
Cardiac arrest is a sudden loss of blood flow resulting from the failure of the heart to pump effectively. Signs include loss of consciousness and abnormal or absent breathing. Some individuals may experience chest pain, shortness of breath, or nausea immediately before entering cardiac arrest. Radiating pain to one arm is a common symptom, as is long term malaise and "general weakness of heart". If not treated within minutes, it typically leads to death.

A cardiac pacemaker, is a medical device that generates electrical impulses delivered by electrodes to cause the heart muscle chambers to contract and therefore pump blood; by doing so this device replaces and/or regulates the function of the electrical conduction system of the heart.

Defibrillation is a treatment for life-threatening cardiac dysrhythmias, specifically ventricular fibrillation (VF) and non-perfusing ventricular tachycardia (VT). A defibrillator delivers a dose of electric current to the heart. Although not fully understood, this process depolarizes a large amount of the heart muscle, ending the dysrhythmia. Subsequently, the body's natural pacemaker in the sinoatrial node of the heart is able to re-establish normal sinus rhythm. A heart which is in asystole (flatline) cannot be restarted by a defibrillator, but would be treated by cardiopulmonary resuscitation (CPR).

Brugada syndrome (BrS) is a genetic disorder in which the electrical activity within the heart is abnormal. It increases the risk of abnormal heart rhythms and sudden cardiac death. Those affected may have episodes of passing out. The abnormal heart rhythms seen in those with Brugada syndrome often occur at rest. They may be triggered by a fever.

Long QT syndrome (LQTS) is a condition in which repolarization of the heart after a heartbeat is affected. It results in an increased risk of an irregular heartbeat which can result in fainting, drowning, seizures, or sudden death. These episodes can be triggered by exercise or stress. Some rare forms of LQTS are associated with other symptoms and signs including deafness and periods of muscle weakness.

An implantable cardioverter-defibrillator (ICD) or automated implantable cardioverter defibrillator (AICD) is a device implantable inside the body, able to perform cardioversion, defibrillation, and pacing of the heart. The device is therefore capable of correcting most life-threatening cardiac arrhythmias. The ICD is the first-line treatment and prophylactic therapy for patients at risk for sudden cardiac death due to ventricular fibrillation and ventricular tachycardia. Current devices can be programmed to detect abnormal heart rhythms and deliver therapy via programmable antitachycardia pacing in addition to low-energy and high-energy shocks.

Arrhythmogenic cardiomyopathy (ACM), arrhythmogenic right ventricular dysplasia (ARVD), or arrhythmogenic right ventricular cardiomyopathy (ARVC), is an inherited heart disease.
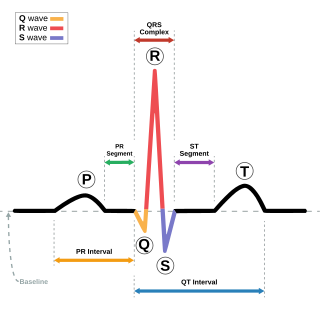
Short QT syndrome (SQT) is a very rare genetic disease of the electrical system of the heart, and is associated with an increased risk of abnormal heart rhythms and sudden cardiac death. The syndrome gets its name from a characteristic feature seen on an electrocardiogram (ECG) – a shortening of the QT interval. It is caused by mutations in genes encoding ion channels that shorten the cardiac action potential, and appears to be inherited in an autosomal dominant pattern. The condition is diagnosed using a 12-lead ECG. Short QT syndrome can be treated using an implantable cardioverter-defibrillator or medications including quinidine. Short QT syndrome was first described in 2000, and the first genetic mutation associated with the condition was identified in 2004.

Ventricular tachycardia is a type of regular, fast heart rate that arises from improper electrical activity in the ventricles of the heart. Although a few seconds may not result in problems, longer periods are dangerous; and multiple episodes over a short period of time is referred to as an Electrical Storm. Short periods may occur without symptoms, or present with lightheadedness, palpitations, or chest pain. Ventricular tachycardia may result in ventricular fibrillation and turn into cardiac arrest. It is found initially in about 7% of people in cardiac arrest.

