
Rhenium is a chemical element with the symbol Re and atomic number 75. It is a silvery-gray, heavy, third-row transition metal in group 7 of the periodic table. With an estimated average concentration of 1 part per billion (ppb), rhenium is one of the rarest elements in the Earth's crust. It has the third-highest melting point and second-highest boiling point of any element at 5869 K. It resembles manganese and technetium chemically and is mainly obtained as a by-product of the extraction and refinement of molybdenum and copper ores. It shows in its compounds a wide variety of oxidation states ranging from −1 to +7.

Group 7, numbered by IUPAC nomenclature, is a group of elements in the periodic table. It contains manganese (Mn), technetium (Tc), rhenium (Re) and bohrium (Bh). This group lies in the d-block of the periodic table, and are hence transition metals. This group is sometimes called the manganese group or manganese family after its lightest member; however, the group itself has not acquired a trivial name because it belongs to the broader grouping of the transition metals.

In organic chemistry, a sulfoxide, also called a sulfoxide, is an organosulfur compound containing a sulfinyl functional group attached to two carbon atoms. It is a polar functional group. Sulfoxides are oxidized derivatives of sulfides. Examples of important sulfoxides are alliin, a precursor to the compound that gives freshly crushed garlic its aroma, and dimethyl sulfoxide (DMSO), a common solvent.

Technetium(VII) oxide is the chemical compound with the formula Tc2O7. This yellow volatile solid is a rare example of a molecular binary metal oxide, the other examples being RuO4, OsO4, and the unstable Mn2O7. It adopts a centrosymmetric corner-shared bi-tetrahedral structure in which the terminal and bridging Tc−O bonds are 167pm and 184 pm respectively and the Tc−O−Tc angle is 180°.
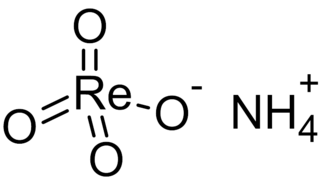
Ammonium perrhenate (APR) is the ammonium salt of perrhenic acid, NH4ReO4. It is the most common form in which rhenium is traded. It is a white salt; soluble in ethanol and water, and mildly soluble in NH4Cl. It was first described soon after the discovery of rhenium.

Manganese(VII) oxide (manganese heptoxide) is an inorganic compound with the formula Mn2O7. This volatile liquid is highly reactive. It is a dangerous oxidizer and was first described in 1860. It is the acid anhydride of permanganic acid.
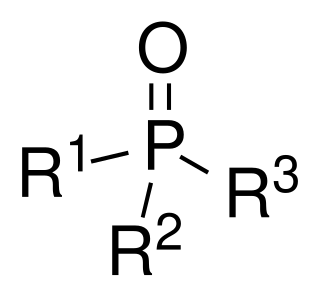
Phosphine oxides are phosphorus compounds with the formula OPX3. When X = alkyl or aryl, these are organophosphine oxides. Triphenylphosphine oxide is an example. An inorganic phosphine oxide is phosphoryl chloride (POCl3).

Rhenium(VII) oxide is the inorganic compound with the formula Re2O7. This yellowish solid is the anhydride of HOReO3. Perrhenic acid, Re2O7·2H2O, is closely related to Re2O7. Re2O7 is the raw material for all rhenium compounds, being the volatile fraction obtained upon roasting the host ore.
Rhenium trioxide or rhenium(VI) oxide is an inorganic compound with the formula ReO3. It is a red solid with a metallic lustre that resembles copper in appearance. It is the only stable trioxide of the Group 7 elements (Mn, Tc, Re).

Sodium perrhenate (also known as sodium rhenate(VII)) is the inorganic compound with the formula NaReO4. It is a white salt that is soluble in water. It is a common precursor to other rhenium compounds. Its structure resembles that of sodium perchlorate and sodium permanganate.

Bromopentacarbonylrhenium(I) is an inorganic compound of rhenium, commonly used for the syntheses of other rhenium complexes.

Dirhenium decacarbonyl is the inorganic compound with the chemical formula Re2(CO)10. Commercially available, it is used as a starting point for the synthesis of many rhenium carbonyl complexes. It was first reported in 1941 by Walter Hieber, who prepared it by reductive carbonylation of rhenium. The compound consists of a pair of square pyramidal Re(CO)5 units joined via a Re-Re bond, which produces a homoleptic carbonyl complex.
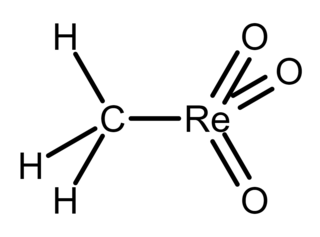
Methylrhenium trioxide, also known as methyltrioxorhenium(VII), is an organometallic compound with the formula CH3−ReO3. It is a volatile, colourless solid that has been used as a catalyst in some laboratory experiments. In this compound, rhenium has a tetrahedral coordination geometry with one methyl and three oxo ligands. The oxidation state of rhenium is +7.

Oxotrichlorobis(triphenylphosphine)rhenium(V) is the chemical compound with the formula ReOCl3(PPh3)2. This yellow, air-stable solid is a precursor to a variety of other rhenium complexes. In this diamagnetic compound, Re has an octahedral coordination environment with one oxo, three chloro and two mutually trans triphenylphosphine ligands. The oxidation state of rhenium is +5 and its configuration is d2.

Rhenium(IV) oxide or rhenium dioxide is the inorganic compound with the formula ReO2. This gray to black crystalline solid is a laboratory reagent that can be used as a catalyst. It adopts the rutile structure.
The perrhenate ion is the anion with the formula ReO−
4, or a compound containing this ion. The perrhenate anion is tetrahedral, being similar in size and shape to perchlorate and the valence isoelectronic permanganate. The perrhenate anion is stable over a broad pH range and can be precipitated from solutions with the use of organic cations. At normal pH, perrhenate exists as metaperrhenate, but at high pH mesoperrhenate forms. Perrhenate, like its conjugate acid perrhenic acid, features rhenium in the oxidation state of +7 with a d0 configuration. Solid perrhenate salts takes on the color of the cation.
Organorhenium chemistry describes the compounds with Re−C bonds. Because rhenium is a rare element, relatively few applications exist, but the area has been a rich source of concepts and a few useful catalysts.
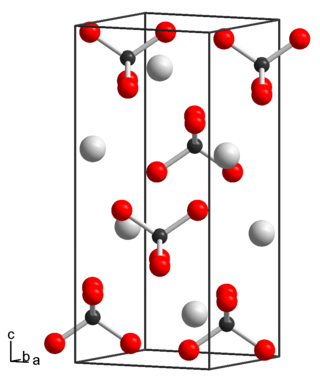
Potassium perrhenate is an inorganic compound with the chemical formula KReO4.
Rhenium compounds are compounds formed by the transition metal rhenium (Re). Rhenium can form in many oxidation states, and compounds are known for every oxidation state from -3 to +7 except -2, although the oxidation states +7, +6, +4, and +2 are the most common. Rhenium is most available commercially as salts of perrhenate, including sodium and ammonium perrhenates. These are white, water-soluble compounds. Tetrathioperrhenate anion [ReS4]− is possible.

Rhenium trioxide chloride is an inorganic compound with the formula ReO3Cl. It is a colorless, distillable, diamagnetic liquid. It is a rhenium oxychloride. The material is used as a reagent in the preparation of rhenium compounds.





















