Related Research Articles
Photoconductivity is an optical and electrical phenomenon in which a material becomes more electrically conductive due to the absorption of electromagnetic radiation such as visible light, ultraviolet light, infrared light, or gamma radiation.

Molybdenum disulfide is an inorganic compound composed of molybdenum and sulfur. Its chemical formula is MoS
2.

Nanoelectromechanical systems (NEMS) are a class of devices integrating electrical and mechanical functionality on the nanoscale. NEMS form the next logical miniaturization step from so-called microelectromechanical systems, or MEMS devices. NEMS typically integrate transistor-like nanoelectronics with mechanical actuators, pumps, or motors, and may thereby form physical, biological, and chemical sensors. The name derives from typical device dimensions in the nanometer range, leading to low mass, high mechanical resonance frequencies, potentially large quantum mechanical effects such as zero point motion, and a high surface-to-volume ratio useful for surface-based sensing mechanisms. Applications include accelerometers and sensors to detect chemical substances in the air.
Hybrid solar cells combine advantages of both organic and inorganic semiconductors. Hybrid photovoltaics have organic materials that consist of conjugated polymers that absorb light as the donor and transport holes. Inorganic materials in hybrid cells are used as the acceptor and electron transporter in the structure. The hybrid photovoltaic devices have a potential for not only low-cost by roll-to-roll processing but also for scalable solar power conversion.

Nanochemistry is an emerging sub-discipline of the chemical and material sciences that deals with the development of new methods for creating nanoscale materials. The term "nanochemistry" was first used by Ozin in 1992 as 'the uses of chemical synthesis to reproducibly afford nanomaterials from the atom "up", contrary to the nanoengineering and nanophysics approach that operates from the bulk "down"'. Nanochemistry focuses on solid-state chemistry that emphasizes synthesis of building blocks that are dependent on size, surface, shape, and defect properties, rather than the actual production of matter. Atomic and molecular properties mainly deal with the degrees of freedom of atoms in the periodic table. However, nanochemistry introduced other degrees of freedom that controls material's behaviors by transformation into solutions. Nanoscale objects exhibit novel material properties, largely as a consequence of their finite small size. Several chemical modifications on nanometer-scaled structures approve size dependent effects.
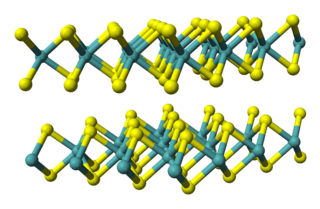
Tungsten disulfide is an inorganic chemical compound composed of tungsten and sulfur with the chemical formula WS2. This compound is part of the group of materials called the transition metal dichalcogenides. It occurs naturally as the rare mineral tungstenite. This material is a component of certain catalysts used for hydrodesulfurization and hydrodenitrification.

Nanocomposite is a multiphase solid material where one of the phases has one, two or three dimensions of less than 100 nanometers (nm) or structures having nano-scale repeat distances between the different phases that make up the material.
ApNano Materials is a nanotechnology company, wholly owned and operated by Nanotech Industrial Solutions (NIS) with R&D lab, manufacturing, blending and packaging facilities in Avenel, New Jersey, United States, and Yavne, Israel. NIS is the only company in the world with an exclusive license to manufacture inorganic fullerene-like tungsten disulfide (IF-WS2) submicron (nanosized) spherical particles on a commercial scale with the patent from the Weizmann Institute. These inorganic fullerene-like tungsten disulfide-based nanomaterials opened up new possibilities for developing extreme performance industrial lubricants, coatings, and polymer composites.

Carbon nanotubes (CNTs) are cylinders of one or more layers of graphene (lattice). Diameters of single-walled carbon nanotubes (SWNTs) and multi-walled carbon nanotubes (MWNTs) are typically 0.8 to 2 nm and 5 to 20 nm, respectively, although MWNT diameters can exceed 100 nm. CNT lengths range from less than 100 nm to 0.5 m.
Molecular wires are molecular chains that conduct electric current. They are the proposed building blocks for molecular electronic devices. Their typical diameters are less than three nanometers, while their lengths may be macroscopic, extending to centimeters or more.
Polymer nanocomposites (PNC) consist of a polymer or copolymer having nanoparticles or nanofillers dispersed in the polymer matrix. These may be of different shape, but at least one dimension must be in the range of 1–50 nm. These PNC's belong to the category of multi-phase systems that consume nearly 95% of plastics production. These systems require controlled mixing/compounding, stabilization of the achieved dispersion, orientation of the dispersed phase, and the compounding strategies for all MPS, including PNC, are similar. Alternatively, polymer can be infiltrated into 1D, 2D, 3D preform creating high content polymer nanocomposites.

