
Paint is a liquid pigment that, after applied to a solid material and allowed to dry, adds a film-like layer, in most cases to create an image, known as a painting. Paint can be made in many colors and types. Most paints are either oil-based or water-based, and each has distinct characteristics.

Surfactants are chemical compounds that decrease the surface tension or interfacial tension between two liquids, a liquid and a gas, or a liquid and a solid. Surfactants may function as emulsifiers, wetting agents, detergents, foaming agents, or dispersants. The word "surfactant" is a blend of surface-active agent, coined c. 1950.
In polymer chemistry, emulsion polymerization is a type of radical polymerization that usually starts with an emulsion incorporating water, monomers, and surfactants. The most common type of emulsion polymerization is an oil-in-water emulsion, in which droplets of monomer are emulsified in a continuous phase of water. Water-soluble polymers, such as certain polyvinyl alcohols or hydroxyethyl celluloses, can also be used to act as emulsifiers/stabilizers. The name "emulsion polymerization" is a misnomer that arises from a historical misconception. Rather than occurring in emulsion droplets, polymerization takes place in the latex/colloid particles that form spontaneously in the first few minutes of the process. These latex particles are typically 100 nm in size, and are made of many individual polymer chains. The particles are prevented from coagulating with each other because each particle is surrounded by the surfactant ('soap'); the charge on the surfactant repels other particles electrostatically. When water-soluble polymers are used as stabilizers instead of soap, the repulsion between particles arises because these water-soluble polymers form a 'hairy layer' around a particle that repels other particles, because pushing particles together would involve compressing these chains.
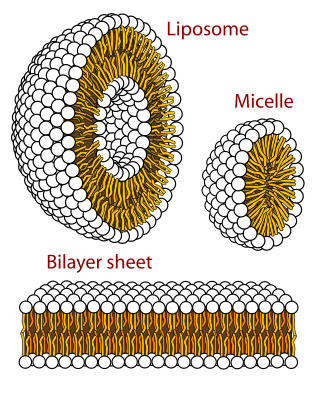
A micelle or micella is an aggregate of surfactant amphipathic lipid molecules dispersed in a liquid, forming a colloidal suspension. A typical micelle in water forms an aggregate with the hydrophilic "head" regions in contact with surrounding solvent, sequestering the hydrophobic single-tail regions in the micelle centre.
Lipophilicity, refers to the ability of a chemical compound to dissolve in fats, oils, lipids, and non-polar solvents such as hexane or toluene. Such non-polar solvents are themselves lipophilic, and the axiom that "like dissolves like" generally holds true. Thus lipophilic substances tend to dissolve in other lipophilic substances, but hydrophilic ("water-loving") substances tend to dissolve in water and other hydrophilic substances.
Microemulsions are clear, thermodynamically stable isotropic liquid mixtures of oil, water and surfactant, frequently in combination with a cosurfactant. The aqueous phase may contain salt(s) and/or other ingredients, and the "oil" may actually be a complex mixture of different hydrocarbons. In contrast to ordinary emulsions, microemulsions form upon simple mixing of the components and do not require the high shear conditions generally used in the formation of ordinary emulsions. The three basic types of microemulsions are direct, reversed and bicontinuous.
An antistatic agent is a compound used for treatment of materials or their surfaces in order to reduce or eliminate buildup of static electricity. Static charge may be generated by the triboelectric effect or by a non-contact process using a high voltage power source. Static charge may be introduced on a surface as part of an in-mold label printing process.

An amphiphile, or amphipath, is a chemical compound possessing both hydrophilic and lipophilic (fat-loving) properties. Such a compound is called amphiphilic or amphipathic. Amphiphilic compounds include surfactants. The phospholipid amphiphiles are the major structural component of cell membranes.
Solution polymerization is a method of industrial polymerization. In this procedure, a monomer is dissolved in a non-reactive solvent that contains a catalyst or initiator.
An anti-graffiti coating is a coating that prevents graffiti paint from bonding to surfaces.
Adsorption of polyelectrolytes on solid substrates is a surface phenomenon where long-chained polymer molecules with charged groups bind to a surface that is charged in the opposite polarity. On the molecular level, the polymers do not actually bond to the surface, but tend to "stick" to the surface via intermolecular forces and the charges created by the dissociation of various side groups of the polymer. Because the polymer molecules are so long, they have a large amount of surface area with which to contact the surface and thus do not desorb as small molecules are likely to do. This means that adsorbed layers of polyelectrolytes form a very durable coating. Due to this important characteristic of polyelectrolyte layers they are used extensively in industry as flocculants, for solubilization, as supersorbers, antistatic agents, as oil recovery aids, as gelling aids in nutrition, additives in concrete, or for blood compatibility enhancement to name a few.

