
The striatum, or corpus striatum, is a nucleus in the subcortical basal ganglia of the forebrain. The striatum is a critical component of the motor and reward systems; receives glutamatergic and dopaminergic inputs from different sources; and serves as the primary input to the rest of the basal ganglia.

Dopamine is a neuromodulatory molecule that plays several important roles in cells. It is an organic chemical of the catecholamine and phenethylamine families. Dopamine constitutes about 80% of the catecholamine content in the brain. It is an amine synthesized by removing a carboxyl group from a molecule of its precursor chemical, L-DOPA, which is synthesized in the brain and kidneys. Dopamine is also synthesized in plants and most animals. In the brain, dopamine functions as a neurotransmitter—a chemical released by neurons to send signals to other nerve cells. Neurotransmitters are synthesized in specific regions of the brain, but affect many regions systemically. The brain includes several distinct dopamine pathways, one of which plays a major role in the motivational component of reward-motivated behavior. The anticipation of most types of rewards increases the level of dopamine in the brain, and many addictive drugs increase dopamine release or block its reuptake into neurons following release. Other brain dopamine pathways are involved in motor control and in controlling the release of various hormones. These pathways and cell groups form a dopamine system which is neuromodulatory.

The nucleus accumbens is a region in the basal forebrain rostral to the preoptic area of the hypothalamus. The nucleus accumbens and the olfactory tubercle collectively form the ventral striatum. The ventral striatum and dorsal striatum collectively form the striatum, which is the main component of the basal ganglia. The dopaminergic neurons of the mesolimbic pathway project onto the GABAergic medium spiny neurons of the nucleus accumbens and olfactory tubercle. Each cerebral hemisphere has its own nucleus accumbens, which can be divided into two structures: the nucleus accumbens core and the nucleus accumbens shell. These substructures have different morphology and functions.

Dopaminergic pathways in the human brain are involved in both physiological and behavioral processes including movement, cognition, executive functions, reward, motivation, and neuroendocrine control. Each pathway is a set of projection neurons, consisting of individual dopaminergic neurons.

The nigrostriatal pathway is a bilateral dopaminergic pathway in the brain that connects the substantia nigra pars compacta (SNc) in the midbrain with the dorsal striatum in the forebrain. It is one of the four major dopamine pathways in the brain, and is critical in the production of movement as part of a system called the basal ganglia motor loop. Dopaminergic neurons of this pathway release dopamine from axon terminals that synapse onto GABAergic medium spiny neurons (MSNs), also known as spiny projection neurons (SPNs), located in the striatum.

The ventral tegmental area (VTA), also known as the ventral tegmental area of Tsai, or simply ventral tegmentum, is a group of neurons located close to the midline on the floor of the midbrain. The VTA is the origin of the dopaminergic cell bodies of the mesocorticolimbic dopamine system and other dopamine pathways; it is widely implicated in the drug and natural reward circuitry of the brain. The VTA plays an important role in a number of processes, including reward cognition and orgasm, among others, as well as several psychiatric disorders. Neurons in the VTA project to numerous areas of the brain, ranging from the prefrontal cortex to the caudal brainstem and several regions in between.
Motivational salience is a cognitive process and a form of attention that motivates or propels an individual's behavior towards or away from a particular object, perceived event or outcome. Motivational salience regulates the intensity of behaviors that facilitate the attainment of a particular goal, the amount of time and energy that an individual is willing to expend to attain a particular goal, and the amount of risk that an individual is willing to accept while working to attain a particular goal.
Neuropharmacology is the study of how drugs affect function in the nervous system, and the neural mechanisms through which they influence behavior. There are two main branches of neuropharmacology: behavioral and molecular. Behavioral neuropharmacology focuses on the study of how drugs affect human behavior (neuropsychopharmacology), including the study of how drug dependence and addiction affect the human brain. Molecular neuropharmacology involves the study of neurons and their neurochemical interactions, with the overall goal of developing drugs that have beneficial effects on neurological function. Both of these fields are closely connected, since both are concerned with the interactions of neurotransmitters, neuropeptides, neurohormones, neuromodulators, enzymes, second messengers, co-transporters, ion channels, and receptor proteins in the central and peripheral nervous systems. Studying these interactions, researchers are developing drugs to treat many different neurological disorders, including pain, neurodegenerative diseases such as Parkinson's disease and Alzheimer's disease, psychological disorders, addiction, and many others.
According to proponents of the concept, sexual addiction, also known as sex addiction, is a state characterized by compulsive participation or engagement in sexual activity, particularly sexual intercourse, despite negative consequences. The concept is contentious; neither of the two major mainstream medical categorization systems recognise sex addiction as a real medical condition, instead categorizing such behavior under labels such as compulsive sexual behavior.
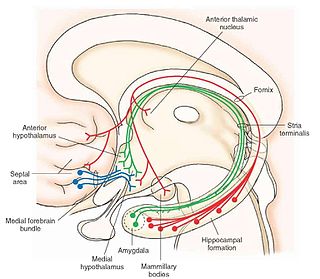
The medial forebrain bundle (MFB) is a neural pathway containing fibers from the basal olfactory regions, the periamygdaloid region and the septal nuclei, as well as fibers from brainstem regions, including the ventral tegmental area and nigrostriatal pathway.

