Polyurethane refers to a class of polymers composed of organic units joined by carbamate (urethane) links. In contrast to other common polymers such as polyethylene and polystyrene, polyurethane is produced from a wide range of starting materials. This chemical variety produces polyurethanes with different chemical structures leading to many different applications. These include rigid and flexible foams, and coatings, adhesives, electrical potting compounds, and fibers such as spandex and polyurethane laminate (PUL). Foams are the largest application accounting for 67% of all polyurethane produced in 2016.

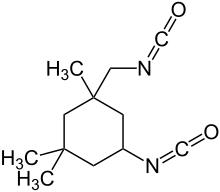
In organic chemistry, isocyanate is the functional group with the formula R−N=C=O. Organic compounds that contain an isocyanate group are referred to as isocyanates. An organic compound with two isocyanate groups is known as a diisocyanate. Diisocyanates are manufactured for the production of polyurethanes, a class of polymers.

Epoxy is the family of basic components or cured end products of epoxy resins. Epoxy resins, also known as polyepoxides, are a class of reactive prepolymers and polymers which contain epoxide groups. The epoxide functional group is also collectively called epoxy. The IUPAC name for an epoxide group is an oxirane.

In materials science, a thermosetting polymer, often called a thermoset, is a polymer that is obtained by irreversibly hardening ("curing") a soft solid or viscous liquid prepolymer (resin). Curing is induced by heat or suitable radiation and may be promoted by high pressure or mixing with a catalyst. Heat is not necessarily applied externally, and is often generated by the reaction of the resin with a curing agent. Curing results in chemical reactions that create extensive cross-linking between polymer chains to produce an infusible and insoluble polymer network.

Polyurea is a type of elastomer that is derived from the reaction product of an isocyanate component and an amine component. The isocyanate can be aromatic or aliphatic in nature. It can be monomer, polymer, or any variant reaction of isocyanates, quasi-prepolymer or a prepolymer. The prepolymer, or quasi-prepolymer, can be made of an amine-terminated polymer resin, or a hydroxyl-terminated polymer resin.
In organic chemistry, a polyol is an organic compound containing multiple hydroxyl groups. The term "polyol" can have slightly different meanings depending on whether it is used in food science or polymer chemistry. Polyols containing two, three and four hydroxyl groups are diols, triols, and tetrols, respectively.

Toluene diisocyanate (TDI) is an organic compound with the formula CH3C6H3(NCO)2. Two of the six possible isomers are commercially important: 2,4-TDI (CAS: 584-84-9) and 2,6-TDI (CAS: 91-08-7). 2,4-TDI is produced in the pure state, but TDI is often marketed as 80/20 and 65/35 mixtures of the 2,4 and 2,6 isomers respectively. It is produced on a large scale, accounting for 34.1% of the global isocyanate market in 2000, second only to MDI. Approximately 1.4 billion kilograms were produced in 2000. All isomers of TDI are colorless, although commercial samples can appear yellow.

Methylene diphenyl diisocyanate (MDI) is an aromatic diisocyanate. Three isomers are common, varying by the positions of the isocyanate groups around the rings: 2,2′-MDI, 2,4′-MDI, and 4,4′-MDI. The 4,4′ isomer is most widely used, and is also known as 4,4′-diphenylmethane diisocyanate. This isomer is also known as Pure MDI. MDI reacts with polyols in the manufacture of polyurethane. It is the most produced diisocyanate, accounting for 61.3% of the global market in the year 2000.