Germanium disulfide or Germanium(IV) sulfide is the inorganic compound with the formula GeS2. It is a white high-melting crystalline solid. The compound is a 3-dimensional polymer, in contrast to silicon disulfide, which is a one-dimensional polymer. The Ge-S distance is 2.19 Å.

Transparent conducting films (TCFs) are thin films of optically transparent and electrically conductive material. They are an important component in a number of electronic devices including liquid-crystal displays, OLEDs, touchscreens and photovoltaics. While indium tin oxide (ITO) is the most widely used, alternatives include wider-spectrum transparent conductive oxides (TCOs), conductive polymers, metal grids and random metallic networks, carbon nanotubes (CNT), graphene, nanowire meshes and ultra thin metal films.

Titanium disulfide is an inorganic compound with the formula TiS2. A golden yellow solid with high electrical conductivity, it belongs to a group of compounds called transition metal dichalcogenides, which consist of the stoichiometry ME2. TiS2 has been employed as a cathode material in rechargeable batteries.
Carbon nanotube supported catalyst is a novel supported catalyst, using carbon nanotubes as the support instead of the conventional alumina or silicon support. The exceptional physical properties of carbon nanotubes (CNTs) such as large specific surface areas, excellent electron conductivity incorporated with the good chemical inertness, and relatively high oxidation stability makes it a promising support material for heterogeneous catalysis.
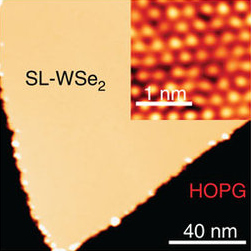
Tungsten diselenide is an inorganic compound with the formula WSe2. The compound adopts a hexagonal crystalline structure similar to molybdenum disulfide. The tungsten atoms are covalently bonded to six selenium ligands in a trigonal prismatic coordination sphere while each selenium is bonded to three tungsten atoms in a pyramidal geometry. The tungsten–selenium bond has a length of 0.2526 nm, and the distance between selenium atoms is 0.334 nm. It is a well studied example of a layered material. The layers stack together via van der Waals interactions. WSe2 is a very stable semiconductor in the group-VI transition metal dichalcogenides.

