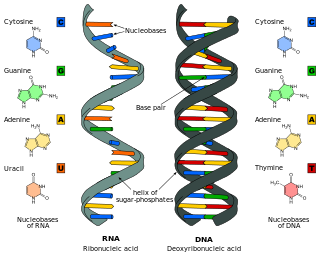
Nucleic acids are biopolymers, macromolecules, essential to all known forms of life. They are composed of nucleotides, which are the monomers made of three components: a 5-carbon sugar, a phosphate group and a nitrogenous base. The two main classes of nucleic acids are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). If the sugar is ribose, the polymer is RNA; if the sugar is the ribose derivative deoxyribose, the polymer is DNA.
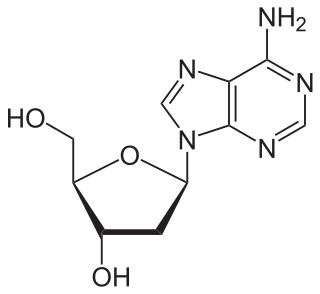
Nucleosides are glycosylamines that can be thought of as nucleotides without a phosphate group. A nucleoside consists simply of a nucleobase and a five-carbon sugar whereas a nucleotide is composed of a nucleobase, a five-carbon sugar, and one or more phosphate groups. In a nucleoside, the anomeric carbon is linked through a glycosidic bond to the N9 of a purine or the N1 of a pyrimidine. Nucleotides are the molecular building-blocks of DNA and RNA.
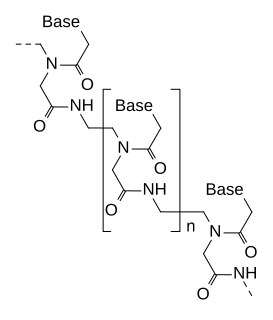
Peptide nucleic acid (PNA) is an artificially synthesized polymer similar to DNA or RNA.
Oligonucleotides are short DNA or RNA molecules, oligomers, that have a wide range of applications in genetic testing, research, and forensics. Commonly made in the laboratory by solid-phase chemical synthesis, these small bits of nucleic acids can be manufactured as single-stranded molecules with any user-specified sequence, and so are vital for artificial gene synthesis, polymerase chain reaction (PCR), DNA sequencing, molecular cloning and as molecular probes. In nature, oligonucleotides are usually found as small RNA molecules that function in the regulation of gene expression, or are degradation intermediates derived from the breakdown of larger nucleic acid molecules.
Antisense therapy is a form of treatment that uses antisense oligonucleotides (ASOs) to target messenger RNA (mRNA). ASOs are capable of altering mRNA expression through a variety of mechanisms, including ribonuclease H mediated decay of the pre-mRNA, direct steric blockage, and exon content modulation through splicing site binding on pre-mRNA. Several ASOs have been approved in the United States, the European Union, and elsewhere.
In molecular biology, a hybridization probe(HP) is a fragment of DNA or RNA of usually 15–10000 nucleotide long which can be radioactively or fluorescently labeled. HP can be used to detect the presence of nucleotide sequences in analyzed RNA or DNA that are complementary to the sequence in the probe. The labeled probe is first denatured into single stranded DNA (ssDNA) and then hybridized to the target ssDNA or RNA immobilized on a membrane or in situ.
Deoxyribozymes, also called DNA enzymes, DNAzymes, or catalytic DNA, are DNA oligonucleotides that are capable of performing a specific chemical reaction, often but not always catalytic. This is similar to the action of other biological enzymes, such as proteins or ribozymes . However, in contrast to the abundance of protein enzymes in biological systems and the discovery of biological ribozymes in the 1980s, there is only little evidence for naturally occurring deoxyribozymes. Deoxyribozymes should not be confused with DNA aptamers which are oligonucleotides that selectively bind a target ligand, but do not catalyze a subsequent chemical reaction.
Threose nucleic acid (TNA) is an artificial genetic polymer in which the natural five-carbon ribose sugar found in RNA has been replaced by an unnatural four-carbon threose sugar. Invented by Albert Eschenmoser as part of his quest to explore the chemical etiology of RNA, TNA has become an important synthetic genetic polymer (XNA) due to its ability to efficiently base pair with complementary sequences of DNA and RNA. However, unlike DNA and RNA, TNA is completely refractory to nuclease digestion, making it a promising nucleic acid analog for therapeutic and diagnostic applications.

Molecular beacons, or molecular beacon probes, are oligonucleotide hybridization probes that can report the presence of specific nucleic acids in homogenous solutions. Molecular beacons are hairpin-shaped molecules with an internally quenched fluorophore whose fluorescence is restored when they bind to a target nucleic acid sequence. This is a novel non-radioactive method for detecting specific sequences of nucleic acids. They are useful in situations where it is either not possible or desirable to isolate the probe-target hybrids from an excess of the hybridization probes.
In molecular biology and genetics, the sense of a nucleic acid molecule, particularly of a strand of DNA or RNA, refers to the nature of the roles of the strand and its complement in specifying a sequence of amino acids. Depending on the context, sense may have slightly different meanings. For example, negative-sense strand of DNA is equivalent to the template strand, whereas the positive-sense strand is the non-template strand whose nucleotide sequence is equivalent to the sequence of the mRNA transcript.

