
Bromine is a chemical element; it has symbol Br and atomic number 35. It is a volatile red-brown liquid at room temperature that evaporates readily to form a similarly coloured vapour. Its properties are intermediate between those of chlorine and iodine. Isolated independently by two chemists, Carl Jacob Löwig and Antoine Jérôme Balard, its name was derived from the Ancient Greek βρῶμος (bromos) meaning "stench", referring to its sharp and pungent smell.

The Montreal Protocol is an international treaty designed to protect the ozone layer by phasing out the production of numerous substances that are responsible for ozone depletion. It was agreed on 16 September 1987, and entered into force on 1 January 1989. Since then, it has undergone nine revisions, in 1990 (London), 1991 (Nairobi), 1992 (Copenhagen), 1993 (Bangkok), 1995 (Vienna), 1997 (Montreal), 1998 (Australia), 1999 (Beijing) and 2016 (Kigali) As a result of the international agreement, the ozone hole in Antarctica is slowly recovering. Climate projections indicate that the ozone layer will return to 1980 levels between 2040 and 2066. Due to its widespread adoption and implementation, it has been hailed as an example of successful international co-operation. Former UN Secretary-General Kofi Annan stated that "perhaps the single most successful international agreement to date has been the Montreal Protocol". In comparison, effective burden-sharing and solution proposals mitigating regional conflicts of interest have been among the success factors for the ozone depletion challenge, where global regulation based on the Kyoto Protocol has failed to do so. In this case of the ozone depletion challenge, there was global regulation already being installed before a scientific consensus was established. Also, overall public opinion was convinced of possible imminent risks.

Ozone depletion consists of two related events observed since the late 1970s: a steady lowering of about four percent in the total amount of ozone in Earth's atmosphere, and a much larger springtime decrease in stratospheric ozone around Earth's polar regions. The latter phenomenon is referred to as the ozone hole. There are also springtime polar tropospheric ozone depletion events in addition to these stratospheric events.

Chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) are fully or partly halogenated hydrocarbons that contain carbon (C), hydrogen (H), chlorine (Cl), and fluorine (F), produced as volatile derivatives of methane, ethane, and propane.

The haloalkanes are alkanes containing one or more halogen substituents. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely used commercially. They are used as flame retardants, fire extinguishants, refrigerants, propellants, solvents, and pharmaceuticals. Subsequent to the widespread use in commerce, many halocarbons have also been shown to be serious pollutants and toxins. For example, the chlorofluorocarbons have been shown to lead to ozone depletion. Methyl bromide is a controversial fumigant. Only haloalkanes that contain chlorine, bromine, and iodine are a threat to the ozone layer, but fluorinated volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting properties and the United States Environmental Protection Agency has designated the compound a non-ozone layer depleter. For more information, see Halomethane. Haloalkane or alkyl halides are the compounds which have the general formula "RX" where R is an alkyl or substituted alkyl group and X is a halogen.

Bromochlorodifluoromethane (BCF), also referred to by the code numbers Halon 1211 and Freon 12B1, is a haloalkane with the chemical formula CF2ClBr. It is used for fire suppression, especially for expensive equipment or items that could be damaged by the residue from other types of extinguishers. It is stored as a liquid under pressure and vaporizes when discharged to suppress fires. The use of halons, including Halon 1211, has decreased over time due to their adverse impact on the ozone layer. Alternatives have been developed to mitigate environmental concerns while still providing effective fire suppression capabilities.
Bromotrifluoromethane, commonly known as Halon 1301, R13B1, Halon 13B1 or BTM, is an organic halide with the chemical formula CBrF3. It is used for gaseous fire suppression as a far less toxic alternative to bromochloromethane.

A refrigerant is a working fluid used in the refrigeration cycle of air conditioning systems and heat pumps where in most cases they undergo a repeated phase transition from a liquid to a gas and back again. Refrigerants are heavily regulated due to their toxicity, flammability and the contribution of CFC and HCFC refrigerants to ozone depletion and that of HFC refrigerants to climate change.

Halomethane compounds are derivatives of methane with one or more of the hydrogen atoms replaced with halogen atoms. Halomethanes are both naturally occurring, especially in marine environments, and human-made, most notably as refrigerants, solvents, propellants, and fumigants. Many, including the chlorofluorocarbons, have attracted wide attention because they become active when exposed to ultraviolet light found at high altitudes and destroy the Earth's protective ozone layer.