Hot-melt adhesive (HMA), also known as hot glue, is a form of thermoplastic adhesive that is commonly sold as solid cylindrical sticks of various diameters designed to be applied using a hot glue gun. The gun uses a continuous-duty heating element to melt the plastic glue, which the user pushes through the gun either with a mechanical trigger mechanism on the gun, or with direct finger pressure. The glue squeezed out of the heated nozzle is initially hot enough to burn and even blister skin. The glue is sticky when hot, and solidifies in a few seconds to one minute. Hot-melt adhesives can also be applied by dipping or spraying, and are popular with hobbyists and crafters both for affixing and as an inexpensive alternative to resin casting.
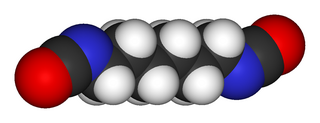
Hexamethylene diisocyanate (HDI) is the organic compound with the formula (CH2)6(NCO)2. It is classified as an diisocyanate. It is a colorless liquid. It has sometimes been called HMDI but this not usually done to avoid confusion with Hydrogenated MDI.
A thermoset polymer matrix is a synthetic polymer reinforcement where polymers act as binder or matrix to secure in place incorporated particulates, fibres or other reinforcements. They were first developed for structural applications, such as glass-reinforced plastic radar domes on aircraft and graphite-epoxy payload bay doors on the Space Shuttle.
In polymer chemistry, the term prepolymer or pre-polymer, refers to a monomer or system of monomers that have been reacted to an intermediate-molecular mass state. This material is capable of further polymerization by reactive groups to a fully cured, high-molecular-mass state. As such, mixtures of reactive polymers with un-reacted monomers may also be referred to as pre-polymers. The term "pre-polymer" and "polymer precursor" may be interchanged.
Polyaspartic ester chemistry was first introduced in the early 1990s making it a relatively new technology. The patents were issued to Bayer in Germany and Miles Corporation in the United States. It utilizes the aza-Michael addition reaction. These products are then used in coatings, adhesives, sealants and elastomers. Pure polyurea reacts extremely quickly making them almost unusable without plural component spray equipment. Polyaspartic technology utilizes a partially blocked amine to react more slowly with the isocyanates and thus produce a modified polyurea. The amine/diamine or even triamine functional coreactant for aliphatic polyisocyanate is typically reacted with a maleate. Polyaspartic esters (PAE) initially found use in conventional solvent-borne two-component polyurethane coatings.
Moisture-cure polyurethanes -- or polyurethane prepolymer -- are isocyanate-terminated prepolymers that are formulated to cure with ambient water. Cured PURs are segmented copolymer polyurethane-ureas exhibiting microphase-separated morphologies. One phase is derived from a typically flexible polyol that is generally referred to as the “soft phase”. Likewise the corresponding “hard phase” is born from the di- or polyisocyanates that through water reaction produce a highly crosslinked material with softening temperature well above room temperature.
4,4'-Diaminodicyclohexylmethane is the name for organic compounds with the formula CH2(C6H10NH2)2. It is classified as a diamine. In the epoxy industry it is often referred to as PACM, short for para-diaminodicyclohexylmethane. It is used as a curing agent for epoxy resins It finds particular use in epoxy flooring. Another use is to produce diisocyanates, which are precursors to polyurethanes. The mixture is a colorless solid, but typical samples are yellowish and oily. The compound is produced as a mixture of three isomers by the hydrogenation of methylenedianiline. These isomers are, in decreasing order of their yield from the hydrogenation, trans-trans, cis-trans, and a small amount of cis-cis.

Isophorone diamine (usually shortened to IPDA) is a chemical compound and specifically a diamine with the formula (CH3)3C6H7(NH2)(CH2NH2). It is a colorless liquid. It is a precursor to polymers and coatings.

Tetramethylxylylene diisocyanate (TMXDI) is an organic compound with the formula C6H4(CMe2NCO)2 (Me = CH3). Introduced in the 1980s by American Cyanamid, the molecule features two isocyanate groups. TMXDI is generally classified as an aliphatic isocyanate, which are generally more UV stable than their aromatic counterparts.

Dimethylolpropionic acid (DMPA) is a chemical compound that has the full IUPAC name of 2,2-bis(hydroxymethyl)propionic acid and is an organic compound with one carboxyl and two hydroxy groups. It has the CAS Registry Number of 4767-03-7.
Hydrogenated MDI (H12MDI or 4,4′-diisocyanato dicyclohexylmethane) is an organic compound in the class known as isocyanates. More specifically, it is an aliphatic diisocyanate. It is a water white liquid at room temperature and is manufactured in relatively small quantities. It is also known as 4,4'-methylenedi(cyclohexyl isocyanate) or methylene bis(4-cyclohexylisocyanate) and has the formula CH2[(C6H10)NCO]2.
Blocked isocyanates are organic compounds that have their isocyanate functionality chemically blocked to control reactivity. They are the product of an isocyanate moiety and a suitable blocking agent. It may also be a polyurethane prepolymer that is NCO terminated but this functionality has also been chemically reacted with a blocking agent. They are usually used in polyurethane applications but not always. They are extensively used in industrial applications such as coatings, sealants and adhesives.