An oil dispersant is a mixture of emulsifiers and solvents that helps break oil into small droplets following an oil spill. Small droplets are easier to disperse throughout a water volume, and small droplets may be more readily biodegraded by microbes in the water. Dispersant use involves a trade-off between exposing coastal life to surface oil and exposing aquatic life to dispersed oil. While submerging the oil with dispersant may lessen exposure to marine life on the surface, it increases exposure for animals dwelling underwater, who may be harmed by toxicity of both dispersed oil and dispersant. Although dispersant reduces the amount of oil that lands ashore, it may allow faster, deeper penetration of oil into coastal terrain, where it is not easily biodegraded.
Macroemulsions are dispersed liquid-liquid, thermodynamically unstable systems with particle sizes ranging from 1 to 100 μm, which, most often, do not form spontaneously. Macroemulsions scatter light effectively and therefore appear milky, because their droplets are greater than a wavelength of light. They are part of a larger family of emulsions along with miniemulsions. As with all emulsions, one phase serves as the dispersing agent. It is often called the continuous or outer phase. The remaining phase(s) are disperse or inner phase(s), because the liquid droplets are finely distributed amongst the larger continuous phase droplets. This type of emulsion is thermodynamically unstable, but can be stabilized for a period of time with applications of kinetic energy. Surfactants are used to reduce the interfacial tension between the two phases, and induce macroemulsion stability for a useful amount of time. Emulsions can be stabilized otherwise with polymers, solid particles or proteins.
Polyelectrolytes are charged polymers capable of stabilizing colloidal emulsions through electrostatic interactions. Their effectiveness can be dependent on molecular weight, pH, solvent polarity, ionic strength, and the hydrophilic-lipophilic balance (HLB). Stabilized emulsions are useful in many industrial processes, including deflocculation, drug delivery, petroleum waste treatment, and food technology.
In colloidal chemistry, the surfactant’s critical micelle concentration (CMC) plays a factor in Gibbs free energy of micellization. The exact concentration of the surfactants that yield the aggregates being thermodynamically soluble is the CMC. The Krafft temperature determines the solubility of the surfactants which in turn is the temperature that CMC is achieved. There are many parameters that affect the CMC. The interaction between the hydrophilic heads and the hydrophobic tails play a part, as well as the concentration of salt within the solution and surfactants.
The surface chemistry of paper is responsible for many important paper properties, such as gloss, waterproofing, and printability. Many components are used in the paper-making process that affect the surface.
Wax emulsions are stable mixtures of one or more waxes in water. Waxes and water are normally immiscible but can be brought together stably by the use of surfactants and a clever preparation process. Strictly speaking a wax emulsion should be called a wax dispersion since the wax is solid at room temperature. However, because the preparation takes place above the melting point of the wax, the actual process is called emulsification, hence the name wax emulsion. In praxis, wax dispersion is used for solvent based systems.
Polyurethane dispersion, or PUD, is understood to be a polyurethane polymer resin dispersed in water, rather than a solvent, although some cosolvent maybe used. Its manufacture involves the synthesis of polyurethanes having carboxylic acid functionality or nonionic hydrophiles like PEG incorporated into, or pendant from, the polymer backbone. Two component polyurethane dispersions are also available.
Waterborne resins are sometimes called water-based resins. They are resins or polymeric resins that use water as the carrying medium as opposed to solvent or solvent-less. Resins are used in the production of coatings, adhesives, sealants, elastomers and composite materials. When the phrase waterborne resin is used, it usually describes all resins which have water as the main carrying solvent. The resin could be water-soluble, water reducible or water dispersed.

Emmie Helena Lucassen-Reynders was a Dutch scientist specialising in colloid chemistry and theoretical physics. She worked in both academia and in industry.










