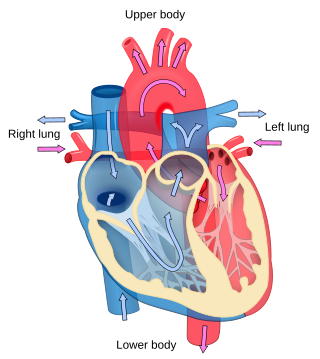
Cardiology is the study of the heart. Cardiology is a branch of medicine that deals with disorders of the heart and the cardiovascular system. The field includes medical diagnosis and treatment of congenital heart defects, coronary artery disease, heart failure, valvular heart disease and electrophysiology. Physicians who specialize in this field of medicine are called cardiologists, a specialty of internal medicine. Pediatric cardiologists are pediatricians who specialize in cardiology. Physicians who specialize in cardiac surgery are called cardiothoracic surgeons or cardiac surgeons, a specialty of general surgery.

Heart murmurs are unique heart sounds produced when blood flows across a heart valve or blood vessel. This occurs when turbulent blood flow creates a sound loud enough to hear with a stethoscope. Turbulent blood flow is not smooth. The sound differs from normal heart sounds by their characteristics. For example, heart murmurs may have a distinct pitch, duration and timing. The major way health care providers examine the heart on physical exam is heart auscultation; another clinical technique is palpation, which can detect by touch when such turbulence causes the vibrations called cardiac thrill. A murmur is a sign found during the cardiac exam. Murmurs are of various types and are important in the detection of cardiac and valvular pathologies.

Tetralogy of Fallot (TOF), formerly known as Steno-Fallot tetralogy, is a congenital heart defect characterized by four specific cardiac defects. Classically, the four defects are:
Pulmonary heart disease, also known as cor pulmonale, is the enlargement and failure of the right ventricle of the heart as a response to increased vascular resistance or high blood pressure in the lungs.

Pulmonary edema, also known as pulmonary congestion, is excessive liquid accumulation in the tissue and air spaces of the lungs. It leads to impaired gas exchange and may cause hypoxemia and respiratory failure. It is due to either failure of the left ventricle of the heart to remove oxygenated blood adequately from the pulmonary circulation, or an injury to the lung tissue directly or blood vessels of the lung.

Atrial septal defect (ASD) is a congenital heart defect in which blood flows between the atria of the heart. Some flow is a normal condition both pre-birth and immediately post-birth via the foramen ovale; however, when this does not naturally close after birth it is referred to as a patent (open) foramen ovale (PFO). It is common in patients with a congenital atrial septal aneurysm (ASA).

Pulmonary hypertension is a condition of increased blood pressure in the arteries of the lungs. Symptoms include shortness of breath, fainting, tiredness, chest pain, swelling of the legs, and a fast heartbeat. The condition may make it difficult to exercise. Onset is typically gradual.
A cyanotic heart defect is any congenital heart defect (CHD) that occurs due to deoxygenated blood bypassing the lungs and entering the systemic circulation, or a mixture of oxygenated and unoxygenated blood entering the systemic circulation. It is caused by structural defects of the heart such as right-to-left or bidirectional shunting, malposition of the great arteries, or any condition which increases pulmonary vascular resistance. The result may be the development of collateral circulation.

A ventricular septal defect (VSD) is a defect in the ventricular septum, the wall dividing the left and right ventricles of the heart. The extent of the opening may vary from pin size to complete absence of the ventricular septum, creating one common ventricle. The ventricular septum consists of an inferior muscular and superior membranous portion and is extensively innervated with conducting cardiomyocytes.
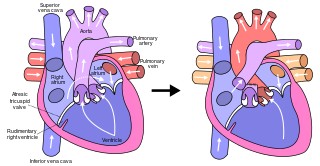
The Fontan procedure or Fontan–Kreutzer procedure is a palliative surgical procedure used in children with univentricular hearts. It involves diverting the venous blood from the inferior vena cava (IVC) and superior vena cava (SVC) to the pulmonary arteries without passing through the morphologic right ventricle; i.e., the systemic and pulmonary circulations are placed in series with the functional single ventricle. The procedure was initially performed in 1968 by Francis Fontan and Eugene Baudet from Bordeaux, France, published in 1971, simultaneously described in 1971 by Guillermo Kreutzer from Buenos Aires, Argentina, and finally published in 1973.

