
In chemistry, a hydrogen bond is primarily an electrostatic force of attraction between a hydrogen (H) atom which is covalently bonded to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a lone pair of electrons—the hydrogen bond acceptor (Ac). Such an interacting system is generally denoted Dn−H···Ac, where the solid line denotes a polar covalent bond, and the dotted or dashed line indicates the hydrogen bond. The most frequent donor and acceptor atoms are the period 2 elements nitrogen (N), oxygen (O), and fluorine (F).
In chemistry, a disulfide is a compound containing a R−S−S−R′ functional group or the S2−
2 anion. The linkage is also called an SS-bond or sometimes a disulfide bridge and usually derived from two thiol groups.
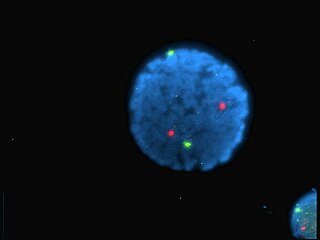
A fluorophore is a fluorescent chemical compound that can re-emit light upon light excitation. Fluorophores typically contain several combined aromatic groups, or planar or cyclic molecules with several π bonds.

In organic chemistry, the Michael reaction or Michael 1,4 addition is a reaction between a Michael donor and a Michael acceptor to produce a Michael adduct by creating a carbon-carbon bond at the acceptor's β-carbon. It belongs to the larger class of conjugate additions and is widely used for the mild formation of carbon-carbon bonds.
In chemical synthesis, click chemistry is a class of simple, atom-economy reactions commonly used for joining two molecular entities of choice. Click chemistry is not a single specific reaction, but describes a way of generating products that follow examples in nature, which also generates substances by joining small modular units. In many applications, click reactions join a biomolecule and a reporter molecule. Click chemistry is not limited to biological conditions: the concept of a "click" reaction has been used in chemoproteomic, pharmacological, biomimetic and molecular machinery applications. However, they have been made notably useful in the detection, localization and qualification of biomolecules.
Dynamic covalent chemistry (DCvC) is a synthetic strategy employed by chemists to make complex molecular and supramolecular assemblies from discrete molecular building blocks. DCvC has allowed access to complex assemblies such as covalent organic frameworks, molecular knots, polymers, and novel macrocycles. Not to be confused with dynamic combinatorial chemistry, DCvC concerns only covalent bonding interactions. As such, it only encompasses a subset of supramolecular chemistries.
A photoswitch is a type of molecule that can change its structural geometry and chemical properties upon irradiation with electromagnetic radiation. Although often used interchangeably with the term molecular machine, a switch does not perform work upon a change in its shape whereas a machine does. However, photochromic compounds are the necessary building blocks for light driven molecular motors and machines. Upon irradiation with light, photoisomerization about double bonds in the molecule can lead to changes in the cis- or trans- configuration. These photochromic molecules are being considered for a range of applications.

2-Mercaptoethanol (also β-mercaptoethanol, BME, 2BME, 2-ME or β-met) is the chemical compound with the formula HOCH2CH2SH. ME or βME, as it is commonly abbreviated, is used to reduce disulfide bonds and can act as a biological antioxidant by scavenging hydroxyl radicals (amongst others). It is widely used because the hydroxyl group confers solubility in water and lowers the volatility. Due to its diminished vapor pressure, its odor, while unpleasant, is less objectionable than related thiols.
Bioconjugation is a chemical strategy to form a stable covalent link between two molecules, at least one of which is a biomolecule.
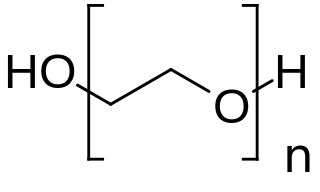
PEGylation is the process of both covalent and non-covalent attachment or amalgamation of polyethylene glycol polymer chains to molecules and macrostructures, such as a drug, therapeutic protein or vesicle, which is then described as PEGylated. PEGylation affects the resulting derivatives or aggregates interactions, which typically slows down their coalescence and degradation as well as elimination in vivo.

The thiol-yne reaction is an organic reaction between a thiol and an alkyne. The reaction product is an alkenyl sulfide. The reaction was first reported in 1949 with thioacetic acid as reagent and rediscovered in 2009. It is used in click chemistry and in polymerization, especially with dendrimers.
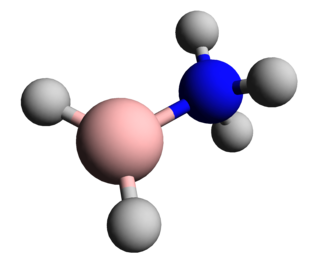
In chemistry, a boranylium ion is an inorganic cation with the chemical formula BR+
2, where R represents a non-specific substituent. Being electron-deficient, boranylium ions form adducts with Lewis bases. Boranylium ions have historical names that depend on the number of coordinated ligands:

Mercury(II) hydride is an inorganic compound with the chemical formula HgH
2. It is both thermodynamically and kinetically unstable at ambient temperature, and as such, little is known about its bulk properties. However, it known as a white, crystalline solid, which is kinetically stable at temperatures below −125 °C (−193 °F), which was synthesised for the first time in 1951.
Radiofluorination is the process by which a radioactive isotope of fluorine is attached to a molecule and is preferably performed by nucleophilic substitution using nitro or halogens as leaving groups. Fluorine-18 is the most common isotope used for this procedure. This is due to its 97% positron emission and relatively long 109.8 min half-life. The half-life allows for a long enough time to be incorporated into the molecule and be used without causing exceedingly harmful effects. This process has many applications especially with the use of positron emission tomography (PET) as the aforementioned low positron energy is able to yield a high resolution in PET imaging.

3-Arylpropiolonitriles (APN) belong to a class of electron-deficient alkyne derivatives substituted by two electron-withdrawing groups – a nitrile and an aryl moieties. Such activation results in improved selectivity towards highly reactive thiol-containing molecules, namely cysteine residues in proteins. APN-based modification of proteins was reported to surpass several important drawbacks of existing strategies in bioconjugation, notably the presence of side reactions with other nucleophilic amino acid residues and the relative instability of the resulting bioconjugates in the blood stream. The latter drawback is especially important for the preparation of targeted therapies, such as antibody-drug conjugates.

Kristi Lynn Kiick is the Blue and Gold Distinguished Professor of Materials Science and Engineering at the University of Delaware. She studies polymers, biomaterials and hydrogels for drug delivery and regenerative medicine. She is a Fellow of the American Chemical Society, the American Institute for Medical and Biological Engineering, and of the National Academy of Inventors. She served for nearly eight years as the deputy dean of the college of engineering at the University of Delaware.

Polymer-protein hybrids are a class of nanostructure composed of protein-polymer conjugates. The protein component generally gives the advantages of biocompatibility and biodegradability, as many proteins are produced naturally by the body and are therefore well tolerated and metabolized. Although proteins are used as targeted therapy drugs, the main limitations—the lack of stability and insufficient circulation times still remain. Therefore, protein-polymer conjugates have been investigated to further enhance pharmacologic behavior and stability. By adjusting the chemical structure of the protein-polymer conjugates, polymer-protein particles with unique structures and functions, such as stimulus responsiveness, enrichment in specific tissue types, and enzyme activity, can be synthesized. Polymer-protein particles have been the focus of much research recently because they possess potential uses including bioseparations, imaging, biosensing, gene and drug delivery.

Boraacenes are polycyclic aromatic hydrocarbons containing at least one boron atom. Structurally, they are related to acenes, linearly fused benzene rings. However, the boron atom is electron deficient and may act as a Lewis Acid when compared to carbon. This results in slightly less negative charge within the ring, smaller HOMO-LUMO gaps, as well as differences in redox chemistry when compared to their acene analogues. When incorporated into acenes, Boron maintains the planarity and aromaticity of carbon acenes, while adding an empty p-orbital, which can be utilized for the fine tuning of organic semiconductor band gaps. Due to this empty p orbital, however, it is also highly reactive when exposed to nucleophiles like water or normal atmosphere, as it will readily be attacked by oxygen, which must be addressed to maintain its stability.
Heteroatomic multiple bonding between group 13 and group 15 elements are of great interest in synthetic chemistry due to their isoelectronicity with C-C multiple bonds. Nevertheless, the difference of electronegativity between group 13 and 15 leads to different character of bondings comparing to C-C multiple bonds. Because of the ineffective overlap between p𝝅 orbitals and the inherent lewis acidity/basicity of group 13/15 elements, the synthesis of compounds containing such multiple bonds is challenging and subject to oligomerization. The most common example of compounds with 13/15 group multiple bonds are those with B=N units. The boron-nitrogen-hydride compounds are candidates for hydrogen storage. In contrast, multiple bonding between aluminium and nitrogen Al=N, Gallium and nitrogen (Ga=N), boron and phosphorus (B=P), or boron and arsenic (B=As) are less common.

An N-heterocyclic olefin (NHO) is a neutral heterocyclic compound with a highly polarized, electron-rich C=C olefin attached to a heterocycle made up of two nitrogen atoms. A derivative of N-heterocyclic carbenes (NHCs), NHO was first synthesized in 1961 by Horst Böhme and Fritz Soldan, but the term NHO was not used until 2011 by Eric Rivard and coworkers. Since its discovery, NHOs have been applied in organocatalysis, metal ligation, and polymerization.

















