
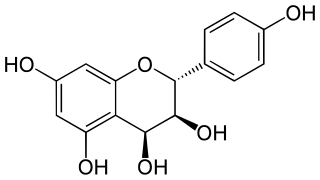
Catechin is a flavan-3-ol, a type of secondary metabolite providing antioxidant roles in plants. It belongs to the subgroup of polyphenols called flavonoids.
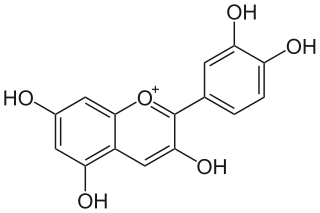
Cyanidin is a natural organic compound. It is a particular type of anthocyanidin. It is a pigment found in many red berries including grapes, bilberry, blackberry, blueberry, cherry, chokeberry, cranberry, elderberry, hawthorn, loganberry, açai berry and raspberry. It can also be found in other fruits such as apples and plums, and in red cabbage and red onion. It has a characteristic reddish-purple color, though this can change with pH; solutions of the compound are red at pH < 3, violet at pH 7-8, and blue at pH > 11. In certain fruits, the highest concentrations of cyanidin are found in the seeds and skin. Cyanidin has been found to be a potent sirtuin 6 (SIRT6) activator.
In enzymology, a flavanone 4-reductase (EC 1.1.1.234) is an enzyme that catalyzes the chemical reaction
In enzymology, an anthocyanidin reductase (EC 1.3.1.77) is an enzyme that catalyzes the chemical reaction

In enzymology, divinyl chlorophyllide a 8-vinyl-reductase (EC 1.3.1.75) is an enzyme that catalyzes the chemical reaction
In enzymology, a leucocyanidin oxygenase (EC 1.14.11.19) is an enzyme that catalyzes the chemical reaction

In enzymology, an aspartate-semialdehyde dehydrogenase is an enzyme that is very important in the biosynthesis of amino acids in prokaryotes, fungi, and some higher plants. It forms an early branch point in the metabolic pathway forming lysine, methionine, leucine and isoleucine from aspartate. This pathway also produces diaminopimelate which plays an essential role in bacterial cell wall formation. There is particular interest in ASADH as disabling this enzyme proves fatal to the organism giving rise to the possibility of a new class of antibiotics, fungicides, and herbicides aimed at inhibiting it.
In enzymology, a leucoanthocyanidin reductase (EC 1.17.1.3) (LAR, aka leucocyanidin reductase or LCR) is an enzyme that catalyzes the chemical reaction
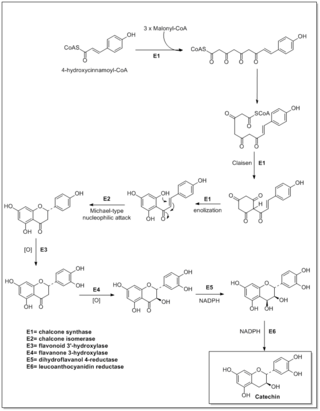
Flavonoids are synthesized by the phenylpropanoid metabolic pathway in which the amino acid phenylalanine is used to produce 4-coumaroyl-CoA. This can be combined with malonyl-CoA to yield the true backbone of flavonoids, a group of compounds called chalcones, which contain two phenyl rings. Conjugate ring-closure of chalcones results in the familiar form of flavonoids, the three-ringed structure of a flavone. The metabolic pathway continues through a series of enzymatic modifications to yield flavanones → dihydroflavonols → anthocyanins. Along this pathway, many products can be formed, including the flavonols, flavan-3-ols, proanthocyanidins (tannins) and a host of other various polyphenolics.

Anthocyanins, also called anthocyans, are water-soluble vacuolar pigments that, depending on their pH, may appear red, purple, blue, or black. In 1835, the German pharmacist Ludwig Clamor Marquart gave the name Anthokyan to a chemical compound that gives flowers a blue color for the first time in his treatise "Die Farben der Blüthen". Food plants rich in anthocyanins include the blueberry, raspberry, black rice, and black soybean, among many others that are red, blue, purple, or black. Some of the colors of autumn leaves are derived from anthocyanins.

Aromadendrin is a flavanonol, a type of flavonoid. It can be found in the wood of Pinus sibirica.

The 3-Deoxyanthocyanidins and their glycosides are molecules with an anthocyanidins backbone lacking an hydroxyl group at position 3 on the C-ring. This nomenclature is the inverse of that which is commonly used in flavonoids, where the hydroxy-group is assumed absent if it is not specified, e. g. flavan-3-ol, flavan-4-ol, flavan-3,4-ol and flavonol.
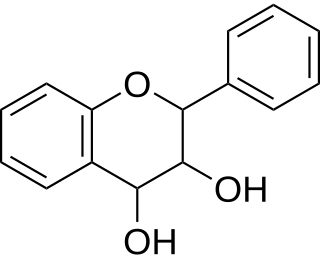
Leucoanthocyanidin (flavan-3,4-diols) are colorless chemical compounds related to anthocyanidins and anthocyanins. Leucoanthocyanins can be found in Anadenanthera peregrina and in several species of Nepenthes including N. burbidgeae, N. muluensis, N. rajah, N. tentaculata, and N. × alisaputrana.

Leucocyanidin is a colorless chemical compound that is a member of the class of natural products known as leucoanthocyanidins.
Leucopelargonidin is a colorless chemical compound related to leucoanthocyanins. It can be found in Albizia lebbeck, in the fruit of Anacardium occidentale (Cashew), in the fruit of Areca catechu, in the fruit of Hydnocarpus wightiana, in the rhizome of Rumex hymenosepalus, in Zea mays (Corn) and in Ziziphus jujuba.
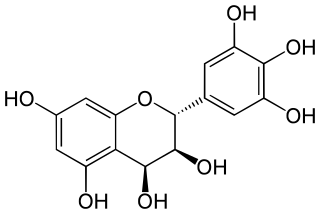
Leucodelphinidin is a colorless chemical compound related to leucoanthocyanidins. It can be found in Acacia auriculiformis, in the bark of Karada and in the kino (gum) from Eucalyptus pilularis.

The flavan-4-ols (3-deoxyflavonoids) are flavone-derived alcohols and a family of flavonoids. Flavan-4-ols are colorless precursor compounds that polymerize to form red phlobaphene pigments. They can be found in the sorghum. Glycosides can be isolated from a methanol extract of the rhizomes of Abacopteris penangiana.

Sinapaldehyde is an organic compound with the formula HO(CH3O)2C6H2CH=CHCHO. It is a derivative of cinnamaldehyde, featuring one hydroxy group and two methoxy groups as substituents. It is an intermediate in the formation of sinapyl alcohol, a lignol that is a major precursor to lignin.
The biosynthesis of phenylpropanoids involves a number of enzymes.

Blue flower colour was always associated with something unusual and desired. Blue roses especially were assumed to be a dream that cannot be realised. Blue colour in flower petals is caused by anthocyanins, which are members of flavonoid class metabolites. We can diversify three main classes of anthocyanin pigments: cyaniding type responsible for red coloration, pelargonidin type responsible for orange colour and delphinidin type responsible for violet/blue flower and fruits coloration. The main difference in the structure of listed anthocyanins type is the number of hydroxyl groups in the B-ring of the anthocyanin. Nevertheless, in the monomeric state anthocyanins never show blue colour in the weak acidic and neutral pH. The mechanism of blue colour formation are very complicated in most cases, presence of delphinidin type pigments is not sufficient, great role play also the pH and the formation of complexes of anthocyanins with flavones and metal ions.