Transition-metal dichalcogenide (TMD or TMDC) monolayers are atomically thin semiconductors of the type MX2, with M a transition-metal atom (Mo, W, etc.) and X a chalcogen atom (S, Se, or Te). One layer of M atoms is sandwiched between two layers of X atoms. They are part of the large family of so-called 2D materials, named so to emphasize their extraordinary thinness. For example, a MoS2 monolayer is only 6.5 Å thick. The key feature of these materials is the interaction of large atoms in the 2D structure as compared with first-row transition-metal dichalcogenides, e.g., WTe2 exhibits anomalous giant magnetoresistance and superconductivity.
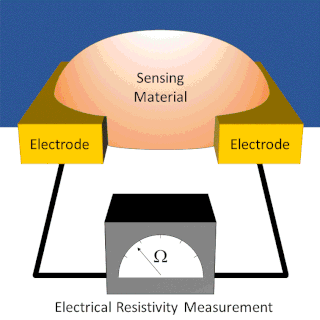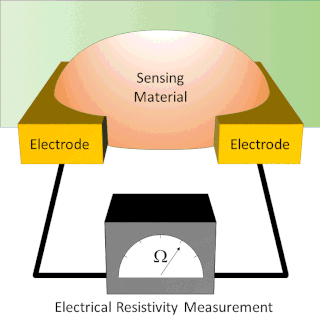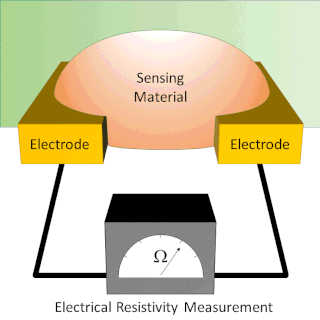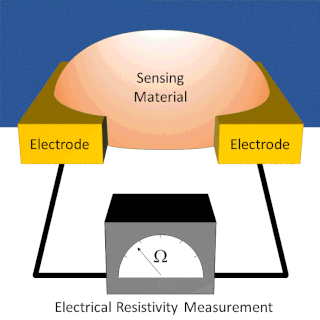
A chemiresistor is a material that changes its electrical resistance in response to changes in the nearby chemical environment. Chemiresistors are a class of chemical sensors that rely on the direct chemical interaction between the sensing material and the analyte. The sensing material and the analyte can interact by covalent bonding, hydrogen bonding, or molecular recognition. Several different materials have chemiresistor properties: metal-oxide semiconductors, some conductive polymers, and nanomaterials like graphene, carbon nanotubes and nanoparticles. Typically these materials are used as partially selective sensors in devices like electronic tongues or electronic noses.
In materials science, a polymer matrix composite (PMC) is a composite material composed of a variety of short or continuous fibers bound together by a matrix of organic polymers. PMCs are designed to transfer loads between fibers of a matrix. Some of the advantages with PMCs include their light weight, high resistance to abrasion and corrosion, and high stiffness and strength along the direction of their reinforcements.
Mixed-anion compounds, heteroanionic materials or mixed-anion materials are chemical compounds containing cations and more than one kind of anion. The compounds contain a single phase, rather than just a mixture.
References
- ↑ Ahmadi A, Beheshtian J, Hadipour NL (2011). "Interaction of NH3 with aluminum nitride nanotube: Electrostatic vs. covalent". Physica E: Low-dimensional Systems and Nanostructures. 43 (9): 1717–1719. Bibcode:2011PhyE...43.1717A. doi:10.1016/j.physe.2011.05.029.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Beheshtian J, Baei MT, Peyghan AA, Bagheri Z (2012). "Electronic sensor for sulfide dioxide based on AlN nanotubes: a computational study". J Mol Model. 18 (10): 4745–4750. doi:10.1007/s00894-012-1476-2. PMID 22678082. S2CID 36157701.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ↑ Harris, P.F.J. (2002). Carbon nanotubes and related structures (1st ed.). Cambridge University Press. pp. 213–32. ISBN 978-0-521-00533-3.
- ↑ Pauling L (1930). "The Structure Of The Chlorites". Proc. Natl. Acad. Sci. U.S.A. 16 (9): 578–82. Bibcode:1930PNAS...16..578P. doi: 10.1073/pnas.16.9.578 . PMC 526695 . PMID 16587609.
- ↑ Bates; et al. (1950). "Tubular Crystals of Chrysotile Asbestos". Science. 111 (2889): 512–513. Bibcode:1950Sci...111..512B. doi:10.1126/science.111.2889.512. PMID 15418177.
- ↑ Cradwick; et al. (1972). "Imogolite, a Hydrated Aluminium Silicate of Tubular Structure". Nature Physical Science. 240 (104): 187–189. Bibcode:1972NPhS..240..187C. doi:10.1038/physci240187a0.
- 1 2 Tenne R, Margulis L, Genut M, Hodes G (1992). "Polyhedral and cylindrical structures of tungsten disulphide". Nature. 360 (6403): 444–446. Bibcode:1992Natur.360..444T. doi:10.1038/360444a0. S2CID 4309310.
