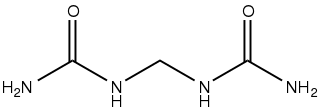
Urea, also called carbamide, is an organic compound with chemical formula CO(NH2)2. This amide has two amino groups joined by a carbonyl functional group. It is thus the simplest amide of carbamic acid.

A fertilizer or fertiliser is any material of natural or synthetic origin that is applied to soil or to plant tissues to supply plant nutrients. Fertilizers may be distinct from liming materials or other non-nutrient soil amendments. Many sources of fertilizer exist, both natural and industrially produced. For most modern agricultural practices, fertilization focuses on three main macro nutrients: nitrogen (N), phosphorus (P), and potassium (K) with occasional addition of supplements like rock flour for micronutrients. Farmers apply these fertilizers in a variety of ways: through dry or pelletized or liquid application processes, using large agricultural equipment or hand-tool methods.

The nitrogen cycle is the biogeochemical cycle by which nitrogen is converted into multiple chemical forms as it circulates among atmospheric, terrestrial, and marine ecosystems. The conversion of nitrogen can be carried out through both biological and physical processes. Important processes in the nitrogen cycle include fixation, ammonification, nitrification, and denitrification. The majority of Earth's atmosphere (78%) is atmospheric nitrogen, making it the largest source of nitrogen. However, atmospheric nitrogen has limited availability for biological use, leading to a scarcity of usable nitrogen in many types of ecosystems.
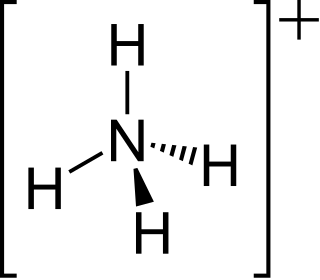
The ammonium cation is a positively charged polyatomic ion with the chemical formula NH+4 or [NH4]+. It is formed by the protonation of ammonia. Ammonium is also a general name for positively charged (protonated) substituted amines and quaternary ammonium cations, where one or more hydrogen atoms are replaced by organic or other groups. Not only is ammonium a source of nitrogen and a key metabolite for many living organisms, but it is an integral part of the global nitrogen cycle. As such, the human impact in recent years could have an effect on the biological communities that depend on it.
Ureases, functionally, belong to the superfamily of amidohydrolases and phosphotriesterases. Ureases are found in numerous bacteria, fungi, algae, plants, and some invertebrates, as well as in soils, as a soil enzyme. They are nickel-containing metalloenzymes of high molecular weight.
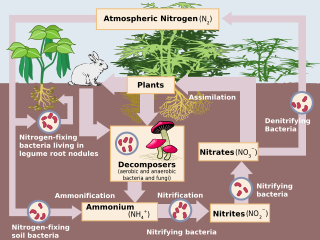
Nitrification is the biological oxidation of ammonia to nitrate via the intermediary nitrite. Nitrification is an important step in the nitrogen cycle in soil. The process of complete nitrification may occur through separate organisms or entirely within one organism, as in comammox bacteria. The transformation of ammonia to nitrite is usually the rate limiting step of nitrification. Nitrification is an aerobic process performed by small groups of autotrophic bacteria and archaea.

In organic chemistry, a carbamate is a category of organic compounds with the general formula R2NC(O)OR and structure >N−C(=O)−O−, which are formally derived from carbamic acid. The term includes organic compounds, formally obtained by replacing one or more of the hydrogen atoms by other organic functional groups; as well as salts with the carbamate anion H2NCOO−.

Nutrient management is the science and practice directed to link soil, crop, weather, and hydrologic factors with cultural, irrigation, and soil and water conservation practices to achieve optimal nutrient use efficiency, crop yields, crop quality, and economic returns, while reducing off-site transport of nutrients (fertilizer) that may impact the environment. It involves matching a specific field soil, climate, and crop management conditions to rate, source, timing, and place of nutrient application.

