
Prostamides are a class of physiologically active, lipid-like substances chemically related to prostaglandins. They occur naturally in humans and other animals. The first prostamide, called prostamide E2, was discovered in 1997. [1]

Prostamides are a class of physiologically active, lipid-like substances chemically related to prostaglandins. They occur naturally in humans and other animals. The first prostamide, called prostamide E2, was discovered in 1997. [1]

Prostamides are synthesized in the body from anandamide, which is the ethanolamide of arachidonic acid (ARA). This pathway is parallel to the synthesis of prostaglandins from ARA itself. The enzymes involved are at least partly the same as those responsible for prostaglandin synthesis. [1] [2]
It is thought that prostamides act via a specific receptor (or several receptors) which is distinct from the prostaglandin receptors. [1] No such receptor has been identified as of 2015 [update] . [2]
The names of prostamides are derived from the corresponding prostaglandin. For example, prostamide E2 is the ethanolamide of prostaglandin E2 (dinoprostone). [3]
The anti-glaucoma drug bimatoprost is a derivative of prostamide F2α, and is thought to have the same mechanism of action. [4] [5]
Importantly, prostaglandin F2α ethanolamide inhibits preadipocyte differentiation and increases their proliferation. [6] This mechanism maintains a reserve of adipocyte progenitor cells. This might allow for healthier development of fat tissue, which is through hyperplasia that outbalances morbid hypertrophy and ectopic fat deposition. Hyperplasia allows for storing excess energy triglycerides in more and smaller fat cells, instead of continually increasing the size of fat cells that leads to inflammation, ectopic fat deposits and fat tissue hypoxia. These findings have been behind the foundation of the Fat Four Ps Hypothesis, namely, Preadipocyte Pool Preservation by prostaglandin F2α ethanolamide. This mechanism could be tightly balanced as it can be controlled through the feedback control loop that involves this anti-adipogenic metabolite and its pro-adipogenic precursor anandamide (AEA). This has further suggested prostaglandin F2α ethanolamide synthetic pharmaceutical analog Bimatoprost as a promising therapy for obesity.

Prostaglandins (PG) are a group of physiologically active lipid compounds called eicosanoids that have diverse hormone-like effects in animals. Prostaglandins have been found in almost every tissue in humans and other animals. They are derived enzymatically from the fatty acid arachidonic acid. Every prostaglandin contains 20 carbon atoms, including a 5-carbon ring. They are a subclass of eicosanoids and of the prostanoid class of fatty acid derivatives.
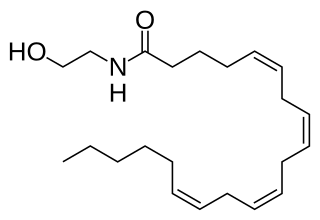
Anandamide (ANA), also known as N-arachidonoylethanolamine (AEA), an N-acylethanolamine (NAE), is a fatty acid neurotransmitter. Anandamide was the first endocannabinoid to be discovered: it participates in the body's endocannabinoid system by binding to cannabinoid receptors, the same receptors that the psychoactive compound THC in cannabis acts on. Anandamide is found in nearly all tissues in a wide range of animals. Anandamide has also been found in plants, including small amounts in chocolate. The name 'anandamide' is taken from the Sanskrit word ananda, which means "joy, bliss, delight", plus amide.

Eicosanoids are signaling molecules made by the enzymatic or non-enzymatic oxidation of arachidonic acid or other polyunsaturated fatty acids (PUFAs) that are, similar to arachidonic acid, around 20 carbon units in length. Eicosanoids are a sub-category of oxylipins, i.e. oxidized fatty acids of diverse carbon units in length, and are distinguished from other oxylipins by their overwhelming importance as cell signaling molecules. Eicosanoids function in diverse physiological systems and pathological processes such as: mounting or inhibiting inflammation, allergy, fever and other immune responses; regulating the abortion of pregnancy and normal childbirth; contributing to the perception of pain; regulating cell growth; controlling blood pressure; and modulating the regional flow of blood to tissues. In performing these roles, eicosanoids most often act as autocrine signaling agents to impact their cells of origin or as paracrine signaling agents to impact cells in the proximity of their cells of origin. Some eicosanoids, such as prostaglandins, may also have endocrine roles as hormones to influence the function of distant cells.
Linoleic acid (LA) is an organic compound with the formula HOOC(CH2)7CH=CHCH2CH=CH(CH2)4CH3. Both alkene groups are cis. It is a fatty acid sometimes denoted 18:2 (n-6) or 18:2 cis-9,12. A linoleate is a salt or ester of this acid.

Prostacyclin (also called prostaglandin I2 or PGI2) is a prostaglandin member of the eicosanoid family of lipid molecules. It inhibits platelet activation and is also an effective vasodilator.