Medium spiny neurons (MSNs), also known as spiny projection neurons (SPNs), are a special type of GABAergic inhibitory cell representing 95% of neurons within the human striatum, a basal ganglia structure. Medium spiny neurons have two primary phenotypes : D1-type MSNs of the direct pathway and D2-type MSNs of the indirect pathway. Most striatal MSNs contain only D1-type or D2-type dopamine receptors, but a subpopulation of MSNs exhibit both phenotypes.

The reward system is a group of neural structures responsible for incentive salience, associative learning, and positively-valenced emotions, particularly ones involving pleasure as a core component. Reward is the attractive and motivational property of a stimulus that induces appetitive behavior, also known as approach behavior, and consummatory behavior. A rewarding stimulus has been described as "any stimulus, object, event, activity, or situation that has the potential to make us approach and consume it is by definition a reward". In operant conditioning, rewarding stimuli function as positive reinforcers; however, the converse statement also holds true: positive reinforcers are rewarding.

Methamphetamine is a potent central nervous system (CNS) stimulant that is mainly used as a recreational drug and less commonly as a second-line treatment for attention deficit hyperactivity disorder and obesity. Methamphetamine was discovered in 1893 and exists as two enantiomers: levo-methamphetamine and dextro-methamphetamine. Methamphetamine properly refers to a specific chemical substance, the racemic free base, which is an equal mixture of levomethamphetamine and dextromethamphetamine in their pure amine forms, but the hydrochloride salt, commonly called crystal meth, is widely used. Methamphetamine is rarely prescribed over concerns involving human neurotoxicity and potential for recreational use as an aphrodisiac and euphoriant, among other concerns, as well as the availability of safer substitute drugs with comparable treatment efficacy such as Adderall and Vyvanse. Dextromethamphetamine is a stronger CNS stimulant than levomethamphetamine.

Protein fosB, also known as FosB and G0/G1 switch regulatory protein 3 (G0S3), is a protein that in humans is encoded by the FBJ murine osteosarcoma viral oncogene homolog B (FOSB) gene.
Behavioral addiction is a form of addiction that involves a compulsion to engage in a rewarding non-substance-related behavior – sometimes called a natural reward – despite any negative consequences to the person's physical, mental, social or financial well-being. Addiction canonically refers to substance abuse; however, the term's connotation has been expanded to include behaviors that may lead to a reward since the 1990s. A gene transcription factor known as ΔFosB has been identified as a necessary common factor involved in both behavioral and drug addictions, which are associated with the same set of neural adaptations in the reward system.
The ventral pallidum (VP) is a structure within the basal ganglia of the brain. It is an output nucleus whose fibres project to thalamic nuclei, such as the ventral anterior nucleus, the ventral lateral nucleus, and the medial dorsal nucleus. The VP is a core component of the reward system which forms part of the limbic loop of the basal ganglia, a pathway involved in the regulation of motivational salience, behavior, and emotions. It is involved in addiction.

Addiction is generally a neuropsychological disorder defining pervasive and intense urge to engage in maladaptive behaviors providing immediate sensory rewards, despite their harmful consequences. Dependence is generally an addiction that can involve withdrawal issues. Addictive disorder is a category of mental disorders defining important intensities of addictions or dependences, which induce functional disabilities. There are no agreed definitions on these terms – see section on 'definitions'.
Cocaine addiction is the compulsive use of cocaine despite adverse consequences. It arises through epigenetic modification and transcriptional regulation of genes in the nucleus accumbens.
Addiction is a state characterized by compulsive engagement in rewarding stimuli, despite adverse consequences. The process of developing an addiction occurs through instrumental learning, which is otherwise known as operant conditioning.
The rostromedial tegmental nucleus (RMTg), also known as the tail of the ventral tegmental area (tVTA), is a GABAergic nucleus which functions as a "master brake" for the midbrain dopamine system. This region was discovered by the researchers, M. Barrot, J.Kaufling and T. Jhou. It is poorly differentiated from the rest of the ventral tegmental area (VTA) and possesses robust functional and structural links to the dopamine pathways. Notably, both acute and chronic exposure to psychostimulants have been shown to induce FosB and ΔFosB expression in the RMTg; no other drug type has been shown to induce these proteins in the RMTg.












