Nibrin, also known as NBN or NBS1, is a protein which in humans is encoded by the NBN gene. [5] [6] [7]
Nibrin, also known as NBN or NBS1, is a protein which in humans is encoded by the NBN gene. [5] [6] [7]
Nibrin is a protein associated with the repair of double strand breaks (DSBs) which pose serious damage to a genome. It is a 754 amino acid protein identified as a member of the NBS1/hMre11/RAD50(N/M/R, more commonly referred to as MRN) double strand DNA break repair complex. [8] This complex recognizes DNA damage and rapidly relocates to DSB sites and forms nuclear foci. It also has a role in regulation of N/M/R (MRN) protein complex activity which includes end-processing of both physiological and mutagenic DNA double strand breaks (DSBs). [9]
Cellular response is performed by damage sensors, effectors of lesion repair and signal transduction. The central role is carried out by ataxia telangiectasia mutated (ATM) by activating the DSB signaling cascade, phosphorylating downstream substrates such as histone H2AX and NBS1. NBS1 relocates to DSB sites by interaction of FHA/BRCT domains with phosphorylated histone H2AX. Once it interacts with nibrin c-terminal hMre11-binding domain, hMre11 and hRad50 relocate from the cytoplasm to the nucleus then to sites of DSBs. They finally relocate to N/M/R where they form the foci at the site of damage. [10]
DSBs occur during V(D)J recombination during early B and T cell development. This is at the point when the cells of the immune system are developing and the DSBs affect the development of lymphoid cells. DSBs also occur in immunoglobulin class switch in mature B cells. [9] More frequently, however, DSBs are caused by mutagenic agents like radiomimetic chemicals and ionizing radiation(IR).
As mentioned, DSBs cause extreme damage to DNA. Mutations that cause defective repair of DSBs tend to accumulate un-repaired DSBs. One such mutation is associated with Nijmegen breakage syndrome (NBS), a radiation hyper-sensitive disease. [11] It is a rare inherited autosomal recessive condition of chrosomal instability. It has been linked to mutations within exons 6–10 in the NBS1 gene which results in a truncated protein. [9] Characteristics of NBS include microcephaly, cranial characteristics, growth retardation, impaired sexual maturation, immunodeficiency/recurring infections and a predisposition to cancer. This predisposition to cancer may be linked to the DSBs occurring at the development of lymphoid cells.
Two adult siblings, both heterozygous for two particular NBS1 nonsense mutations displayed cellular sensitivity to radiation, chromosome instability and fertility defects, but not the developmental defects that are typically found in other NBS patients. [12] These individuals appear to be primarily defective in homologous recombination, a process that accurately repairs double-strand breaks, both in somatic cells and during meiosis.
Orthologs of NBS1 have been studied in mice [13] and the plant arabidopsis. [14] NBS1 mutant mice display cellular radiation sensitivity and female mice are sterile due to oogenesis failure. [13] Studies of NBS1 mutants in Arabidopsis revealed that NBS1 has a role in recombination during early stages of meiosis. [14]
NBS1 has a role in microhomology-mediated end joining (MMEJ) repair of double strand breaks. It is one of 6 enzymes required for this error prone DNA repair pathway. [15] NBS1 is often over-expressed in prostate cancer, [16] in liver cancer, [17] in esophageal squamous cell carcinoma, [18] in non-small cell lung carcinoma, hepatoma, and esophageal cancer, [19] in head and neck cancer, [20] and in squamous cell carcinoma of the oral cavity. [21]
Cancers are very often deficient in expression of one or more DNA repair genes, but over-expression of a DNA repair gene is less usual in cancer. For instance, at least 36 DNA repair enzymes, when mutationally defective in germ line cells, cause increased risk of cancer (hereditary cancer syndromes).[ citation needed ] (Also see DNA repair-deficiency disorder.) Similarly, at least 12 DNA repair genes have frequently been found to be epigenetically repressed in one or more cancers.[ citation needed ] (See also Epigenetically reduced DNA repair and cancer.) Ordinarily, deficient expression of a DNA repair enzyme results in increased un-repaired DNA damages which, through replication errors (translesion synthesis), lead to mutations and cancer. However, NBS1 mediated MMEJ repair is highly inaccurate, so in this case, over-expression, rather than under-expression, apparently leads to cancer.
HSV-1 infects more than 90% of adults over the age of 50. Alphaherpesviruses alone can cause the host to have mild symptoms, but these viruses can be associated with severe disease when they are transferred to a new species. Humans can even pass and also get an HSV-1 infection from other primate species. However, because of evolutionary differences between primate species, only some species can pass HSV-1 in an interspecies interaction. Also, though HSV-1 transmission from humans to other species primates can occur, there is no known sustained transmission chains that have resulted from constant transmission. A study found that Nbs1 is the most diverged in DNA sequence in the MRN complex between different primate species and that there is a high degree of species specificity, causing variability in promotion of the HSV-1 life cycle. The same study found that Nbs1 interacts with HSV-1's ICP0 proteins in an area of structural disorder of the nibrin. This suggests that in general, viruses commonly interact in intrinsically disordered domains in host proteins. It is possible that there are differences in the mammalian genomes that create unique environments for the viruses. Host proteins that are specific to the species might determine how the viruses must adapt to be able to ignite an infection in a new species. The evolution of increased disorder in nibrin benefits the host in decreasing ICP0 interaction and virus hijack. Nbs1 may not be the only host protein that evolves this way. [22]
HSV-1-infection has been shown to result from the phosphorylation of Nbs1. It has been shown in studies that activation of the MRN complex and ATM biochemical cascade is consistent for a resulting HSV-1 infection. When there is an HSV-1 infection, the nucleus is reorganized causing the formation of RCs (replication compartments) where gene expression and DNA replication occurs. Proteins in the host used for DNA repair and damage response are needed for virus production. ICP8, which is a viral single-strand binding protein, is known to interact with several DNA repair proteins, such as Rad50, Mre11, BRG1, and DNA-PKcs. Ul12 and ICP8 viral proteins function together as a recombinase, possibly showing that while working with the host's recombination factors, work to form a concatemer by stimulating homologous recombination. These proteins may move the MRN complex towards the viral genome so it is able to promote homologous recombination, and to prevent non-homologous recombination as non-homologous recombination can have anti-viral effects. This possibly shows that the reaction between UL12 and MRN regulates the complex in a way that benefits the herpes virus. [23]
Nibrin has been shown to interact with:

DNA repair is a collection of processes by which a cell identifies and corrects damage to the DNA molecules that encode its genome. In human cells, both normal metabolic activities and environmental factors such as radiation can cause DNA damage, resulting in tens of thousands of individual molecular lesions per cell per day. Many of these lesions cause structural damage to the DNA molecule and can alter or eliminate the cell's ability to transcribe the gene that the affected DNA encodes. Other lesions induce potentially harmful mutations in the cell's genome, which affect the survival of its daughter cells after it undergoes mitosis. As a consequence, the DNA repair process is constantly active as it responds to damage in the DNA structure. When normal repair processes fail, and when cellular apoptosis does not occur, irreparable DNA damage may occur. This can eventually lead to malignant tumors, or cancer as per the two-hit hypothesis.

Non-homologous end joining (NHEJ) is a pathway that repairs double-strand breaks in DNA. It is called "non-homologous" because the break ends are directly ligated without the need for a homologous template, in contrast to homology directed repair (HDR), which requires a homologous sequence to guide repair. NHEJ is active in both non-dividing and proliferating cells, while HDR is not readily accessible in non-dividing cells. The term "non-homologous end joining" was coined in 1996 by Moore and Haber.

ATM serine/threonine kinase or Ataxia-telangiectasia mutated, symbol ATM, is a serine/threonine protein kinase that is recruited and activated by DNA double-strand breaks, oxidative stress, topoisomerase cleavage complexes, splicing intermediates, R-loops and in some cases by single-strand DNA breaks. It phosphorylates several key proteins that initiate activation of the DNA damage checkpoint, leading to cell cycle arrest, DNA repair or apoptosis. Several of these targets, including p53, CHK2, BRCA1, NBS1 and H2AX are tumor suppressors.
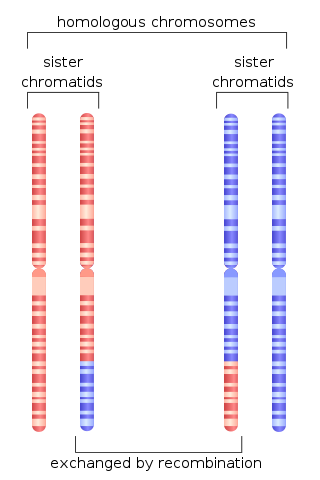
Homologous recombination is a type of genetic recombination in which genetic information is exchanged between two similar or identical molecules of double-stranded or single-stranded nucleic acids.

Werner syndrome ATP-dependent helicase, also known as DNA helicase, RecQ-like type 3, is an enzyme that in humans is encoded by the WRN gene. WRN is a member of the RecQ Helicase family. Helicase enzymes generally unwind and separate double-stranded DNA. These activities are necessary before DNA can be copied in preparation for cell division. Helicase enzymes are also critical for making a blueprint of a gene for protein production, a process called transcription. Further evidence suggests that Werner protein plays a critical role in repairing DNA. Overall, this protein helps maintain the structure and integrity of a person's DNA.