The endocannabinoid system (ECS) is a biological system composed of endocannabinoids, which are endogenous lipid-based retrograde neurotransmitters that bind to cannabinoid receptors, and cannabinoid receptor proteins that are expressed throughout the vertebrate central nervous system and peripheral nervous system. The endocannabinoid system remains under preliminary research, but may be involved in regulating physiological and cognitive processes, including fertility, pregnancy, pre- and postnatal development, various activity of immune system, appetite, pain-sensation, mood, and memory, and in mediating the pharmacological effects of cannabis. The ECS plays an important role in multiple aspects of neural functions, including the control of movement and motor coordination, learning and memory, emotion and motivation, addictive-like behavior and pain modulation, among others.

Lipid signaling, broadly defined, refers to any biological cell signaling event involving a lipid messenger that binds a protein target, such as a receptor, kinase or phosphatase, which in turn mediate the effects of these lipids on specific cellular responses. Lipid signaling is thought to be qualitatively different from other classical signaling paradigms because lipids can freely diffuse through membranes. One consequence of this is that lipid messengers cannot be stored in vesicles prior to release and so are often biosynthesized "on demand" at their intended site of action. As such, many lipid signaling molecules cannot circulate freely in solution but, rather, exist bound to special carrier proteins in serum.

Bimatoprost, sold under the brand name Lumigan among others, is a medication used to treat high pressure inside the eye including glaucoma. Specifically it is used for open angle glaucoma when other agents are not sufficient. It may also be used to increase the size of the eyelashes. It is used as an eye drop and effects generally occur within four hours.

The transient receptor potential cation channel subfamily V member 1 (TRPV1), also known as the capsaicin receptor and the vanilloid receptor 1, is a protein that, in humans, is encoded by the TRPV1 gene. It was the first isolated member of the transient receptor potential vanilloid receptor proteins that in turn are a sub-family of the transient receptor potential protein group. This protein is a member of the TRPV group of transient receptor potential family of ion channels. Fatty acid metabolites with affinity for this receptor are produced by cyanobacteria, which diverged from eukaryotes at least 2000 million years ago (MYA). The function of TRPV1 is detection and regulation of body temperature. In addition, TRPV1 provides a sensation of scalding heat and pain (nociception). In primary afferent sensory neurons, it cooperates with TRPA1 to mediate the detection of noxious environmental stimuli.

AM404, also known as N-arachidonoylphenolamine, is an active metabolite of paracetamol (acetaminophen), responsible for all or part of its analgesic action and anticonvulsant effects. Chemically, it is the amide formed from 4-aminophenol and arachidonic acid.

N-Arachidonyl glycine receptor, also known as G protein-coupled receptor 18 (GPR18), is a protein that in humans is encoded by the GPR18 gene. Along with the other previously "orphan" receptors GPR55 and GPR119, GPR18 has been found to be a receptor for endogenous lipid neurotransmitters, several of which also bind to cannabinoid receptors. It has been found to be involved in the regulation of intraocular pressure.

Prostaglandin EP3 receptor (53kDa), also known as EP3, is a prostaglandin receptor for prostaglandin E2 (PGE2) encoded by the human gene PTGER3; it is one of four identified EP receptors, the others being EP1, EP2, and EP4, all of which bind with and mediate cellular responses to PGE2 and also, but generally with lesser affinity and responsiveness, certain other prostanoids (see Prostaglandin receptors). EP has been implicated in various physiological and pathological responses.

Prostaglandin F receptor (FP) is a receptor belonging to the prostaglandin (PG) group of receptors. FP binds to and mediates the biological actions of Prostaglandin F2α (PGF2α). It is encoded in humans by the PTGFR gene.

Prostaglandin F2α, pharmaceutically termed dinoprost is a naturally occurring prostaglandin used in medicine to induce labor and as an abortifacient. Prostaglandins are lipids throughout the entire body that have a hormone-like function. In pregnancy, PGF2 is medically used to sustain contracture and provoke myometrial ischemia to accelerate labor and prevent significant blood loss in labor. Additionally, PGF2 has been linked to being naturally involved in the process of labor. It has been seen that there are higher levels of PGF2 in maternal fluid during labor when compared to at term. This signifies that there is likely a biological use and significance to the production and secretion of PGF2 in labor. Prostaglandin is also used to treat uterine infections in domestic animals.