- ↑ Peter J. F. Harris; Peter John Frederich Harris (12 November 2001). Carbon nanotubes and related structures: new materials for the twenty-first century. Cambridge University Press. pp. 213–. ISBN 978-0-521-00533-3 . Retrieved 3 November 2011.
- ↑ R. Tenne (2002). "Fullerene-like materials and nanotubes from inorganic compounds with a layered (2-D) structure". Colloids and Surfaces A. 208 (1–3): 83–92. doi:10.1016/S0927-7757(02)00104-8.
- ↑ A. Zak; L. Sallacan Ecker; N. Fleischer; R. Tenne (2011). "Large-Scale Synthesis of WS2 Multiwall Nanotubes: Update". J. Sensors& Transducers. 12 (10): 1–10.
- ↑ G. Radovsky; R. Popovitz-Biro; M. Staiger; K. Gartsman; C. Thomsen; T. Lorenz; G. Seifert; R. Tenne (2011). "Synthesis of Copious Amounts of SnS2 and SnS2/SnS Nanotubes with Ordered Superstructures". Angew. Chem. Int. Ed. 50 (51): 12316–12320. doi:10.1002/anie.201104520. PMID 22038979.
- ↑ Muhammad, Ibrahim D.; Awang, Mokhtar. "Review of the Geometric Dimensions of Cubic Zirconia Nanotubes". www.academia.edu. Retrieved 2016-02-20.
- ↑ S.I. Na; S.S. Kim; W. K. Hong; J.W. Park; J. Jo; Y.C. Nah; T. Lee; D.Y. Kim (2008). "Fabrication of TiO2 nanotubes by using electrodeposited ZnO nanorod template and their application to hybrid solar cells". Electrochimica Acta. 53 (5): 2560–2566. doi:10.1016/j.electacta.2007.10.041.
- ↑ A. Kis; D. Mihailovic; M. Remskar; A. Mrzel; A. Jesih; I. Piwonski; A. J. Kulik; W. Benoit; L. Forro (2003). "Shear and Young's Moduli of MoS2 Nanotube Ropes". Advanced Materials. 15 (9): 733–736. Bibcode:2003AdM....15..733K. doi:10.1002/adma.200304549. S2CID 136420653.
- ↑ Yang, Dachi; Meng, Guowen; Zhang, Shuyuan; Hao, Yufeng; An, Xiaohong; Wei, Qing; Ye, Min; Zhang, Lide (2007). "Electrochemical synthesis of metal and semimetal nanotube–nanowire heterojunctions and their electronic transport properties". Chem. Commun. (17): 1733–1735. doi:10.1039/B614147A. PMID 17457424. S2CID 2534957.
- ↑ M. Krause; A. Mucklich; A. Zak; G. Seifert; S. Gemming (2011). "High resolution TEM study of WS2 nanotubes". Physica Status Solidi B. 248 (11): 2716–2719. Bibcode:2011PSSBR.248.2716K. doi: 10.1002/pssb.201100076 . S2CID 121701880.
- ↑ Y. Q. Zhu; H. W. Kroto (2003). "Shock-Wave Resistance of WS2 Nanotubes". J. Am. Chem. Soc. 125 (5): 1329–1333. doi:10.1021/ja021208i. PMID 12553835.
- ↑ ApNano Materials Announces Major Breakthrough in Industrial Nanotube Production for Bullet Proof Vests. Nanotech now
- ↑ "Inorganic Menagerie. Unusual properties of nanotubes made from inorganic materials offer intriguing possibilities for applications". Chemical and Engineering News. 83 (35): 30–33. August 2005. doi:10.1021/cen-v083n040.p030.
- ↑ S.J. Chin; P. Hornsby; D. Vengust; D. Mihailović; J. Mitra; P. Dawson; T. McNally (2011). "Composites of poly(ε-caprolactone) and Mo6S3I6 Nanowires". Polymers for Advanced Technologies. 23 (2): 149–160. doi:10.1002/pat.1838.
- ↑ E. Zohar; S. Baruch; M. Shneider; H. Dodiu; S. Kenig; D.H. Wagner; A. Zak; A. Moshkovith; L. Rapoport; R. Tenne (2011). "The Mechanical and Tribological Properties of Epoxy Nanocomposites with WS2 Nanotubes". Sensors & Transducers Journal. 12 (Special Issue): 53–65.
- ↑ C. S. Reddy; A. Zak; E. Zussman (2011). "WS2 nanotubes embedded in PMMA nanofibers as energy absorptive material". J. Mater. Chem. 21 (40): 16086–16093. doi:10.1039/C1JM12700D.
- ↑ R. Kreizman; A. N. Enyashin; F. L. Deepak; A. Albu-Yaron; R. Popovitz-Biro; G. Seifert; R. Tenne (2010). "Synthesis of Core-Shell Inorganic Nanotubes". Adv. Funct. Mater. 20 (15): 2459–2468. doi:10.1002/adfm.201000490. S2CID 136725896.
- ↑ Lalwani G, Henslee AM, Farshid B, Parmar P, Lin L, Qin YX, Kasper FK, Mikos AG, Sitharaman B (2013). "Tungsten disulfide nanotubes reinforced biodegradable polymers for bone tissue engineering". Acta Biomater. 9 (9): 8365–73. doi:10.1016/j.actbio.2013.05.018. PMC 3732565 . PMID 23727293.