5α-Reductase 2 deficiency (5αR2D) is an autosomal recessive condition caused by a mutation in SRD5A2, a gene encoding the enzyme 5α-reductase type 2 (5αR2). The condition is rare, affects only genetic males, and has a broad spectrum.
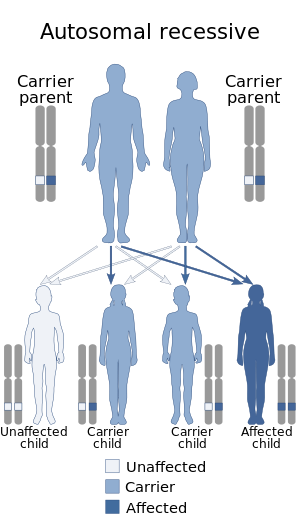
Congenital adrenal hyperplasia (CAH) is a group of autosomal recessive disorders characterized by impaired cortisol synthesis. It results from the deficiency of one of the five enzymes required for the synthesis of cortisol in the adrenal cortex. Most of these disorders involve excessive or deficient production of hormones such as glucocorticoids, mineralocorticoids, or sex steroids, and can alter development of primary or secondary sex characteristics in some affected infants, children, or adults. It is one of the most common autosomal recessive disorders in humans.

Lipoid congenital adrenal hyperplasia is an endocrine disorder that is an uncommon and potentially lethal form of congenital adrenal hyperplasia (CAH). It arises from defects in the earliest stages of steroid hormone synthesis: the transport of cholesterol into the mitochondria and the conversion of cholesterol to pregnenolone—the first step in the synthesis of all steroid hormones. Lipoid CAH causes mineralocorticoid deficiency in affected infants and children. Male infants are severely undervirilized causing their external genitalia to look feminine. The adrenals are large and filled with lipid globules derived from cholesterol.

Congenital adrenal hyperplasia due to 3β-hydroxysteroid dehydrogenase deficiency is an uncommon form of congenital adrenal hyperplasia (CAH) resulting from a mutation in the gene for one of the key enzymes in cortisol synthesis by the adrenal gland, 3β-hydroxysteroid dehydrogenase (3β-HSD) type II (HSD3B2). As a result, higher levels of 17α-hydroxypregnenolone appear in the blood with adrenocorticotropic hormone (ACTH) challenge, which stimulates adrenal corticosteroid synthesis.
Congenital adrenal hyperplasia due to 17α-hydroxylase deficiency is an uncommon form of congenital adrenal hyperplasia resulting from a defect in the gene CYP17A1, which encodes for the enzyme 17α-hydroxylase. It causes decreased synthesis of cortisol and sex steroids, with resulting increase in mineralocorticoid production. Thus, common symptoms include mild hypocortisolism, ambiguous genitalia in genetic males or failure of the ovaries to function at puberty in genetic females, and hypokalemic hypertension (respectively). However, partial (incomplete) deficiency is notable for having inconsistent symptoms between patients, and affected genetic (XX) females may be wholly asymptomatic except for infertility.
Kallmann syndrome (KS) is a genetic disorder that prevents a person from starting or fully completing puberty. Kallmann syndrome is a form of a group of conditions termed hypogonadotropic hypogonadism. To distinguish it from other forms of hypogonadotropic hypogonadism, Kallmann syndrome has the additional symptom of a total lack of sense of smell (anosmia) or a reduced sense of smell. If left untreated, people will have poorly defined secondary sexual characteristics, show signs of hypogonadism, almost invariably are infertile and are at increased risk of developing osteoporosis. A range of other physical symptoms affecting the face, hands and skeletal system can also occur.

Cytochrome P450 17A1 is an enzyme of the hydroxylase type that in humans is encoded by the CYP17A1 gene on chromosome 10. It is ubiquitously expressed in many tissues and cell types, including the zona reticularis and zona fasciculata of the adrenal cortex as well as gonadal tissues. It has both 17α-hydroxylase and 17,20-lyase activities, and is a key enzyme in the steroidogenic pathway that produces progestins, mineralocorticoids, glucocorticoids, androgens, and estrogens. More specifically, the enzyme acts upon pregnenolone and progesterone to add a hydroxyl (-OH) group at carbon 17 position (C17) of the steroid D ring, or acts upon 17α-hydroxyprogesterone and 17α-hydroxypregnenolone to split the side-chain off the steroid nucleus.

Sexual differentiation in humans is the process of development of sex differences in humans. It is defined as the development of phenotypic structures consequent to the action of hormones produced following gonadal determination. Sexual differentiation includes development of different genitalia and the internal genital tracts and body hair plays a role in sex identification.

Disorders of sex development (DSDs), also known as differences in sex development, diverse sex development and variations in sex characteristics (VSC), are congenital conditions affecting the reproductive system, in which development of chromosomal, gonadal, or anatomical sex is atypical.

Aromatase deficiency is a rare condition characterized by extremely low levels or complete absence of the enzyme aromatase activity in the body. It is an autosomal recessive disease resulting from various mutations of gene CYP19 (P450arom) which can lead to ambiguous genitalia and delayed puberty in females, continued linear growth into adulthood and osteoporosis in males and virilization in pregnant mothers. As of 2020, fewer than 15 cases have been identified in genetically male individuals and at least 30 cases in genetically female individuals.
Hypergonadotropic hypogonadism (HH), also known as primary or peripheral/gonadal hypogonadism or primary gonadal failure, is a condition which is characterized by hypogonadism which is due to an impaired response of the gonads to the gonadotropins, follicle-stimulating hormone (FSH) and luteinizing hormone (LH), and in turn a lack of sex steroid production. As compensation and the lack of negative feedback, gonadotropin levels are elevated. Individuals with HH have an intact and functioning hypothalamus and pituitary glands so they are still able to produce FSH and LH. HH may present as either congenital or acquired, but the majority of cases are of the former nature. HH can be treated with hormone replacement therapy.
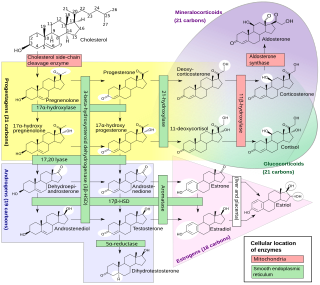
An inborn error of steroid metabolism is an inborn error of metabolism due to defects in steroid metabolism.

