Related Research Articles
In physics, statistical mechanics is a mathematical framework that applies statistical methods and probability theory to large assemblies of microscopic entities. It does not assume or postulate any natural laws, but explains the macroscopic behavior of nature from the behavior of such ensembles.
Geochemistry is the science that uses the tools and principles of chemistry to explain the mechanisms behind major geological systems such as the Earth's crust and its oceans. The realm of geochemistry extends beyond the Earth, encompassing the entire Solar System, and has made important contributions to the understanding of a number of processes including mantle convection, the formation of planets and the origins of granite and basalt. It is an integrated field of chemistry and geology.

Solvation describes the interaction of a solvent with dissolved molecules. Both ionized and uncharged molecules interact strongly with a solvent, and the strength and nature of this interaction influence many properties of the solute, including solubility, reactivity, and color, as well as influencing the properties of the solvent such as its viscosity and density. In the process of solvation, ions are surrounded by a concentric shell of solvent. Solvation is the process of reorganizing solvent and solute molecules into solvation complexes. Solvation involves bond formation, hydrogen bonding, and van der Waals forces. Solvation of a solute by water is called hydration.
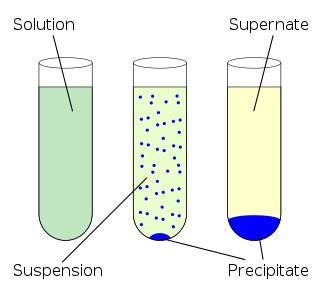
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution.
Solubility equilibrium is a type of dynamic equilibrium that exists when a chemical compound in the solid state is in chemical equilibrium with a solution of that compound. The solid may dissolve unchanged, with dissociation, or with chemical reaction with another constituent of the solution, such as acid or alkali. Each solubility equilibrium is characterized by a temperature-dependent solubility product which functions like an equilibrium constant. Solubility equilibria are important in pharmaceutical, environmental and many other scenarios.
Extractive metallurgy is a branch of metallurgical engineering wherein process and methods of extraction of metals from their natural mineral deposits are studied. The field is a materials science, covering all aspects of the types of ore, washing, concentration, separation, chemical processes and extraction of pure metal and their alloying to suit various applications, sometimes for direct use as a finished product, but more often in a form that requires further working to achieve the given properties to suit the applications.
In chemical thermodynamics, the fugacity of a real gas is an effective partial pressure which replaces the mechanical partial pressure in an accurate computation of the chemical equilibrium constant. It is equal to the pressure of an ideal gas which has the same temperature and molar Gibbs free energy as the real gas.
Robert Minard Garrels was an American geochemist. Garrels applied experimental physical chemistry data and techniques to geology and geochemistry problems. The book Solutions, Minerals, and Equilibria co-authored in 1965 by Garrels and Charles L. Christ revolutionized aqueous geochemistry.
Chemical WorkBench is a proprietary simulation software tool aimed at the reactor scale kinetic modeling of homogeneous gas-phase and heterogeneous processes and kinetic mechanism development. It can be effectively used for the modeling, optimization, and design of a wide range of industrially and environmentally important chemistry-loaded processes. Chemical WorkBench is a modeling environment based on advanced scientific approaches, complementary databases, and accurate solution methods. Chemical WorkBench is developed and distributed by Kintech Lab.

DWSIM is an open-source CAPE-OPEN compliant chemical process simulator for Windows, Linux and macOS. DWSIM is built on top of the Microsoft .NET and Mono Platforms and features a Graphical User Interface (GUI), advanced thermodynamics calculations, reactions support and petroleum characterization / hypothetical component generation tools.
The Geochemist's Workbench (GWB) is an integrated set of interactive software tools for solving a range of problems in aqueous chemistry. The graphical user interface simplifies the use of the geochemical code.
Reactive transport modeling in porous media refers to the creation of computer models integrating chemical reaction with transport of fluids through the Earth's crust. Such models predict the distribution in space and time of the chemical reactions that occur along a flowpath. Reactive transport modeling in general can refer to many other processes, including reactive flow of chemicals through tanks, reactors, or membranes; particles and species in the atmosphere; gases exiting a smokestack; and migrating magma.
Aqion is a hydrochemistry software tool. It bridges the gap between scientific software and the calculation/handling of "simple" water-related tasks in daily routine practice. The software aqion is free for private users, education and companies.

Frederick T. Mackenzie is an American sedimentary and global biogeochemist. Mackenzie applies experimental and field data coupled to a sound theoretical framework to the solution of geological, geochemical, and oceanographic problems at various time and space scales.
Reverse weathering generally refers to the formation of a clay neoformation that utilizes cations and alkalinity in a process unrelated to the weathering of silicates. More specifically reverse weathering refers to the formation of authigenic clay minerals from the reaction of 1) biogenic silica with aqueous cations or cation bearing oxides or 2) cation poor precursor clays with dissolved cations or cation bearing oxides.

