
Amyl nitrite is a chemical compound with the formula C5H11ONO. A variety of isomers are known, but they all feature an amyl group attached to the nitrite functional group. The alkyl group is unreactive and the chemical and biological properties are mainly due to the nitrite group. Like other alkyl nitrites, amyl nitrite is bioactive in mammals, being a vasodilator, which is the basis of its use as a prescription medicine. As an inhalant, it also has a psychoactive effect, which has led to its recreational use, with its smell being described as that of old socks or dirty feet. It was first documented in 1844 and came into medical use in 1867.
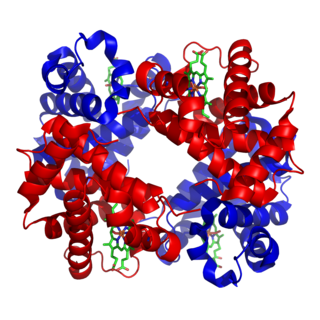
Hemoglobin is a protein containing iron that facilitates the transport of oxygen in red blood cells. Almost all vertebrates contain hemoglobin, with the exception of the fish family Channichthyidae. Hemoglobin in the blood carries oxygen from the respiratory organs to the other tissues of the body, where it releases the oxygen to enable aerobic respiration which powers the animal's metabolism. A healthy human has 12 to 20 grams of hemoglobin in every 100 mL of blood. Hemoglobin is a metalloprotein, a chromoprotein, and globulin.

Hemoglobinopathy is the medical term for a group of inherited blood disorders and diseases that primarily affect red blood cells. They are single-gene disorders and, in most cases, they are inherited as autosomal co-dominant traits.

Heme, or haem, is a ring-shaped iron-containing molecular component of hemoglobin, which is necessary to bind oxygen in the bloodstream. It is composed of four pyrrole rings with 2 vinyl and 2 propionic acid side chains. Heme is biosynthesized in both the bone marrow and the liver.

Methylthioninium chloride, commonly called methylene blue, is a salt used as a dye and as a medication. As a medication, it is mainly used to treat methemoglobinemia by chemically reducing the ferric iron in hemoglobin to ferrous iron. Specifically, it is used to treat methemoglobin levels that are greater than 30% or in which there are symptoms despite oxygen therapy. It has previously been used for treating cyanide poisoning and urinary tract infections, but this use is no longer recommended.

Methemoglobinemia, or methaemoglobinaemia, is a condition of elevated methemoglobin in the blood. Symptoms may include headache, dizziness, shortness of breath, nausea, poor muscle coordination, and blue-colored skin (cyanosis). Complications may include seizures and heart arrhythmias.
The nitrite ion has the chemical formula NO−
2. Nitrite is widely used throughout chemical and pharmaceutical industries. The nitrite anion is a pervasive intermediate in the nitrogen cycle in nature. The name nitrite also refers to organic compounds having the –ONO group, which are esters of nitrous acid.

Cyanosis is the change of body tissue color to a bluish-purple hue, as a result of decrease in the amount of oxygen bound to the hemoglobin in the red blood cells of the capillary bed. Cyanosis is apparent usually in the body tissues covered with thin skin, including the mucous membranes, lips, nail beds, and ear lobes. Some medications may cause discoloration such as medications containing amiodarone or silver. Furthermore, mongolian spots, large birthmarks, and the consumption of food products with blue or purple dyes can also result in the bluish skin tissue discoloration and may be mistaken for cyanosis. Appropriate physical examination and history taking is a crucial part to diagnose cyanosis. Management of cyanosis involves treating the main cause, as cyanosis isn’t a disease, it is a symptom.

