
Dermatitis is inflammation of the skin, typically characterized by itchiness, redness and a rash. In cases of short duration, there may be small blisters, while in long-term cases the skin may become thickened. The area of skin involved can vary from small to covering the entire body. Dermatitis is often called eczema, and the difference between those terms is not standardized.

Dandruff is a skin condition that mainly affects the scalp. Symptoms include flaking and sometimes mild itchiness. It can result in social or self-esteem problems. A more severe form of the condition, which includes inflammation of the skin, is known as seborrhoeic dermatitis.
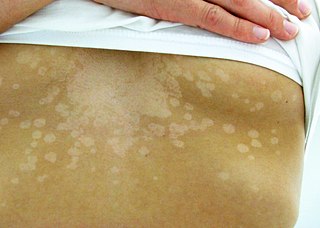
Tinea versicolor is a condition characterized by a skin eruption on the trunk and proximal extremities. The majority of tinea versicolor is caused by the fungus Malassezia globosa, although Malassezia furfur is responsible for a small number of cases. These yeasts are normally found on the human skin and become troublesome only under certain conditions, such as a warm and humid environment, although the exact conditions that cause initiation of the disease process are poorly understood.
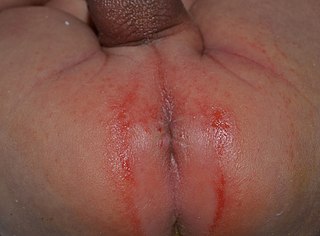
Irritant diaper dermatitis is a generic term applied to skin rash in the diaper area that are caused by various skin disorders and/or irritants.

Seborrhoeic dermatitis is a long-term skin disorder. Symptoms include flaky, scaly, greasy, and occasionally itchy and inflamed skin. Areas of the skin rich in oil-producing glands are often affected including the scalp, face, and chest. It can result in social or self-esteem problems. In babies, when the scalp is primarily involved, it is called cradle cap. Seborrhoeic dermatitis of the scalp may be described in lay terms as dandruff due to the dry, flaky character of the skin. However, as dandruff may refer to any dryness or scaling of the scalp, not all dandruff is seborrhoeic dermatitis. Seborrhoeic dermatitis is sometimes inaccurately referred to as seborrhoea.

An antifungal medication, also known as an antimycotic medication, is a pharmaceutical fungicide or fungistatic used to treat and prevent mycosis such as athlete's foot, ringworm, candidiasis (thrush), serious systemic infections such as cryptococcal meningitis, and others. Such drugs are usually obtained by a doctor's prescription, but a few are available over the counter (OTC). The evolution of antifungal resistance is a growing threat to health globally.

Ketoconazole, sold under the brand name Nizoral among others, is an antiandrogen, antifungal, and antiglucocorticoid medication used to treat a number of fungal infections. Applied to the skin it is used for fungal skin infections such as tinea, cutaneous candidiasis, pityriasis versicolor, dandruff, and seborrheic dermatitis. Taken by mouth it is a less preferred option and only recommended for severe infections when other agents cannot be used. Other uses include treatment of excessive male-patterned hair growth in women and Cushing's syndrome.
Keratolytic therapy is a type of medical treatment to remove warts, calluses and other lesions in which the epidermis produces excess skin. In this therapy, acidic topical medicines, such as Whitfield's ointment or Jessner's solution, are applied to the lesion in order to thin the skin on and around it. This therapy causes the outer layer of the skin to loosen and shed.