Disopyramide is an antiarrhythmic medication used in the treatment of ventricular tachycardia. It is a sodium channel blocker and therefore classified as a Class 1a anti-arrhythmic agent. Disopyramide has a negative inotropic effect on the ventricular myocardium, significantly decreasing the contractility. Disopyramide also has an anticholinergic effect on the heart which accounts for many adverse side effects. Disopyramide is available in both oral and intravenous forms, and has a low degree of toxicity.
Clinical cardiac electrophysiology, is a branch of the medical specialty of cardiology and is concerned with the study and treatment of rhythm disorders of the heart. Cardiologists with expertise in this area are usually referred to as electrophysiologists. Electrophysiologists are trained in the mechanism, function, and performance of the electrical activities of the heart. Electrophysiologists work closely with other cardiologists and cardiac surgeons to assist or guide therapy for heart rhythm disturbances (arrhythmias). They are trained to perform interventional and surgical procedures to treat cardiac arrhythmia.

Catecholaminergic polymorphic ventricular tachycardia (CPVT) is an inherited genetic disorder that predisposes those affected to potentially life-threatening abnormal heart rhythms or arrhythmias. The arrhythmias seen in CPVT typically occur during exercise or at times of emotional stress, and classically take the form of bidirectional ventricular tachycardia or ventricular fibrillation. Those affected may be asymptomatic, but they may also experience blackouts or even sudden cardiac death.

Cardiac resynchronisation therapy is the insertion of electrodes in the left and right ventricles of the heart, as well as on occasion the right atrium, to treat heart failure by coordinating the function of the left and right ventricles via a pacemaker, a small device inserted into the interior chest wall.
Management of heart failure requires a multimodal approach. It involves a combination of lifestyle modifications, medications, and possibly the use of devices or surgery.

A wearable cardioverter defibrillator (WCD) is a device worn by patients who are at risk for sudden cardiac arrest (SCA). A WCD allows physicians time to assess for their patient's arrhythmic risk and make appropriate plans.
Boxer cardiomyopathy is a disease of the myocardium primarily affecting Boxer dogs. It is characterized by the development of ventricular tachyarrhythmias, resulting in syncope and sudden cardiac death. Myocardial failure and congestive heart failure are uncommon manifestations of the disease.
Cameron Health was a medical device developer based in San Clemente, California, USA. Cameron Health had its European office, Cameron Health BV, in Arnhem, The Netherlands. The privately held company's focus was on a new generation of minimally invasive implantable cardioverter-defibrillator (ICD) which they called a Subcutaneous Implantable Defibrillator (S-ICD). Cameron Health's approach avoided implanting transvenous leads into the heart, which had been the usual procedure for cardiac devices. Instead, the Cameron ICD was entirely implanted outside the thoracic wall.
Cardiac Contractility Modulation is a therapy which is intended for the treatment of patients with moderate to severe heart failure with symptoms despite optimal medical therapy who can benefit from an improvement in cardiac output. The short- and long-term use of this therapy enhances the strength of ventricular contraction and therefore the heart's pumping capacity by modulating (adjusting) the myocardial contractility. This is provided by a pacemaker-like device that applies non-excitatory electrical signals adjusted to and synchronized with the electrical action in the cardiac cycle.

Yaariv Khaykin is a Canadian cardiologist and a clinical researcher in the area of electrophysiology. He is the director of the Newmarket Electrophysiology Research Group at the Southlake Regional Health Centre. He has published research into complex ablation and pioneered cardiac ablation methods.
Defibrillation threshold indicates the minimum amount of energy needed to return normal rhythm to a heart that is beating in a cardiac dysrhythmia. Typical examples are the minimum amount of energy, expressed in joules, delivered by external defibrillator paddles or pads, required to break atrial fibrillation and restore normal sinus rhythm. Other common scenarios are restoring normal rhythm from atrial flutter, ventricular tachycardia or ventricular fibrillation. The defibrillation threshold ranking in these settings, from lowest to highest, would be, in order, ventricular tachycardia, atrial flutter, atrial fibrillation, ventricular fibrillation. The highest amount of energy that an external defibrillator can deliver at the present time is 360 joules biphasic. In clinical practice, the real threshold can be approximated but not exactly established, since the defibrillating shock can be delivered only once. Certain medications, in particular sotalol, tend to lower such threshold, while others, such as amiodarone, may increase it.