An N-acylethanolamine (NAE) is a type of fatty acid amide where one of several types of acyl groups is linked to the nitrogen atom of ethanolamine, and highly metabolic formed by intake of essential fatty acids through diet by 20:4, n-6 and 22:6, n-3 fatty acids, and when the body is physically and psychologically active,. The endocannabinoid signaling system (ECS) is the major pathway by which NAEs exerts its physiological effects in animal cells with similarities in plants, and the metabolism of NAEs is an integral part of the ECS, a very ancient signaling system, being clearly present from the divergence of the protostomian/deuterostomian, and even further back in time, to the very beginning of bacteria, the oldest organisms on Earth known to express phosphatidylethanolamine, the precursor to endocannabinoids, in their cytoplasmic membranes. Fatty acid metabolites with affinity for CB receptors are produced by cyanobacteria, which diverged from eukaryotes at least 2000 million years ago (MYA), by brown algae which diverged about 1500 MYA, by sponges, which diverged from eumetazoans about 930 MYA, and a lineages that predate the evolution of CB receptors, as CB1 – CB2 duplication event may have occurred prior to the lophotrochozoan-deuterostome divergence 590 MYA. Fatty acid amide hydrolase (FAAH) evolved relatively recently, either after the evolution of fish 400 MYA, or after the appearance of mammals 300 MYA, but after the appearance of vertebrates. Linking FAAH, vanilloid receptors (VR1) and anandamide implies a coupling among the remaining ‘‘older’’ parts of the endocannabinoid system, monoglyceride lipase (MGL), CB receptors, that evolved prior to the metazoan–bilaterian divergence, but were secondarily lost in the Ecdysozoa, and 2-Arachidonoylglycerol (2-AG).

Lithocholic acid, also known as 3α-hydroxy-5β-cholan-24-oic acid or LCA, is a bile acid that acts as a detergent to solubilize fats for absorption. Bacterial action in the colon produces LCA from chenodeoxycholic acid by reduction of the hydroxyl functional group at carbon-7 in the "B" ring of the steroid framework.

Adipogenesis is the formation of adipocytes from stem cells. It involves 2 phases, determination, and terminal differentiation. Determination is mesenchymal stem cells committing to the adipocyte precursor cells, also known as lipoblasts or preadipocytes which lose the potential to differentiate to other types of cells such as chondrocytes, myocytes, and osteoblasts. Terminal differentiation is that preadipocytes differentiate into mature adipocytes. Adipocytes can arise either from preadipocytes resident in adipose tissue, or from bone-marrow derived progenitor cells that migrate to adipose tissue.

N-Arachidonylglycine (NAGly) is a carboxylic metabolite of the endocannabinoid anandamide (AEA). Since it was first synthesized in 1996, NAGly has been a primary focus of the relatively contemporary field of lipidomics due to its wide range of signaling targets in the brain, the immune system and throughout various other bodily systems. In combination with 2‐arachidonoyl glycerol (2‐AG), NAGly has enabled the identification of a family of lipids often referred to as endocannabinoids. Recently, NAGly has been found to bind to G-protein coupled receptor 18 (GPR18), the putative abnormal cannabidiol receptor. NaGly is an endogenous inhibitor of fatty acid amide hydrolase (FAAH) and thereby increases the ethanolamide endocannabinoids AEA, oleoylethanolamide (OEA) and palmitoylethanolamide (PEA) levels. NaGly is found throughout the body and research on its explicit functions is ongoing.
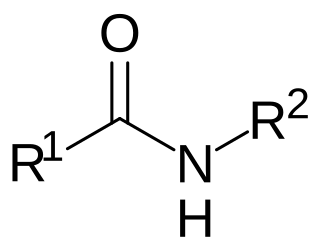
N-acyl amides are a general class of endogenous fatty acid compounds characterized by a fatty acyl group linked to a primary amine metabolite by an amide bond. Broadly speaking, N-acyl amides fall into several categories: amino acid conjugates, neurotransmitter conjugates, ethanolamine conjugates, and taurine conjugates. N-acyl amides have pleiotropic signaling functions in physiology, including in cardiovascular function, metabolic homeostasis, memory, cognition, pain, motor control and others. Initial attention focused on N-acyl amides present in mammalian organisms, however recently lipid signaling systems consisting of N-acyl amides have also been found to be present in invertebrates, such as Drosophila melanogaster. N-acyl amides play important roles in many biochemical pathways involved in a variety of physiological and pathological processes, as well as the metabolic enzymes, transporters, and receptors that regulate their signaling.
Specialized pro-resolving mediators are a large and growing class of cell signaling molecules formed in cells by the metabolism of polyunsaturated fatty acids (PUFA) by one or a combination of lipoxygenase, cyclooxygenase, and cytochrome P450 monooxygenase enzymes. Pre-clinical studies, primarily in animal models and human tissues, implicate SPM in orchestrating the resolution of inflammation. Prominent members include the resolvins and protectins.