Leydig cell hypoplasia (LCH), also known as Leydig cell agenesis, is a rare autosomal recessive genetic and endocrine syndrome affecting an estimated 1 in 1,000,000 genetic males. It is characterized by an inability of the body to respond to luteinizing hormone (LH), a gonadotropin which is normally responsible for signaling Leydig cells of the testicles to produce testosterone and other androgen sex hormones. The condition manifests itself as pseudohermaphroditism, hypergonadotropic hypogonadism, reduced or absent puberty, and infertility.
Follicle-stimulating hormone (FSH) insensitivity, or ovarian insensitivity to FSH in females, also referable to as ovarian follicle hypoplasia or granulosa cell hypoplasia in females, is a rare autosomal recessive genetic and endocrine syndrome affecting both females and males, with the former presenting with much greater severity of symptomatology. It is characterized by a resistance or complete insensitivity to the effects of follicle-stimulating hormone (FSH), a gonadotropin which is normally responsible for the stimulation of estrogen production by the ovaries in females and maintenance of fertility in both sexes. The condition manifests itself as hypergonadotropic hypogonadism, reduced or absent puberty, amenorrhea, and infertility in females, whereas males present merely with varying degrees of infertility and associated symptoms.

Seviteronel is an experimental cancer medication which is under development by Viamet Pharmaceuticals and Innocrin Pharmaceuticals for the treatment of prostate cancer and breast cancer. It is a nonsteroidal CYP17A1 inhibitor and works by inhibiting the production of androgens and estrogens in the body. As of July 2017, seviteronel is in phase II clinical trials for both prostate cancer and breast cancer. In January 2016, it was designated fast-track status by the United States Food and Drug Administration for prostate cancer. In April 2017, seviteronel received fast-track designation for breast cancer as well.

A CYP17A1 inhibitor is a type of drug which inhibits the enzyme CYP17A1. It may inhibit both of the functions of the enzyme, 17α-hydroxylase and 17,20-lyase, or may be selective for inhibition of one of these two functions. These drugs prevent the conversion of pregnane steroids into androgens like testosterone and therefore are androgen biosynthesis inhibitors and functional antiandrogens. Examples of CYP17A1 inhibitors include the older drug ketoconazole and the newer drugs abiraterone acetate, orteronel, galeterone, and seviteronel. The CYP17A1 inhibitors that have been marketed, like abiraterone acetate, are used mainly in the treatment of prostate cancer. CYP17A1 inhibitors that are not selective for inhibition of 17,20-lyase must be combined with a glucocorticoid such as prednisone in order to avoid adrenal insufficiency and mineralocorticoid excess caused by prevention of cortisol production.
An androgen synthesis inhibitor is a type of drug which inhibits the enzymatic synthesis of androgens, such as testosterone and dihydrotestosterone (DHT). They include:
Cytochrome b5 deficiency is a rare condition and form of isolated 17,20-lyase deficiency caused by deficiency in cytochrome b5, a small hemoprotein that acts as an allosteric factor to facilitate the interaction of CYP17A1 (17α-hydroxylase/17,20-lyase) with P450 oxidoreductase (POR), thereby allowing for the 17,20-lyase activity of CYP17A1. The condition affects both adrenal and gonadal androgen biosynthesis and results in male pseudohermaphroditism. The principal biological role of cytochrome b5 is reduction of methemoglobin, so cytochrome b5 deficiency can also result in elevated methemoglobin levels and/or methemoglobinemia, similarly to deficiency of cytochrome b5 reductase.
Cytochrome P450 oxidoreductase deficiency (PORD) is a rare disease and inborn error of metabolism caused by deficiency of cytochrome P450 oxidoreductase (POR). POR is a 2-flavin protein that is responsible for the transfer of electrons from NADPH to all 50 microsomal cytochrome P450 (CYP450) enzymes. This includes the steroidogenic enzymes CYP17A1 (17α-hydroxylase/17,20-lyase), CYP19A1 (aromatase), and CYP21A2 (21-hydroxylase); CYP26B1 ; and the hepatic drug-metabolizing CYP450 enzymes, among many other CYP450 enzymes. Symptoms of severe forms of PORD include ambiguous genitalia in males and females, congenital adrenal hyperplasia, cortisol deficiency, and Antley–Bixler skeletal malformation syndrome (ABS), while symptoms of mild forms include polycystic ovary syndrome in women and hypogonadism in men. Maternal virilization also occurs in severe forms, due to aromatase deficiency in the placenta. Virilization of female infants in PORD may also be caused by alternative biosynthesis of 5α-dihydrotestosterone via the so-called "androgen backdoor pathway". The ABS component of severe forms of PORD is probably caused by CYP26B1 deficiency, which results in retinoic acid excess and defects during skeletal embryogenesis. All forms of PORD in humans are likely partial, as POR knockout in mice results in death during prenatal development.
The androgen backdoor pathway is a collective name for all metabolic pathways where clinically relevant androgens are synthesized from 21-carbon steroids (pregnanes) by their 5α-reduction with a roundabout of testosterone and/or androstenedione.