The endometrium is the inner epithelial layer, along with its mucous membrane, of the mammalian uterus. It has a basal layer and a functional layer: the basal layer contains stem cells which regenerate the functional layer. The functional layer thickens and then is shed during menstruation in humans and some other mammals, including apes, Old World monkeys, some species of bat, the elephant shrew and the Cairo spiny mouse. In most other mammals, the endometrium is reabsorbed in the estrous cycle. During pregnancy, the glands and blood vessels in the endometrium further increase in size and number. Vascular spaces fuse and become interconnected, forming the placenta, which supplies oxygen and nutrition to the embryo and fetus. The speculated presence of an endometrial microbiota has been argued against.
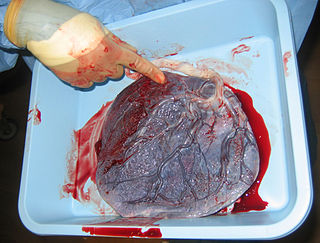
The placenta is a temporary embryonic and later fetal organ that begins developing from the blastocyst shortly after implantation. It plays critical roles in facilitating nutrient, gas and waste exchange between the physically separate maternal and fetal circulations, and is an important endocrine organ, producing hormones that regulate both maternal and fetal physiology during pregnancy. The placenta connects to the fetus via the umbilical cord, and on the opposite aspect to the maternal uterus in a species-dependent manner. In humans, a thin layer of maternal decidual (endometrial) tissue comes away with the placenta when it is expelled from the uterus following birth. Placentas are a defining characteristic of placental mammals, but are also found in marsupials and some non-mammals with varying levels of development.

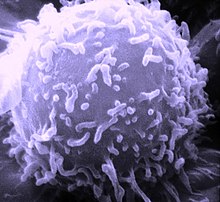
Natural killer cells, also known as NK cells or large granular lymphocytes (LGL), are a type of cytotoxic lymphocyte critical to the innate immune system. They belong to the rapidly expanding family of known innate lymphoid cells (ILC) and represent 5–20% of all circulating lymphocytes in humans. The role of NK cells is analogous to that of cytotoxic T cells in the vertebrate adaptive immune response. NK cells provide rapid responses to virus-infected cell and other intracellular pathogens acting at around 3 days after infection, and respond to tumor formation. Most immune cells detect the antigen presented on major histocompatibility complex (MHC) on infected cell surfaces, but NK cells can recognize and kill stressed cells in the absence of antibodies and MHC, allowing for a much faster immune reaction. They were named "natural killers" because of the notion that they do not require activation to kill cells that are missing "self" markers of MHC class I. This role is especially important because harmful cells that are missing MHC I markers cannot be detected and destroyed by other immune cells, such as T lymphocyte cells.

Eosinophils, sometimes called eosinophiles or, less commonly, acidophils, are a variety of white blood cells and one of the immune system components responsible for combating multicellular parasites and certain infections in vertebrates. Along with mast cells and basophils, they also control mechanisms associated with allergy and asthma. They are granulocytes that develop during hematopoiesis in the bone marrow before migrating into blood, after which they are terminally differentiated and do not multiply. They form about 2 to 3% of white blood cells in the body.

Gestational hypertension or pregnancy-induced hypertension (PIH) is the development of new hypertension in a pregnant woman after 20 weeks' gestation without the presence of protein in the urine or other signs of pre-eclampsia. Gestational hypertension is defined as having a blood pressure greater than 140/90 on two occasions at least 6 hours apart.

The trophoblast is the outer layer of cells of the blastocyst. Trophoblasts are present four days after fertilization in humans. They provide nutrients to the embryo and develop into a large part of the placenta. They form during the first stage of pregnancy and are the first cells to differentiate from the fertilized egg to become extraembryonic structures that do not directly contribute to the embryo. After blastulation, the trophoblast is contiguous with the ectoderm of the embryo and is referred to as the trophectoderm. After the first differentiation, the cells in the human embryo lose their totipotency because they can no longer form a trophoblast. They become pluripotent stem cells.
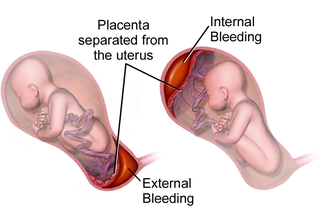
Placental abruption is when the placenta separates early from the uterus, in other words separates before childbirth. It occurs most commonly around 25 weeks of pregnancy. Symptoms may include vaginal bleeding, lower abdominal pain, and dangerously low blood pressure. Complications for the mother can include disseminated intravascular coagulopathy and kidney failure. Complications for the baby can include fetal distress, low birthweight, preterm delivery, and stillbirth.