Dimitri Alexander Sverjensky is a professor in Earth and Planetary Sciences at Johns Hopkins University where his research is focused on geochemistry.
Joint Expert Speciation System (JESS) is a package of computer software and data developed collaboratively at Murdoch University and elsewhere by researchers interested in the chemical thermodynamics of water solutions with important applications in industry, biochemistry, medicine and the environment. Using information from the chemical literature, stored in databases for numerous chemical properties, JESS achieves coherence between frequently conflicting sources by automatic methods.

Patricia Martin Dove is an American geochemist. She is a University Distinguished Professor and the C.P. Miles Professor of Science at Virginia Tech with appointments in the Department of Geosciences and Department of Chemistry. Her research focuses on the kinetics and thermodynamics of mineral reactions with aqueous solutions in biogeochemical systems. Much of her work is on crystal nucleation and growth during biomineralization and biomaterial synthesis. She was elected a member of the National Academy of Sciences (NAS) in 2012 and currently serves as chair of Class I, Physical and Mathematical Sciences.
A Lippmann diagram is a graphical plot showing the solidus/solutus equilibrium states for a given binary solid solution in equilibrium with an aqueous solution containing the two substituting ions: Ba2+
and Sr2+
. It was proposed in the 1970s by F. Lippmann to determine excess Gibbs functions. This diagram summarizes the thermodynamic basis of solid-solution aqueous-solution systems (SS-AS) equilibria and helps to predict the nucleation kinetics for solid solutions crystallizing from an aqueous solution.
Groundwater contamination by pharmaceuticals, which belong to the category of contaminants of emerging concern (CEC) or emerging organic pollutants (EOP), has been receiving increasing attention in the fields of environmental engineering, hydrology and hydrogeochemistry since the last decades of the twentieth century.
References
- ↑ Zhu, C. and G. Anderson, 2002, Environmental Applications of Geochemical Modeling. Cambridge University Press, 300 pp.
- 1 2 3 4 Bethke, C.M., 2008, Geochemical and Biogeochemical Reaction Modeling. Cambridge University Press, 547 pp.
- ↑ Garrels, R.M. and F.T. Mackenzie, 1967, Origin of the chemical compositions of some springs and lakes. Equilibrium Concepts in Natural Waters, Advances in Chemistry Series67, American Chemical Society, Washington, DC, pp. 222-242
- ↑ Helgeson, H.C., 1968, Evaluation of irreversible reactions in geochemical processes involving minerals and aqueous solutions, I. Thermodynamic relations. Geochemica et Cosmochimica Acta32, 853-877
- ↑ Helgeson, H.C., R.M. Garrels and F.T. Mackenzie, 1969, Evaluation of irreversible reactions in geochemical processes involving minerals and aqueous solutions, II. Applications. Geochemica et Cosmochimica Acta33, 455-481
- ↑ Zhu, C., 2009, Geochemical Modeling of Reaction Paths and Geochemical Reaction Networks. In E.H. Oelkers and J. Schott(eds.), 2009, Thermodynamics and Kinetics of Water-Rock Interaction. Reviews in Mineralogy and Geochemistry70, 533-569
- ↑ Brady, P.V. and C.M. Bethke, 2000, Beyond the Kd approach. Ground Water38, 321-322
- 1 2 Anderson, G.M. 2009, Thermodynamics of Natural Systems. Cambridge University Press, 664 pp.
- ↑ Bethke, C.M., ChemPlugin User's Guide Release 15. Aqueous Solutions LLC, Champaign, IL USA https://www.chemplugin.gwb.com/documentation.php
- ↑ Dethlefsen, Frank; Haase, Christoph; Ebert, Markus; Dahmke, Andreas (2011-01-01). "Effects of the variances of input parameters on water-mineral interactions during CO2 sequestration modeling". Energy Procedia. 10th International Conference on Greenhouse Gas Control Technologies. 4: 3770–3777. doi: 10.1016/j.egypro.2011.02.311 .
- ↑ Haase, Christoph; Dethlefsen, Frank; Ebert, Markus; Dahmke, Andreas (2013-06-01). "Uncertainty in geochemical modelling of CO2 and calcite dissolution in NaCl solutions due to different modelling codes and thermodynamic databases". Applied Geochemistry. 33: 306–317. doi:10.1016/j.apgeochem.2013.03.001.
- 1 2 Haase, Christoph; Ebert, Markus; Dethlefsen, Frank (2016-04-01). "Uncertainties of geochemical codes and thermodynamic databases for predicting the impact of carbon dioxide on geologic formations". Applied Geochemistry. 67: 81–92. doi:10.1016/j.apgeochem.2016.01.008.
