Personal protective equipment (PPE) is protective clothing, helmets, goggles, or other garments or equipment designed to protect the wearer's body from injury or infection. The hazards addressed by protective equipment include physical, electrical, heat, chemical, biohazards, and airborne particulate matter. Protective equipment may be worn for job-related occupational safety and health purposes, as well as for sports and other recreational activities. Protective clothing is applied to traditional categories of clothing, and protective gear applies to items such as pads, guards, shields, or masks, and others. PPE suits can be similar in appearance to a cleanroom suit.

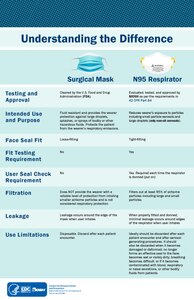
A surgical mask, also known by other names such as a medical face mask or procedure mask, is a personal protective equipment used by healthcare professionals that serves as a mechanical barrier that interferes with direct airflow in and out of respiratory orifices. This helps reduce airborne transmission of pathogens and other aerosolized contaminants between the wearer and nearby people via respiratory droplets ejected when sneezing, coughing, forceful expiration or unintentionally spitting when talking, etc. Surgical masks may be labeled as surgical, isolation, dental or medical procedure masks.

A respirator is a device designed to protect the wearer from inhaling hazardous atmospheres including fumes, vapours, gases and particulate matter such as dusts and airborne pathogens such as viruses. There are two main categories of respirators: the air-purifying respirator, in which respirable air is obtained by filtering a contaminated atmosphere, and the air-supplied respirator, in which an alternate supply of breathable air is delivered. Within each category, different techniques are employed to reduce or eliminate noxious airborne contaminants.

The term immediately dangerous to life or health (IDLH) is defined by the US National Institute for Occupational Safety and Health (NIOSH) as exposure to airborne contaminants that is "likely to cause death or immediate or delayed permanent adverse health effects or prevent escape from such an environment." Examples include smoke or other poisonous gases at sufficiently high concentrations. It is calculated using the LD50 or LC50. The Occupational Safety and Health Administration (OSHA) regulation defines the term as "an atmosphere that poses an immediate threat to life, would cause irreversible adverse health effects, or would impair an individual's ability to escape from a dangerous atmosphere."

A dust mask is a flexible paper pad held over the nose and mouth by elastic or rubber straps for personal comfort against non-toxic nuisance dusts. They are not intended to provide protection from toxic airborne hazards. The European FFP1 mask, the lowest-grade mechanical filter respirator available in the jurisdiction, is also used as a dust mask.

A respirator fit test checks whether a respirator properly fits the face of someone who wears it. The fitting characteristic of a respirator is the ability of the mask to separate a worker's respiratory system from ambient air.

A respirator cartridge or canister is a type of filter that removes gases, volatile organic compounds (VOCs), and other vapors from air through adsorption, absorption, or chemisorption. It is one of two basic types of filters used by air-purifying respirators. The other is a mechanical filter, which removes only particulates. Hybrid filters combine the two.
The National Personal Protective Technology Laboratory (NPPTL) is a research center within the National Institute for Occupational Safety and Health located in Pittsburgh, Pennsylvania, devoted to research on personal protective equipment (PPE). The NPPTL was created in 2001 at the request of the U.S. Congress, in response to a recognized need for improved research in PPE and technologies. It focuses on experimentation and recommendations for respirator masks, by ensuring a level of standard filter efficiency, and develops criteria for testing and developing PPE.

The NIOSH air filtration rating is the U.S. National Institute for Occupational Safety and Health (NIOSH)'s classification of filtering respirators. The ratings describe the ability of the device to protect the wearer from solid and liquid particulates in the air. The certification and approval process for respiratory protective devices is governed by Part 84 of Title 42 of the Code of Federal Regulations. Respiratory protective devices so classified include air-purifying respirators (APR) such as filtering facepiece respirators and chemical protective cartridges that have incorporated particulate filter elements.

