
1,4-Dichlorobenzene (1,4-DCB, p-DCB, or para-dichlorobenzene, sometimes abbreviated as PDCB or para) is an aryl chloride and isomer of dichlorobenzene with the formula C6H4Cl2. This colorless solid has a strong odor. The molecule consists of a benzene ring with two chlorine atoms (replacing hydrogen atoms) on opposing sites of the ring.

Trichloroethylene (TCE) is a halocarbon with the formula C2HCl3, commonly used as an industrial degreasing solvent. It is a clear, colourless, non-flammable, volatile liquid with a chloroform-like pleasant mild smell and sweet taste. Its IUPAC name is trichloroethene. Trichloroethylene has been sold under a variety of trade names. Industrial abbreviations include TCE, trichlor, Trike, Tricky and tri. Under the trade names Trimar and Trilene, it was used as a volatile anesthetic and as an inhaled obstetrical analgesic. It should not be confused with the similar 1,1,1-trichloroethane, which is commonly known as chlorothene.

Environmental remediation is the cleanup of hazardous substances dealing with the removal, treatment and containment of pollution or contaminants from environmental media such as soil, groundwater, sediment. Remediation may be required by regulations before development of land revitalization projects. Developers who agree to voluntary cleanup may be offered incentives under state or municipal programs like New York State's Brownfield Cleanup Program. If remediation is done by removal the waste materials are simply transported off-site for disposal at another location. The waste material can also be contained by physical barriers like slurry walls. The use of slurry walls is well-established in the construction industry. The application of (low) pressure grouting, used to mitigate soil liquefaction risks in San Francisco and other earthquake zones, has achieved mixed results in field tests to create barriers, and site-specific results depend upon many variable conditions that can greatly impact outcomes.

Bioremediation broadly refers to any process wherein a biological system, living or dead, is employed for removing environmental pollutants from air, water, soil, flue gasses, industrial effluents etc., in natural or artificial settings. The natural ability of organisms to adsorb, accumulate, and degrade common and emerging pollutants has attracted the use of biological resources in treatment of contaminated environment. In comparison to conventional physicochemical treatment methods bioremediation may offer advantages as it aims to be sustainable, eco-friendly, cheap, and scalable.

Pentachlorophenol (PCP) is an organochlorine compound used as a pesticide and a disinfectant. First produced in the 1930s, it is marketed under many trade names. It can be found as pure PCP, or as the sodium salt of PCP, the latter of which dissolves easily in water. It can be biodegraded by some bacteria, including Sphingobium chlorophenolicum.
Chloropicrin, also known as PS and nitrochloroform, is a chemical compound currently used as a broad-spectrum antimicrobial, fungicide, herbicide, insecticide, and nematicide. It was used as a poison gas in World War I and allegedly by Russia in the Russian invasion of Ukraine. Its chemical structural formula is Cl3C−NO2.

Toxaphene was an insecticide used primarily for cotton in the southern United States during the late 1960s and the 1970s. Toxaphene is a mixture of over 670 different chemicals and is produced by reacting chlorine gas with camphene. It can be most commonly found as a yellow to amber waxy solid.

Soil contamination, soil pollution, or land pollution as a part of land degradation is caused by the presence of xenobiotic (human-made) chemicals or other alteration in the natural soil environment. It is typically caused by industrial activity, agricultural chemicals or improper disposal of waste. The most common chemicals involved are petroleum hydrocarbons, polynuclear aromatic hydrocarbons, solvents, pesticides, lead, and other heavy metals. Contamination is correlated with the degree of industrialization and intensity of chemical substance. The concern over soil contamination stems primarily from health risks, from direct contact with the contaminated soil, vapour from the contaminants, or from secondary contamination of water supplies within and underlying the soil. Mapping of contaminated soil sites and the resulting clean ups are time-consuming and expensive tasks, and require expertise in geology, hydrology, chemistry, computer modelling, and GIS in Environmental Contamination, as well as an appreciation of the history of industrial chemistry.
Dehalococcoides is a genus of bacteria within class Dehalococcoidia that obtain energy via the oxidation of hydrogen and subsequent reductive dehalogenation of halogenated organic compounds in a mode of anaerobic respiration called organohalide respiration. They are well known for their great potential to remediate halogenated ethenes and aromatics. They are the only bacteria known to transform highly chlorinated dioxins, PCBs. In addition, they are the only known bacteria to transform tetrachloroethene to ethene.