The decidua is the modified mucosal lining of the uterus that forms every month, in preparation for pregnancy. It is shed off each month when there is no fertilised egg to support. The decidua is under the influence of progesterone. Endometrial cells become highly characteristic. The decidua forms the maternal part of the placenta and remains for the duration of the pregnancy. After birth the decidua is shed together with the placenta.

Placentation refers to the formation, type and structure, or arrangement of the placenta. The function of placentation is to transfer nutrients, respiratory gases, and water from maternal tissue to a growing embryo, and in some instances to remove waste from the embryo. Placentation is best known in live-bearing mammals (theria), but also occurs in some fish, reptiles, amphibians, a diversity of invertebrates, and flowering plants. In vertebrates, placentas have evolved more than 100 times independently, with the majority of these instances occurring in squamate reptiles.

Implantation, also known as nidation is the stage in the embryonic development of mammals in which the blastocyst hatches, attaches, adheres, and invades into the wall of the female's uterus. Implantation is the first stage of gestation, and, when successful, the female is considered to be pregnant. An implanted embryo is detected by the presence of increased levels of human chorionic gonadotropin (hCG) in a pregnancy test. The implanted embryo will receive oxygen and nutrients in order to grow.

Decidualization is a process that results in significant changes to cells of the endometrium in preparation for, and during, pregnancy. This includes morphological and functional changes to endometrial stromal cells (ESCs), the presence of decidual white blood cells (leukocytes), and vascular changes to maternal arteries. The sum of these changes results in the endometrium changing into a structure called the decidua. In humans, the decidua is shed during childbirth.

HLA-G histocompatibility antigen, class I, G, also known as human leukocyte antigen G (HLA-G), is a protein that in humans is encoded by the HLA-G gene.

CD244 also known as 2B4 or SLAMF4 is a protein that in humans is encoded by the CD244 gene.

Natural cytotoxicity triggering receptor 3 is a protein that in humans is encoded by the NCR3 gene. NCR3 has also been designated as CD337 and as NKp30. NCR3 belongs to the family of NCR membrane receptors together with NCR1 (NKp46) and NCR2 (NKp44).
Reproductive immunology refers to a field of medicine that studies interactions between the immune system and components related to the reproductive system, such as maternal immune tolerance towards the fetus, or immunological interactions across the blood-testis barrier. The concept has been used by fertility clinics to explain fertility problems, recurrent miscarriages and pregnancy complications observed when this state of immunological tolerance is not successfully achieved. Immunological therapy is a method for treating many cases of previously "unexplained infertility" or recurrent miscarriage.
Immune tolerance in pregnancy or maternal immune tolerance is the immune tolerance shown towards the fetus and placenta during pregnancy. This tolerance counters the immune response that would normally result in the rejection of something foreign in the body, as can happen in cases of spontaneous abortion. It is studied within the field of reproductive immunology.

NKG2D is an activating receptor (transmembrane protein) belonging to the NKG2 family of C-type lectin-like receptors. NKG2D is encoded by KLRK1 (killer cell lectin like receptor K1) gene which is located in the NK-gene complex (NKC) situated on chromosome 6 in mice and chromosome 12 in humans. In mice, it is expressed by NK cells, NK1.1+ T cells, γδ T cells, activated CD8+ αβ T cells and activated macrophages. In humans, it is expressed by NK cells, γδ T cells and CD8+ αβ T cells. NKG2D recognizes induced-self proteins from MIC and RAET1/ULBP families which appear on the surface of stressed, malignant transformed, and infected cells.
Natural killer T (NKT) cells are a heterogeneous group of T cells that share properties of both T cells and natural killer cells. Many of these cells recognize the non-polymorphic CD1d molecule, an antigen-presenting molecule that binds self and foreign lipids and glycolipids. They constitute only approximately 1% of all peripheral blood T cells. Natural killer T cells should neither be confused with natural killer cells nor killer T cells.
Barbara Anne Croy is a Canadian reproductive immunologist and professor emerita in Biomedical and Molecular Sciences at Queen's University. From 2004 until 2016, Croy was a Canada Research Chair in Reproduction, Development and Sexual Function. In 2017, she was elected a Fellow of the Royal Society of Canada. Her research focus is on mice pregnancy and natural killer cells.
Extravillous trophoblasts(EVTs), are one form of differentiated trophoblast cells of the placenta. They are invasive mesenchymal cells which function to establish critical tissue connection in the developing placental-uterine interface. EVTs derive from progenitor cytotrophoblasts (CYTs), as does the other main trophoblast subtype, syncytiotrophoblast (SYN). They are sometimes called intermediate trophoblast.