Nijmegen breakage syndrome (NBS) is a rare autosomal recessive congenital disorder causing chromosomal instability, probably as a result of a defect in the double Holliday junction DNA repair mechanism and/or the synthesis dependent strand annealing mechanism for repairing double strand breaks in DNA.

H2A histone family member X is a type of histone protein from the H2A family encoded by the H2AFX gene. An important phosphorylated form is γH2AX (S139), which forms when double-strand breaks appear.

Double-strand break repair protein MRE11 is an enzyme that in humans is encoded by the MRE11 gene. The gene has been designated MRE11A to distinguish it from the pseudogene MRE11B that is nowadays named MRE11P1.
Artemis is a protein that in humans is encoded by the DCLRE1C gene.

DNA repair protein RAD50, also known as RAD50, is a protein that in humans is encoded by the RAD50 gene.

RAD52 homolog , also known as RAD52, is a protein which in humans is encoded by the RAD52 gene.

Mediator of DNA damage checkpoint protein 1 is a 2080 amino acid long protein that in humans is encoded by the MDC1 gene located on the short arm (p) of chromosome 6. MDC1 protein is a regulator of the Intra-S phase and the G2/M cell cycle checkpoints and recruits repair proteins to the site of DNA damage. It is involved in determining cell survival fate in association with tumor suppressor protein p53. This protein also goes by the name Nuclear Factor with BRCT Domain 1 (NFBD1).
SAE2 is a gene in budding yeast, coding for the protein Sae2, which is involved in DNA repair. Sae2 is a part of the homologous recombination process in response to double-strand breaks. It is best characterized in the yeast model organism Saccharomyces cerevisiae. Homologous genes in other organisms include Ctp1 in fission yeast, Com1 in plants, and CtIP in higher eukaryotes including humans.

E3 ubiquitin-protein ligase RNF8 is an enzyme that in humans is encoded by the RNF8 gene. RNF8 has activity both in immune system functions and in DNA repair.
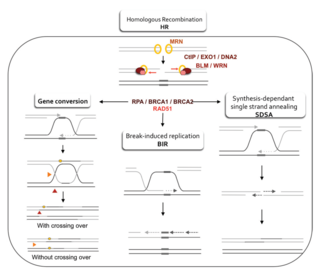
Homology-directed repair (HDR) is a mechanism in cells to repair double-strand DNA lesions. The most common form of HDR is homologous recombination. The HDR mechanism can only be used by the cell when there is a homologous piece of DNA present in the nucleus, mostly in G2 and S phase of the cell cycle. Other examples of homology-directed repair include single-strand annealing and breakage-induced replication. When the homologous DNA is absent, another process called non-homologous end joining (NHEJ) takes place instead.

DNA replication licensing factor MCM8 is a protein that in humans is encoded by the MCM8 gene.
The MRX complex is a heterotrimeric protein complex consisting of Mre11, Rad50, and Xrs2. It is a budding yeast homolog of the mammalian Mre11-Rad50-Nbs1 (MRN) DNA damage repair complex.
The MRN complex is a protein complex consisting of Mre11, Rad50 and Nbs1. In eukaryotes, the MRN/X complex plays an important role in the initial processing of double-strand DNA breaks prior to repair by homologous recombination or non-homologous end joining. The MRN complex binds avidly to double-strand breaks both in vitro and in vivo and may serve to tether broken ends prior to repair by non-homologous end joining or to initiate DNA end resection prior to repair by homologous recombination. The MRN complex also participates in activating the checkpoint kinase ATM in response to DNA damage. Production of short single-strand oligonucleotides by Mre11 endonuclease activity has been implicated in ATM activation by the MRN complex.
DNA damage is an alteration in the chemical structure of DNA, such as a break in a strand of DNA, a nucleobase missing from the backbone of DNA, or a chemically changed base such as 8-OHdG. DNA damage can occur naturally or via environmental factors, but is distinctly different from mutation, although both are types of error in DNA. DNA damage is an abnormal chemical structure in DNA, while a mutation is a change in the sequence of base pairs. DNA damages cause changes in the structure of the genetic material and prevents the replication mechanism from functioning and performing properly. The DNA damage response (DDR) is a complex signal transduction pathway which recognizes when DNA is damaged and initiates the cellular response to the damage.

DNA end resection, also called 5′–3′ degradation, is a biochemical process where the blunt end of a section of double-stranded DNA (dsDNA) is modified by cutting away some nucleotides from the 5' end to produce a 3' single-stranded sequence. The presence of a section of single-stranded DNA (ssDNA) allows the broken end of the DNA to line up accurately with a matching sequence, so that it can be accurately repaired.