A powered air-purifying respirator (PAPR) is a type of respirator used to safeguard workers against contaminated air. PAPRs consist of a headgear-and-fan assembly that takes ambient air contaminated with one or more type of pollutant or pathogen, actively removes (filters) a sufficient proportion of these hazards, and then delivers the clean air to the user's face or mouth and nose. They have a higher assigned protection factor than filtering facepiece respirators such as N95 masks. PAPRs are sometimes called positive-pressure masks, blower units, or just blowers.

The respiratory protective devices (RPD) can protect workers only if their protective properties are adequate to the conditions in the workplace. Therefore, specialists have developed criteria for the selection of proper, adequate respirators, including the Assigned Protection Factors (APF) - the decrease of the concentration of harmful substances in the inhaled air, which to be provided with timely and proper use of a certified respirator of certain types (design) by taught and trained workers, when the employer performs an effective respiratory protective device programme.

Respirators, also known as respiratory protective equipment (RPE) or respiratory protective devices (RPD), are used in some workplaces to protect workers from air contaminants. Initially, respirator effectiveness was tested in laboratories, but in the late 1960s it was found that these tests gave misleading results regarding the level of protection provided. In the 1970s, workplace-based respirator testing became routine in industrialized countries, leading to a dramatic reduction in the claimed efficacy of many respirator types and new guidelines on how to select the appropriate respirator for a given environment.

Hazard controls for COVID-19 in workplaces are the application of occupational safety and health methodologies for hazard controls to the prevention of COVID-19. Vaccination is the most effective way to protect against severe illness or death from COVID-19. Multiple layers of controls are recommended, including measures such as remote work and flextime, increased ventilation, personal protective equipment (PPE) and face coverings, social distancing, and enhanced cleaning programs.

A cloth face mask is a mask made of common textiles, usually cotton, worn over the mouth and nose. When more effective masks are not available, and when physical distancing is impossible, cloth face masks are recommended by public health agencies for disease "source control" in epidemic situations to protect others from virus laden droplets in infected mask wearers' breath, coughs, and sneezes. Because they are less effective than N95 masks, surgical masks, or physical distancing in protecting the wearer against viruses, they are not considered to be personal protective equipment by public health agencies. They are used by the general public in household and community settings as protection against both infectious diseases and particulate air pollution.

Mechanical filters are a class of filter for air-purifying respirators that mechanically stops particulates from reaching the wearer's nose and mouth. They come in multiple physical forms.

During the COVID-19 pandemic, face masks or coverings, including N95, FFP2, surgical, and cloth masks, have been employed as public and personal health control measures against the spread of SARS-CoV-2, the virus that causes COVID-19.

Source control is a strategy for reducing disease transmission by blocking respiratory secretions produced through speaking, coughing, sneezing or singing. Surgical masks are commonly used for this purpose, with cloth face masks recommended for use by the public only in epidemic situations when there are shortages of surgical masks. In addition, respiratory etiquette such as covering the mouth and nose with a tissue when coughing can be considered source control. In diseases transmitted by droplets or aerosols, understanding air flow, particle and aerosol transport may lead to rational infrastructural source control measures that minimize exposure of susceptible persons.

Elastomeric respirators, also called reusable air-purifying respirators, seal to the face with elastomeric material, which may be a natural or synthetic rubber. They are generally reusable. Full-face versions of elastomeric respirators seal better and protect the eyes.
FFP standards refer to the filtering half mask classification by EN 149, a European standard of testing and marking requirements for filtering half masks. FFP standard masks cover the nose, mouth and chin and may have inhalation and/or exhalation valves.

A supplied-air respirator (SAR) or air-line respirator is a breathing apparatus used in places where the ambient air may not be safe to breathe. It uses an air hose to supply air from outside the danger zone. It is similar to a self-contained breathing apparatus (SCBA), except that SCBA users carry their air with them in high pressure cylinders, while SAR users get it from a remote stationary air supply connected to them by a hose.