1,3-Dichloropropene, sold under diverse trade names, is an organochlorine compound with the formula C3H4Cl2. It is a colorless liquid with a sweet smell. It is feebly soluble in water and evaporates easily. It is used mainly in farming as a pesticide, specifically as a preplant fumigant and nematicide. It is widely used in the US and other countries, but is banned in 34 countries.
A dense non-aqueous phase liquid or DNAPL is a denser-than-water NAPL, i.e. a liquid that is both denser than water and is immiscible in or does not dissolve in water.
Groundwater remediation is the process that is used to treat polluted groundwater by removing the pollutants or converting them into harmless products. Groundwater is water present below the ground surface that saturates the pore space in the subsurface. Globally, between 25 per cent and 40 per cent of the world's drinking water is drawn from boreholes and dug wells. Groundwater is also used by farmers to irrigate crops and by industries to produce everyday goods. Most groundwater is clean, but groundwater can become polluted, or contaminated as a result of human activities or as a result of natural conditions.

Electrical resistance heating (ERH) is an intensive in situ environmental remediation method that uses the flow of alternating current electricity to heat soil and groundwater and evaporate contaminants. Electric current is passed through a targeted soil volume between subsurface electrode elements. The resistance to electrical flow that exists in the soil causes the formation of heat; resulting in an increase in temperature until the boiling point of water at depth is reached. After reaching this temperature, further energy input causes a phase change, forming steam and removing volatile contaminants. ERH is typically more cost effective when used for treating contaminant source areas.
In situ chemical oxidation (ISCO), a form of advanced oxidation process, is an environmental remediation technique used for soil and/or groundwater remediation to lower the concentrations of targeted environmental contaminants to acceptable levels. ISCO is accomplished by introducing strong chemical oxidizers into the contaminated medium to destroy chemical contaminants in place. It can be used to remediate a variety of organic compounds, including some that are resistant to natural degradation. The in situ in ISCO is just Latin for "in place", signifying that ISCO is a chemical oxidation reaction that occurs at the site of the contamination.
A permeable reactive barrier (PRB), also referred to as a permeable reactive treatment zone (PRTZ), is a developing technology that has been recognized as being a cost-effective technology for in situ groundwater remediation. PRBs are barriers which allow some—but not all—materials to pass through. One definition for PRBs is an in situ treatment zone that passively captures a plume of contaminants and removes or breaks down the contaminants, releasing uncontaminated water. The primary removal methods include: (1) sorption and precipitation, (2) chemical reaction, and (3) reactions involving biological mechanisms.
In situ chemical reduction (ISCR) is a type of environmental remediation technique used for soil and/or groundwater remediation to reduce the concentrations of targeted environmental contaminants to acceptable levels. It is the mirror process of In Situ Chemical Oxidation (ISCO). ISCR is usually applied in the environment by injecting chemically reductive additives in liquid form into the contaminated area or placing a solid medium of chemical reductants in the path of a contaminant plume. It can be used to remediate a variety of organic compounds, including some that are resistant to natural degradation.

Zerovalent iron (ZVI) is jargon that describes forms of iron metal that are proposed for used in Groundwater remediation.
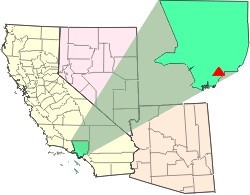
The Waste Disposal Inc. Superfund site is an oil-related contaminated site in the highly industrialized city of Santa Fe Springs in Los Angeles County, California. It is approximately 38 acres (15 ha), with St Paul's high school immediately adjacent to the northeast corner of the site. Approximately 15,000 residents of Santa Fe Springs obtain drinking water from wells within three miles (4.8 km) of the site.
Dehalogenimonas lykanthroporepellens is an anaerobic, Gram-negative bacteria in the phylum Chloroflexota isolated from a Superfund site in Baton Rouge, Louisiana. It is useful in bioremediation for its ability to reductively dehalogenate chlorinated alkanes.

Non-aqueous phase liquids, or NAPLs, are organic liquid contaminants characterized by their relative immiscibility with water. Common examples of NAPLs are petroleum products, coal tars, chlorinated solvents, and pesticides. Strategies employed for their removal from the subsurface environment have expanded since the late-20th century.














