
Chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) are fully or partly halogenated hydrocarbons that contain carbon (C), hydrogen (H), chlorine (Cl), and fluorine (F), produced as volatile derivatives of methane, ethane, and propane.
Isobutane, also known as i-butane, 2-methylpropane or methylpropane, is a chemical compound with molecular formula HC(CH3)3. It is an isomer of butane. Isobutane is a colorless, odorless gas. It is the simplest alkane with a tertiary carbon atom. Isobutane is used as a precursor molecule in the petrochemical industry, for example in the synthesis of isooctane.

Bromochlorodifluoromethane (BCF), also referred to by the code numbers Halon 1211 and Freon 12B1, is a haloalkane with the chemical formula CF2ClBr. It is used for fire suppression, especially for expensive equipment or items that could be damaged by the residue from other types of extinguishers. It is stored as a liquid under pressure and vaporizes when discharged to suppress fires. The use of halons, including Halon 1211, has decreased over time due to their adverse impact on the ozone layer. Alternatives have been developed to mitigate environmental concerns while still providing effective fire suppression capabilities.

A refrigerant is a working fluid used in the refrigeration cycle of air conditioning systems and heat pumps where in most cases they undergo a repeated phase transition from a liquid to a gas and back again. Refrigerants are heavily regulated due to their toxicity, flammability and the contribution of CFC and HCFC refrigerants to ozone depletion and that of HFC refrigerants to climate change.

Halomethane compounds are derivatives of methane with one or more of the hydrogen atoms replaced with halogen atoms. Halomethanes are both naturally occurring, especially in marine environments, and human-made, most notably as refrigerants, solvents, propellants, and fumigants. Many, including the chlorofluorocarbons, have attracted wide attention because they become active when exposed to ultraviolet light found at high altitudes and destroy the Earth's protective ozone layer.

Methyl formate, also called methyl methanoate, is the methyl ester of formic acid. The simplest example of a carboxylate ester, it is a colorless liquid with an ethereal odour, high vapor pressure, and low surface tension. It is a precursor to many other compounds of commercial interest.
Dichlorodifluoromethane (R-12) is a colorless gas usually sold under the brand name Freon-12, and a chlorofluorocarbon halomethane (CFC) used as a refrigerant and aerosol spray propellant. In compliance with the Montreal Protocol, its manufacture was banned in developed countries in 1996, and in developing countries in 2010 out of concerns about its damaging effect on the ozone layer. Its only allowed usage is as a fire retardant in submarines and aircraft. It is soluble in many organic solvents. R-12 cylinders are colored white.
1,1,1,2-Tetrafluoroethane (also known as norflurane (INN), R-134a, Klea 134a,Freon 134a, Forane 134a, Genetron 134a, Green Gas, Florasol 134a, Suva 134a, HFA-134a, or HFC-134a) is a hydrofluorocarbon (HFC) and haloalkane refrigerant with thermodynamic properties similar to R-12 (dichlorodifluoromethane) but with insignificant ozone depletion potential and a lower 100-year global warming potential (1,430, compared to R-12's GWP of 10,900). It has the formula CF3CH2F and a boiling point of −26.3 °C (−15.34 °F) at atmospheric pressure. R-134a cylinders are colored light blue. A phaseout and transition to HFO-1234yf and other refrigerants, with GWPs similar to CO2, began in 2012 within the automotive market.
Chlorodifluoromethane or difluoromonochloromethane is a hydrochlorofluorocarbon (HCFC). This colorless gas is better known as HCFC-22, or R-22, or CHClF
2. It was commonly used as a propellant and refrigerant. These applications were phased out under the Montreal Protocol in developed countries in 2020 due to the compound's ozone depletion potential (ODP) and high global warming potential (GWP), and in developing countries this process will be completed by 2030. R-22 is a versatile intermediate in industrial organofluorine chemistry, e.g. as a precursor to tetrafluoroethylene.
Fluoroform, or trifluoromethane, is the chemical compound with the formula CHF3. It is a Hydrofluorocarbon as well as being apart of the haloforms, a class of compounds with the formula CHX3 with C3v symmetry. Fluoroform is used in diverse applications in organic synthesis. It is not an ozone depleter but is a greenhouse gas.

1,1,1,2,3,3,3-Heptafluoropropane, also called heptafluoropropane, HFC-227ea, HFC-227 or FM-200, as well as apaflurane (INN), is a colourless, odourless gaseous halocarbon commonly used as a gaseous fire suppression agent.
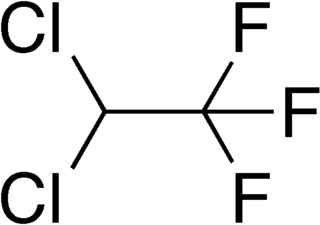
2,2-Dichloro-1,1,1-trifluoroethane or HCFC-123 is considered as an alternative to CFC-11 in low pressure refrigeration and HVAC systems, and should not be used in foam blowing processes or solvent applications. It is also the primary component of the Halotron I fire-extinguishing mixture.

Bromochloromethane or methylene bromochloride and Halon 1011 is a mixed halomethane. It is a heavy low-viscosity liquid with refractive index 1.4808.

A fire extinguisher is a handheld active fire protection device usually filled with a dry or wet chemical used to extinguish or control small fires, often in emergencies. It is not intended for use on an out-of-control fire, such as one which has reached the ceiling, endangers the user, or otherwise requires the equipment, personnel, resources or expertise of a fire brigade. Typically, a fire extinguisher consists of a hand-held cylindrical pressure vessel containing an agent that can be discharged to extinguish a fire. Fire extinguishers manufactured with non-cylindrical pressure vessels also exist but are less common.

Automatic fire suppression systems control and extinguish fires without human intervention. Examples of automatic systems include fire sprinkler system, gaseous fire suppression, and condensed aerosol fire suppression. When fires are extinguished in the early stages loss of life is minimal since 93% of all fire-related deaths occur once the fire has progressed beyond the early stages.
HC-12a, also called ES-12a, OZ-12a, DURACOOL 12a and Hydrocarbon Blend B, is a "drop-in" replacement refrigerant for Freon-12 and to a lesser extent, R-134a. HC-12a is a mixture of hydrocarbons, specifically propane (R-290) and isobutane (R-600a), and is therefore considered nearly non-ozone-depleting when compared to dichlorodifluoromethane or 1,1,1,2-tetrafluoroethane (R-134a). The mixture can be used in refrigeration systems designed for R-12. HC-12a provides better cooling than an R-12 system retrofitted to R-134a, with much greater energy efficiency as well. Unlike R-134a, HC-12a is completely compatible with the hoses and oils used in R-12 systems, making the conversion much easier to accomplish. HC-12a is also patent-free due to its non-synthetic nature.

Pentafluoroethane is a fluorocarbon with the formula CF3CHF2. Pentafluoroethane is currently used as a refrigerant (known as R-125) and also used as a fire suppression agent in fire suppression systems.

1-Chloro-1,1-difluoroethane (HCFC-142b) is a haloalkane with the chemical formula CH3CClF2. It belongs to the hydrochlorofluorocarbon (HCFC) family of man-made compounds that contribute significantly to both ozone depletion and global warming when released into the environment. It is primarily used as a refrigerant where it is also known as R-142b and by trade names including Freon-142b.

Clean agent FS 49 C2 is an environmentally engineered, human safe, fast acting Clean Agent fire extinguishing gas for gaseous fire suppression installed in a suited fire suppression system. The gas consists of tetrafluoroethane, pentafluoroethane and carbon dioxide.
Fluorinated gases (F-gases) are chemical compounds containing fluorine that are gases near room temperature.


