- ↑ Muller, B., 2004, CHEMEQL V3.0, A program to calculate chemical speciation equilibria, titrations, dissolution, precipitation, adsorption, kinetics, pX-pY diagrams, solubility diagrams. Limnological Research Center EAWAG/ETH, Kastanienbaum, Switzerland
- ↑ van der Lee, J., and L. De Windt, 2000, CHESS, another speciation and complexation computer code. Technical Report no. LHM/RD/93/39, Ecole des Mines de Paris, Fontainebleau
- ↑ Reed, M.H., 1982, Calculation of multicomponent chemical equilibria and reaction processes in systems involving minerals, gases, and aqueous phase. Geochimica et Cosmochemica Acta46, 513-528.
- ↑ Steefel, C.I. and A.C. Lasaga, 1994, A coupled model for transport of multiple chemical species and kinetic precipitation/dissolution reactions with application to reactive flow in single phase hydrothermal systems. American Journal of Science294, 529-592
- ↑ Steefel, C.I., 2001, GIMRT, Version 1.2: Software for modeling multicomponent, multidimensional reactive transport, User's Guide. Report UCRL-MA-143182, Lawrence Livermore National Laboratory, Livermore, California.
- ↑ Wolery, T.J., 1992a, EQ3/EQ6, a software package for geochemical modeling of aqueous systems, package overview and installation guide (version 7.0). Lawrence Livermore National Laboratory Report UCRL-MA-110662(1).
- ↑ Shaff, J.E., B.A. Schultz, E.J. Craft, R.T. Clark, and L.V. Kochian, 2010, GEOCHEM-EZ: a chemical speciation program with greater power and flexibility. Plant Soil330(1), 207-214
- ↑ Bethke, C.M., B. Farrell, and M. Sharifi, 2021, The Geochemist's Workbench Release 15 (five volumes). Aqueous Solutions LLC, Champaign, IL USA
- ↑ Kulik, D.A., 2002, Gibbs energy minimization approach to model sorption equilibria at the mineral-water interface: Thermodynamic relations for multi-site surface complexation. American Journal of Science302, 227-279
- ↑ Cheng, H.P. and G.T. Yeh, 1998, Development of a three-dimensional model of subsurface flow, heat transfer, and reactive chemical transport: 3DHYDROGEOCHEM. Journal of Contaminant Hydrology34, 47-83
- ↑ Westall, J.C., J.L. Zachary and F.F.M. Morel, 1976, MINEQL, a computer program for the calculation of chemical equilibrium composition of aqueous systems. Technical Note 18, R.M. Parsons Laboratory, Department of Civil and Environmental Engineering, Massachusetts Institute of Technology, Cambridge, MA.
- ↑ Scherer, W.D. and D.C. McAvoy, 1994, MINEQL+, A Chemical Equilibrium Program for Personal Computers, User's Manual, version 3.0. Environmental Research Software, Inc., Hallowell, ME.
- ↑ Allison, J.D., D.S. Brown and K.J. Novo-Gradac, 1991, MINTEQA2/ PRODEFA2, a geochemical assessment model for environmental systems, version 3.0 user's manual. US Environmental Protectiona Agency Report EPA/600/3-91/021.
- ↑ "ORCHESTRA | Geochemical and Transport Modelling" . Retrieved 2022-09-29.
- ↑ Parkhurst, D.L., 1995, User's Guide to PHREEQC, a computer model for speciation, reaction-path, advective-transport and inverse geochemical calculations. US Geological Survey Water-Resources Investigations Report 95-4227.
- ↑ Parkhurst, D.L. and C.A.J. Appelo, 1999, User's Guide to PHREEQC (version 2), a computer program for speciation, batch-reaction, one-dimensional transport and inverse geochemical calculations. US Geological Survey Water-Resources Investigations Report 99-4259.
- ↑ Leal, A.M.M., Kulik, D.A., Smith, W.R. and Saar, M.O., 2017, An overview of computational methods for chemical equilibrium and kinetic calculations for geochemical and reactive transport modeling. Pure and Applied Chemistry. 89 (5), 145–166.
- ↑ Perkins, E.H., 1992, Integration of intensive variable diagrams and fluid phase equilibria with SOLMINEQ.88 pc/shell. In Y.K. Kharaka and A.S. Maest (eds.), Water-Rock Interaction, Balkema, Rotterdam, p. 1079-1081.
- ↑ Xu, T., E.L. Sonnenthal, N. Spycher and K. Pruess, 2004, TOUGHREACT user's guide: A simulation program for non-isothermal multiphase reactive geochemical transport in variably saturated geologic media. Report LBNL-55460, Lawrence Berkeley National Laboratory, Berkeley, California.
- ↑ hem.bredband.net/b108693/-VisualMINTEQ_references.pdf
- ↑ Ball, J.W. and D.K. Nordstrom, 1991, User's manual for WATEQ4F, with revised thermodynamic data base and test cases for calculating speciation of major, trace, and redox elements in natural waters. US Geological Survey Open File Report 91-183.
- ↑ Tipping E., 1994, WHAM - a chemical equilibrium model and computer code for waters, sediments and soils incorporating a discrete site/electrostatic model of ion-binding by humic substances. Computers and Geosciences20, 973-1023.
- ↑ "Water Resources Geochemical Software". water.usgs.gov. Retrieved 2020-09-25.