Systematic evolution of ligands by exponential enrichment (SELEX), also referred to as in vitro selection or in vitro evolution, is a combinatorial chemistry technique in molecular biology for producing oligonucleotides of either single-stranded DNA or RNA that specifically bind to a target ligand or ligands. These single-stranded DNA or RNA are commonly referred to as aptamers. Although SELEX has emerged as the most commonly used name for the procedure, some researchers have referred to it as SAAB and CASTing SELEX was first introduced in 1990. In 2015, a special issue was published in the Journal of Molecular Evolution in the honor of quarter century of the discovery of SELEX.
Oligonucleotide synthesis is the chemical synthesis of relatively short fragments of nucleic acids with defined chemical structure (sequence). The technique is extremely useful in current laboratory practice because it provides a rapid and inexpensive access to custom-made oligonucleotides of the desired sequence. Whereas enzymes synthesize DNA and RNA only in a 5' to 3' direction, chemical oligonucleotide synthesis does not have this limitation, although it is most often carried out in the opposite, 3' to 5' direction. Currently, the process is implemented as solid-phase synthesis using phosphoramidite method and phosphoramidite building blocks derived from protected 2'-deoxynucleosides, ribonucleosides, or chemically modified nucleosides, e.g. LNA or BNA.

Nucleic acid analogues are compounds which are analogous to naturally occurring RNA and DNA, used in medicine and in molecular biology research. Nucleic acids are chains of nucleotides, which are composed of three parts: a phosphate backbone, a pentose sugar, either ribose or deoxyribose, and one of four nucleobases. An analogue may have any of these altered. Typically the analogue nucleobases confer, among other things, different base pairing and base stacking properties. Examples include universal bases, which can pair with all four canonical bases, and phosphate-sugar backbone analogues such as PNA, which affect the properties of the chain . Nucleic acid analogues are also called Xeno Nucleic Acid and represent one of the main pillars of xenobiology, the design of new-to-nature forms of life based on alternative biochemistries.
Santaris Pharma A/S was a biopharmaceutical company founded in 2003 in Copenhagen, Denmark. The company also had a branch in San Diego, California that opened in 2009. Created by a merger between Cureon and Pantheco, Santaris developed RNA-targeted medicines using a Locked Nucleic Acid (LNA) Drug Platform and Drug Development Engine.
Twisted intercalating nucleic acid (TINA) is a nucleic acid molecule that, when added to triplex-forming oligonucleotides (TFOs), stabilizes Hoogsteen triplex DNA formation from double-stranded DNA (dsDNA) and TFOs. Its ability to twist around a triple bond increases ease of intercalation within double stranded DNA in order to form triplex DNA. Certain configurations have been shown to stabilize Watson-Crick antiparallel duplex DNA. TINA-DNA primers have been shown to increase the specificity of binding in PCR. The use of TINA insertions in G-quadruplexes has also been shown to enhance anti-HIV-1 activity. TINA stabilized PT demonstrates improved sensitivity and specificity of DNA based clinical diagnostic assays.
A bridged nucleic acid (BNA) is a modified RNA nucleotide. They are sometimes also referred to as constrained or inaccessible RNA molecules. BNA monomers can contain a five-membered, six-membered or even a seven-membered bridged structure with a "fixed" C3'-endo sugar puckering. The bridge is synthetically incorporated at the 2', 4'-position of the ribose to afford a 2', 4'-BNA monomer. The monomers can be incorporated into oligonucleotide polymeric structures using standard phosphoamidite chemistry. BNAs are structurally rigid oligo-nucleotides with increased binding affinities and stability.
Anti-miRNA oligonucleotides have many uses in cellular mechanics. These synthetically designed molecules are used to neutralize microRNA (miRNA) function in cells for desired responses. miRNA are complementary sequences to mRNA that are involved in the cleavage of RNA or the suppression of the translation. By controlling the miRNA that regulate mRNAs in cells, AMOs can be used as further regulation as well as for therapeutic treatment for certain cellular disorders. This regulation can occur through a steric blocking mechanism as well as hybridization to miRNA. These interactions, within the body between miRNA and AMOs, can be for therapeutics in disorders in which over/under expression occurs or aberrations in miRNA lead to coding issues. Some of the miRNA linked disorders that are encountered in the humans include cancers, muscular diseases, autoimmune disorders, and viruses. In order to determine the functionality of certain AMOs, the AMO/miRNA binding expression must be measured against the expressions of the isolated miRNA. The direct detection of differing levels of genetic expression allow the relationship between AMOs and miRNAs to be shown. This can be detected through luciferase activity. Understanding the miRNA sequences involved in these diseases can allow us to use anti miRNA Oligonucleotides to disrupt pathways that lead to the under/over expression of proteins of cells that can cause symptoms for these diseases.
A hybridization assay comprises any form of quantifiable hybridization i.e. the quantitative annealing of two complementary strands of nucleic acids, known as nucleic acid hybridization.
Miravirsen is an experimental drug for the treatment of hepatitis C, being developed by Santaris Pharma. As of 2017 it was in Phase II clinical trials.
Gapmers are short DNA antisense oligonucleotide structures with RNA-like segments on both sides of the sequence. These linear pieces of genetic information are designed to hybridize to a target piece of RNA and silence the gene through the induction of RNase H cleavage. Binding of the gapmer to the target has a higher affinity due to the modified RNA flanking regions, as well as resistance to degradation by nucleases. Gapmers are currently being developed as therapeutics for a variety of cancers, viruses, and other chronic genetic disorders.