In chemistry, an ionophore is a chemical species that reversibly binds ions. Many ionophores are lipid-soluble entities that transport ions across the cell membrane. Ionophores catalyze ion transport across hydrophobic membranes, such as liquid polymeric membranes or lipid bilayers found in the living cells or synthetic vesicles (liposomes). Structurally, an ionophore contains a hydrophilic center and a hydrophobic portion that interacts with the membrane.
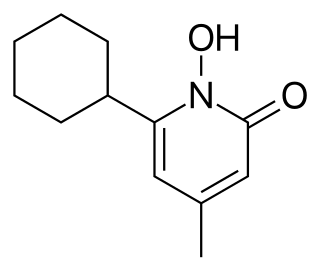
Ciclopirox is a synthetic antifungal agent for topical dermatologic treatment of superficial mycoses. It is most useful against tinea versicolor. It is often used clinically as ciclopirox olamine, the olamine salt of ciclopirox.
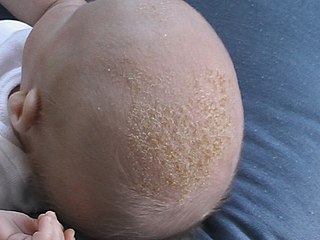
Cradle cap causes crusty or oily scaly patches on a baby's scalp. The condition is not painful or itchy, but it can cause thick white or yellow scales that are not easy to remove. Cradle cap most commonly begins sometime in the first three months but can occur in later years. Similar symptoms in older children are more likely to be dandruff than cradle cap. The rash is often prominent around the ear, the eyebrows or the eyelids. It may appear in other locations as well, where it is called infantile seborrhoeic dermatitis. Cradle cap is just a special—and more benign—case of this condition. The exact cause of cradle cap is not known. Cradle cap is not spread from person to person (contagious). It is also not caused by poor hygiene. It is not an allergy, and it is not dangerous. Cradle cap often lasts a few months. In some children, the condition can last until age 2 or 3.
Selenium disulfide, also known as selenium sulfide, is a chemical compound and medication used to treat seborrheic dermatitis, dandruff, and pityriasis versicolor. It is applied to the affected area as a lotion or shampoo. Symptoms frequently return if treatment is stopped.
Selsun Blue is an over-the-counter brand of dandruff shampoo now owned by Sanofi. First made by Abbott Laboratories, the brand was bought by Chattem in 2002. Chattem was acquired by Sanofi in 2010, although in some markets, it is sold by Rohto Pharmaceutical instead. Selsun Blue has been marketed as a more effective alternative to brand leader Head & Shoulders due to its superior performance in randomized trials.

Malassezia furfur is a species of yeast that is naturally found on the skin surfaces of humans and some other mammals. It is associated with a variety of dermatological conditions caused by fungal infections, notably seborrhoeic dermatitis and tinea versicolor. As an opportunistic pathogen, it has further been associated with dandruff, malassezia folliculitis, pityriasis versicolor (alba), and malassezia intertrigo, as well as catheter-related fungemia and pneumonia in patients receiving hematopoietic transplants.

Climbazole is a topical antifungal agent commonly used in the treatment of human fungal skin infections such as dandruff, seborrhoeic dermatitis and eczema. Climbazole has shown a high in vitro and in vivo efficacy against Malassezia spp. that appear to play an important role in the pathogenesis of dandruff. Its chemical structure and properties are similar to other azole fungicides such as ketoconazole, clotrimazole and miconazole.
Anti-seborrheics are drugs effective in seborrheic dermatitis. Selenium sulfide, zinc pyrithione, corticosteroids, imidazole antifungals, and salicylic acid are common anti-seborrheics.
Malassezia pachydermatis is a zoophilic yeast in the division Basidiomycota. It was first isolated in 1925 by Fred Weidman, and it was named pachydermatis after the original sample taken from an Indian rhinoceros with severe exfoliative dermatitis. Within the genus Malassezia, M. pachydermatis is most closely related to the species M. furfur. A commensal fungus, it can be found within the microflora of healthy mammals such as humans, cats and dogs, However, it is capable of acting as an opportunistic pathogen under special circumstances and has been seen to cause skin and ear infections, most often occurring in canines.
Malassezia sympodialis is a species in the genus Malassezia. It is characterized by a pronounced lipophily, unilateral, percurrent or sympodial budding and an irregular, corrugated cell wall ultrastructure. It is one of the most common species found on the skin of healthy and diseased individuals. It is considered to be part of the skin's normal human microbiota and begins to colonize the skin of humans shortly after birth. Malassezia sympodialis, often has a symbiotic or commensal relationship with its host, but it can act as a pathogen causing a number of different skin diseases, such as atopic dermatitis.

Pyrithione is the common name of an organosulfur compound with molecular formula C
5H
5NOS, chosen as an abbreviation of pyridinethione, and found in the Persian shallot. It exists as a pair of tautomers, the major form being the thione 1-hydroxy-2(1H)-pyridinethione and the minor form being the thiol 2-mercaptopyridine N-oxide; it crystallises in the thione form. It is usually prepared from either 2-bromopyridine, 2-chloropyridine, or 2-chloropyridine N-oxide, and is commercially available as both the neutral compound and its sodium salt. It is used to prepare zinc pyrithione, which is used primarily to treat dandruff and seborrhoeic dermatitis in medicated shampoos, though is also an anti-fouling agent in paints.
Topical antifungaldrugs are used to treat fungal infections on the skin, scalp, nails, vagina or inside the mouth. These medications come as creams, gels, lotions, ointments, powders, shampoos, tinctures and sprays. Most antifungal drugs induce fungal cell death by destroying the cell wall of the fungus. These drugs inhibit the production of ergosterol, which is a fundamental component of the fungal cell membrane and wall.