The infrared atmospheric window refers to a region of the Infrared spectrum where there is relatively little absorption of terrestrial thermal radiation by atmospheric gases. The window plays an important role in the atmospheric greenhouse effect by maintaining the balance between incoming solar radiation and outgoing IR to space. In the Earth's atmosphere this window is roughly the region between 8 and 14 μm although it can be narrowed or closed at times and places of high humidity because of the strong absorption in the water vapor continuum or because of blocking by clouds. It covers a substantial part of the spectrum from surface thermal emission which starts at roughly 5 μm. Principally it is a large gap in the absorption spectrum of water vapor. Carbon dioxide plays an important role in setting the boundary at the long wavelength end. Ozone partly blocks transmission in the middle of the window.
Fluoroform, or trifluoromethane, is the chemical compound with the formula CHF3. It is a Hydrofluorocarbon as well as being apart of the haloforms, a class of compounds with the formula CHX3 with C3v symmetry. Fluoroform is used in diverse applications in organic synthesis. It is not an ozone depleter but is a greenhouse gas.
Trifluoroiodomethane, also referred to as trifluoromethyl iodide is a halomethane with the formula CF3I. It is an experimental alternative to Halon 1301 (CBrF3) in unoccupied areas. It would be used as a gaseous fire suppression flooding agent for in-flight aircraft and electronic equipment fires.
Organofluorine chemistry describes the chemistry of organofluorine compounds, organic compounds that contain a carbon–fluorine bond. Organofluorine compounds find diverse applications ranging from oil and water repellents to pharmaceuticals, refrigerants, and reagents in catalysis. In addition to these applications, some organofluorine compounds are pollutants because of their contributions to ozone depletion, global warming, bioaccumulation, and toxicity. The area of organofluorine chemistry often requires special techniques associated with the handling of fluorinating agents.

Dibromodifluoromethane is a mixed halomethane. It is a colorless non-flammable liquid. Along with Halons 1211, 2402, and 1301, it is one of the most effective fire extinguishers, however, it is also very toxic. It is a class I ozone depleting substance (ODS).
PhostrEx is a fire suppression agent developed for use in aviation applications to replace halon, a greenhouse gas (GHG). It was developed by Eclipse Aviation for use aboard their Eclipse 500 very light jets as an engine fire suppression system, and is now being marketed to other aviation manufacturers.

Automatic fire suppression systems control and extinguish fires without human intervention. Examples of automatic systems include fire sprinkler system, gaseous fire suppression, and condensed aerosol fire suppression. When fires are extinguished in the early stages loss of life is minimal since 93% of all fire-related deaths occur once the fire has progressed beyond the early stages.

Fluorine is a chemical element; it has symbol F and atomic number 9. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. As the most electronegative reactive element, it is extremely reactive, as it reacts with all other elements except for the light inert gases.

Pentafluoroethane is a fluorocarbon with the formula CF3CHF2. Pentafluoroethane is currently used as a refrigerant (known as R-125) and also used as a fire suppression agent in fire suppression systems.

Condensed aerosol fire suppression is a particle-based method of fire extinction. It is similar to but not identical to dry chemical fire extinction methods, using an innovative pyrogenic, condensed aerosol fire suppressant. It is a highly effective fire suppression method for class A, B, C, E and F. Some aerosol-generating compounds produce a corrosive by-product that may damage electronic equipment, although later generations lower the effect.
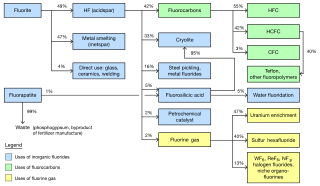
The global market for chemicals from fluorine was about US$16 billion per year as of 2006. The industry was predicted to reach 2.6 million metric tons per year by 2015. The largest market is the United States. Western Europe is the second largest. Asia Pacific is the fastest growing region of production. China in particular has experienced significant growth as a fluorochemical market and is becoming a producer of them as well. Fluorite mining was estimated in 2003 to be a $550 million industry, extracting 4.5 million tons per year.
















