
Physical chemistry is the study of macroscopic and microscopic phenomena in chemical systems in terms of the principles, practices, and concepts of physics such as motion, energy, force, time, thermodynamics, quantum chemistry, statistical mechanics, analytical dynamics and chemical equilibria.
In chemistry, photoisomerization is a form of isomerization induced by photoexcitation. Both reversible and irreversible photoisomerizations are known for photoswitchable compounds. The term "photoisomerization" usually, however, refers to a reversible process.
Koopmans' theorem states that in closed-shell Hartree–Fock theory (HF), the first ionization energy of a molecular system is equal to the negative of the orbital energy of the highest occupied molecular orbital (HOMO). This theorem is named after Tjalling Koopmans, who published this result in 1934.

The Journal of Physical Chemistry B is a peer-reviewed scientific journal that covers research on several fields of material chemistry as well as statistical mechanics, thermodynamics, and biophysical chemistry. It has been published weekly since 1997 by the American Chemical Society. According to the Journal Citation Reports, the journal had an impact factor of 3.466 for 2021.
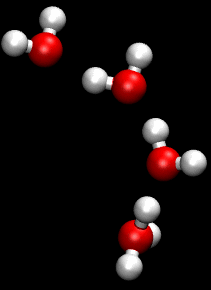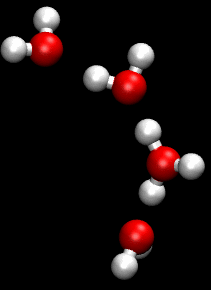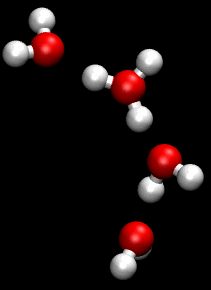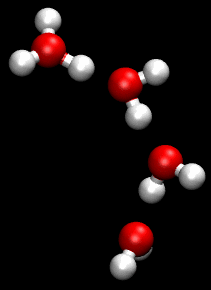
The Grotthuss mechanism is the process by which an 'excess' proton or proton defect diffuses through the hydrogen bond network of water molecules or other hydrogen-bonded liquids through the formation and concomitant cleavage of covalent bonds involving neighboring molecules.

Spartan is a molecular modelling and computational chemistry application from Wavefunction. It contains code for molecular mechanics, semi-empirical methods, ab initio models, density functional models, post-Hartree–Fock models, and thermochemical recipes including G3(MP2) and T1. Quantum chemistry calculations in Spartan are powered by Q-Chem.

Carbon trioxide (CO3) is an unstable oxide of carbon (an oxocarbon). The possible isomers of carbon trioxide include ones with molecular symmetry point groups Cs, D3h, and C2v. The C2v state, consisting of a dioxirane, has been shown to be the ground state of the molecule. Carbon trioxide should not be confused with the stable carbonate ion (CO2−
3).
ReaxFF (for “reactive force field”) is a bond order-based force field developed by Adri van Duin, William A. Goddard, III, and co-workers at the California Institute of Technology. One of its applications is molecular dynamics simulations. Whereas traditional force fields are unable to model chemical reactions because of the requirement of breaking and forming bonds (a force field's functional form depends on having all bonds defined explicitly), ReaxFF eschews explicit bonds in favor of bond orders, which allows for continuous bond formation/breaking. ReaxFF aims to be as general as possible and has been parameterized and tested for hydrocarbon reactions, alkoxysilane gelation, transition-metal-catalyzed nanotube formation, and many advanced material applications such as Li ion batteries, TiO2, polymers, and high-energy materials.

Anna I. Krylov is a Professor of Chemistry at the University of Southern California (USC), working in the field of theoretical and computational quantum chemistry. She is the inventor of the spin-flip method. Krylov is the president of Q-Chem, Inc. and an elected member of the International Academy of Quantum Molecular Science and the Academia Europaea.

In chemistry, a water cluster is a discrete hydrogen bonded assembly or cluster of molecules of water. Many such clusters have been predicted by theoretical models (in silico), and some have been detected experimentally in various contexts such as ice, bulk liquid water, in the gas phase, in dilute mixtures with non-polar solvents, and as water of hydration in crystal lattices. The simplest example is the water dimer (H2O)2.

The Journal of Physical Chemistry C publishes scientific articles reporting research on several subdisciplines of physical chemistry:
Molecular modeling on GPU is the technique of using a graphics processing unit (GPU) for molecular simulations.

Avogadro is a molecule editor and visualizer designed for cross-platform use in computational chemistry, molecular modeling, bioinformatics, materials science, and related areas. It is extensible via a plugin architecture.
Binary compounds of hydrogen are binary chemical compounds containing just hydrogen and one other chemical element. By convention all binary hydrogen compounds are called hydrides even when the hydrogen atom in it is not an anion. These hydrogen compounds can be grouped into several types.

Chlorine peroxide is a molecular compound with formula ClOOCl. Chemically, it is a dimer of the chlorine monoxide radical (ClO·). It is important in the formation of the ozone hole. Chlorine peroxide catalytically converts ozone into oxygen when it is irradiated by ultraviolet light.
Tamar Seideman is the Dow Chemical Company Professor of Chemistry and Professor of Physics at Northwestern University. She specialises in coherence spectroscopies and coherent control in isolated molecules and dissipative media as well as in ultrafast nanoplasmonics, current-driven phenomena in nanoelectronics and mathematical models.

Patricia Ann Thiel was an American chemist and materials scientist who served as a distinguished professor of chemistry at Iowa State University. She was known for her research on atomic-scale structures and processes on solid surfaces.

Majed Chergui is a Swiss and French physicist specialized in ultrafast dynamics of light-induced processes. He is a professor at EPFL, head of the Laboratory of Ultrafast Spectroscopy at EPFL's School of Basic Sciences, and founding director of the Lausanne Centre for Ultrafast Science (LACUS).
Anne B. McCoy is a theoretical chemist. She is the Natt-Lingafelter Professor of Chemistry at the University of Washington, and her research interests include vibrational spectroscopy, hydrogen bonding, and charge-transfer bands.

Alexander I. Boldyrev is a Russian-American computational chemist and R. Gaurth Hansen Professor at Utah State University. Professor Boldyrev is known for his pioneering works on superhalogens, superalkalis, tetracoordinated planar carbon, inorganic double helix, boron and aluminum clusters, and chemical bonding theory, especially aromaticity/antiaromaticity in all-metal structures, and development of the Adaptive Natural Density Partitioning (AdNDP) method.