Cardiac catheterization is the insertion of a catheter into a chamber or vessel of the heart. This is done both for diagnostic and interventional purposes.
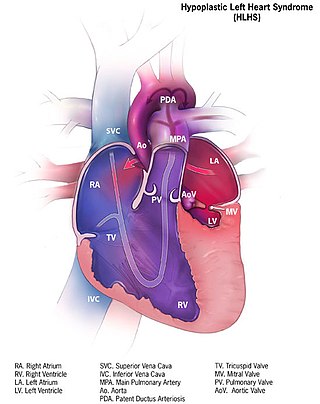
Hypoplastic left heart syndrome (HLHS) is a rare congenital heart defect in which the left side of the heart is severely underdeveloped and incapable of supporting the systemic circulation. It is estimated to account for 2-3% of all congenital heart disease. Early signs and symptoms include poor feeding, cyanosis, and diminished pulse in the extremities. The etiology is believed to be multifactorial resulting from a combination of genetic mutations and defects resulting in altered blood flow in the heart.

Atrioventricular septal defect (AVSD) or atrioventricular canal defect (AVCD), also known as "common atrioventricular canal" (CAVC) or "endocardial cushion defect" (ECD), is characterized by a deficiency of the atrioventricular septum of the heart that creates connections between all four of its chambers. It is caused by an abnormal or inadequate fusion of the superior and inferior endocardial cushions with the mid portion of the atrial septum and the muscular portion of the ventricular septum.

Hypoxemia is an abnormally low level of oxygen in the blood. More specifically, it is oxygen deficiency in arterial blood. Hypoxemia has many causes, and often causes hypoxia as the blood is not supplying enough oxygen to the tissues of the body.
A right-to-left shunt is a cardiac shunt which allows blood to flow from the right heart to the left heart. This terminology is used both for the abnormal state in humans and for normal physiological shunts in reptiles.

Right ventricular hypertrophy (RVH) is a condition defined by an abnormal enlargement of the cardiac muscle surrounding the right ventricle. The right ventricle is one of the four chambers of the heart. It is located towards the lower-end of the heart and it receives blood from the right atrium and pumps blood into the lungs.
A cardiac shunt is a pattern of blood flow in the heart that deviates from the normal circuit of the circulatory system. It may be described as right-left, left-right or bidirectional, or as systemic-to-pulmonary or pulmonary-to-systemic. The direction may be controlled by left and/or right heart pressure, a biological or artificial heart valve or both. The presence of a shunt may also affect left and/or right heart pressure either beneficially or detrimentally.
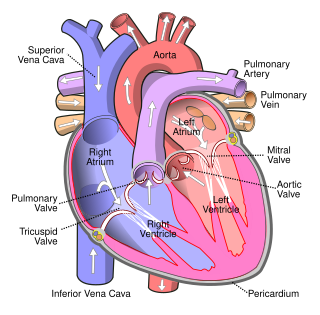
Lutembacher's syndrome is a very rare form of congenital heart disease that affects one of the chambers of the heart as well as a valve. It is commonly known as both congenital atrial septal defect (ASD) and acquired mitral stenosis (MS). Congenital atrial septal defect refers to a hole being in the septum or wall that separates the two atria; this condition is usually seen in fetuses and infants. Mitral stenosis refers to mitral valve leaflets sticking to each other making the opening for blood to pass from the atrium to the ventricles very small. With the valve being so small, blood has difficulty passing from the left atrium into the left ventricle. Septal defects that may occur with Lutembacher's syndrome include: Ostium primum atrial septal defect or ostium secundum which is more prevalent.
The following outline is provided as an overview of and topical guide to cardiology, the branch of medicine dealing with disorders of the human heart. The field includes medical diagnosis and treatment of congenital heart defects, coronary artery disease, heart failure, valvular heart disease and electrophysiology. Physicians who specialize in cardiology are called cardiologists.
Raghib syndrome is rare a congenital heart defect where the left superior vena cava (LSVC) is draining into the left atrium in addition to an absent coronary sinus and an atrial septal defect. This can be considered a dangerous heart condition because it puts the individual at a high risk of stroke. Other defects that are often associated with Raghib syndrome can include ventricular septal defects, enlargement of the tricuspid annulus, and pulmonary stenosis. While this is considered an extremely rare developmental complex, cases regarding a persistent left superior vena cava (PLSVC) are relatively common among congenital heart defects. It is also important to note that the PLSVC often drains into the right atrium, and only drains into the left atrium in approximately 10 to 20% of individuals with the defect.
