Sodium nitrite is an inorganic compound with the chemical formula NaNO2. It is a white to slightly yellowish crystalline powder that is very soluble in water and is hygroscopic. From an industrial perspective, it is the most important nitrite salt. It is a precursor to a variety of organic compounds, such as pharmaceuticals, dyes, and pesticides, but it is probably best known as a food additive used in processed meats and (in some countries) in fish products.
Blue baby syndrome can refer to conditions that cause cyanosis, or blueness of the skin, in babies as a result of low oxygen levels in the blood. This term has traditionally been applied to cyanosis as a result of:.
- Cyanotic heart disease, which is a category of congenital heart defect that results in low levels of oxygen in the blood. This can be caused by either reduced blood flow to the lungs or mixing of oxygenated and deoxygenated blood.
- Methemoglobinemia, which is a disease defined by high levels of methemoglobin in the blood. Increased levels of methemoglobin prevent oxygen from being released into the tissues and result in hypoxemia.
Histotoxic hypoxia is the inability of cells to take up or use oxygen from the bloodstream, despite physiologically normal delivery of oxygen to such cells and tissues. Histotoxic hypoxia results from tissue poisoning, such as that caused by cyanide and certain other poisons like hydrogen sulfide.
The oxygen–hemoglobin dissociation curve, also called the oxyhemoglobin dissociation curve or oxygen dissociation curve (ODC), is a curve that plots the proportion of hemoglobin in its saturated (oxygen-laden) form on the vertical axis against the prevailing oxygen tension on the horizontal axis. This curve is an important tool for understanding how our blood carries and releases oxygen. Specifically, the oxyhemoglobin dissociation curve relates oxygen saturation (SO2) and partial pressure of oxygen in the blood (PO2), and is determined by what is called "hemoglobin affinity for oxygen"; that is, how readily hemoglobin acquires and releases oxygen molecules into the fluid that surrounds it.

Cytochromes b5 are ubiquitous electron transport hemoproteins found in animals, plants, fungi and purple phototrophic bacteria. The microsomal and mitochondrial variants are membrane-bound, while bacterial and those from erythrocytes and other animal tissues are water-soluble. The family of cytochrome b5-like proteins includes hemoprotein domains covalently associated with other redox domains in flavocytochrome cytochrome b2, sulfite oxidase, plant and fungal nitrate reductases, and plant and fungal cytochrome b5/acyl lipid desaturase fusion proteins.

Nitrate reductases are molybdoenzymes that reduce nitrate to nitrite. This reaction is critical for the production of protein in most crop plants, as nitrate is the predominant source of nitrogen in fertilized soils.

Cytochrome-b5 reductase is a NADH-dependent enzyme that converts ferricytochrome from a Fe3+ form to a Fe2+ form. It contains FAD and catalyzes the reaction:

Iron is an important biological element. It is used in both the ubiquitous iron-sulfur proteins and in vertebrates it is used in hemoglobin which is essential for blood and oxygen transport.

Nitric oxide dioxygenase (EC 1.14.12.17) is an enzyme that catalyzes the conversion of nitric oxide (NO) to nitrate (NO−
3) . The net reaction for the reaction catalyzed by nitric oxide dioxygenase is shown below:

NADH-cytochrome b5 reductase 3 is an enzyme that in humans is encoded by the CYB5R3 gene.
The phytoglobin-nitric oxide cycle is a metabolic pathway induced in plants under hypoxic conditions which involves nitric oxide (NO) and phytoglobin (Pgb). It provides an alternative type of respiration to mitochondrial electron transport under the conditions of limited oxygen supply. Phytoglobin in hypoxic plants acts as part of a soluble terminal nitric oxide dioxygenase system, yielding nitrate ion from the reaction of oxygenated phytoglobin with NO. Class 1 phytoglobins are induced in plants under hypoxia, bind oxygen very tightly at nanomolar concentrations, and can effectively scavenge NO at oxygen levels far below the saturation of cytochrome c oxidase. In the course of the reaction, phytoglobin is oxidized to metphytoglobin which has to be reduced for continuous operation of the cycle. Nitrate is reduced to nitrite by nitrate reductase, while NO is mainly formed due to anaerobic reduction of nitrite which may take place in mitochondria by complex III and complex IV in the absence of oxygen, in the side reaction of nitrate reductase, or by electron transport proteins on the plasma membrane. The overall reaction sequence of the cycle consumes NADH and can contribute to the maintenance of ATP level in highly hypoxic conditions.

Hemoglobin M disease is a rare form of hemoglobinopathy, characterized by the presence of hemoglobin M (HbM) and elevated methemoglobin (metHb) level in blood. HbM is an altered form of hemoglobin (Hb) due to point mutation occurring in globin-encoding genes, mostly involving tyrosine substitution for proximal (F8) or distal (E7) histidine residues. HbM variants are inherited as autosomal dominant disorders and have altered oxygen affinity. The pathophysiology of hemoglobin M disease involves heme iron autoxidation promoted by heme pocket structural alteration.

