Nitrosomonas is a genus of Gram-negative bacteria, belonging to the Betaproteobacteria. It is one of the five genera of ammonia-oxidizing bacteria and, as an obligate chemolithoautotroph, uses ammonia as an energy source and carbon dioxide as a carbon source in presence of oxygen. Nitrosomonas are important in the global biogeochemical nitrogen cycle, since they increase the bioavailability of nitrogen to plants and in the denitrification, which is important for the release of nitrous oxide, a powerful greenhouse gas. This microbe is photophobic, and usually generate a biofilm matrix, or form clumps with other microbes, to avoid light. Nitrosomonas can be divided into six lineages: the first one includes the species Nitrosomonas europea, Nitrosomonas eutropha, Nitrosomonas halophila, and Nitrosomonas mobilis. The second lineage presents the species Nitrosomonas communis, N. sp. I and N. sp. II, meanwhile the third lineage includes only Nitrosomonas nitrosa. The fourth lineage includes the species Nitrosomonas ureae and Nitrosomonas oligotropha and the fifth and sixth lineages include the species Nitrosomonas marina, N. sp. III, Nitrosomonas estuarii and Nitrosomonas cryotolerans.
Selective catalytic reduction (SCR) means of converting nitrogen oxides, also referred to as NO
x with the aid of a catalyst into diatomic nitrogen, and water. A reductant, typically anhydrous ammonia, aqueous ammonia, or a urea solution, is added to a stream of flue or exhaust gas and is reacted onto a catalyst. As the reaction drives toward completion, nitrogen, and carbon dioxide, in the case of urea use, are produced.
Carbamic acid, which might also be called aminoformic acid or aminocarboxylic acid, is the chemical compound with the formula H2NCOOH. It can be obtained by the reaction of ammonia NH3 and carbon dioxide CO2 at very low temperatures, which also yields ammonium carbamate [NH4]+[NH2CO2]−. The compound is stable only up to about 250 K (−23 °C); at higher temperatures it decomposes into those two gases. The solid apparently consists of dimers, with the two molecules connected by hydrogen bonds between the two carboxyl groups –COOH.
Berthelot's reagent is an alkaline solution of phenol and hypochlorite, used in analytical chemistry. It is named after its inventor, Marcellin Berthelot. Ammonia reacts with Berthelot's reagent to form a blue product which is used in a colorimetric method for determining ammonia. The reagent can also be used for determining urea. In this case the enzyme urease is used to catalyze the hydrolysis of urea into carbon dioxide and ammonia. The ammonia is then determined with Berthelot's reagent.
A controlled-release fertiliser (CRF) is a granulated fertiliser that releases nutrients gradually into the soil. Controlled-release fertilizer is also known as controlled-availability fertilizer, delayed-release fertilizer, metered-release fertilizer, or slow-acting fertilizer. Usually CRF refers to nitrogen-based fertilizers. Slow- and controlled-release involve only 0.15% of the fertilizer market (1995).
Sporosarcina pasteurii formerly known as Bacillus pasteurii from older taxonomies, is a gram positive bacterium with the ability to precipitate calcite and solidify sand given a calcium source and urea; through the process of microbiologically induced calcite precipitation (MICP) or biological cementation. S. pasteurii has been proposed to be used as an ecologically sound biological construction material. Researchers studied the bacteria in conjunction with plastic and hard mineral; forming a material stronger than bone. It is a commonly used for MICP since it is non-pathogenic and is able to produce high amounts of the enzyme urease which hydrolyzes urea to carbonate and ammonia.
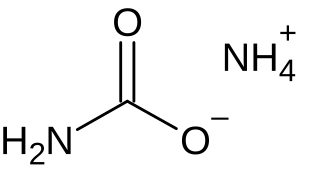
Ammonium carbamate is a chemical compound with the formula [NH4][H2NCO2] consisting of ammonium cation NH+4 and carbamate anion NH2COO−. It is a white solid that is extremely soluble in water, less so in alcohol. Ammonium carbamate can be formed by the reaction of ammonia NH3 with carbon dioxide CO2, and will slowly decompose to those gases at ordinary temperatures and pressures. It is an intermediate in the industrial synthesis of urea (NH2)2CO, an important fertilizer.
Sporosarcina ureae is a type of bacteria of the genus Sporosarcina, and is closely related to the genus Bacillus. S. ureae is an aerobic, motile, spore-forming, Gram-positive coccus, originally isolated in the early 20th century from soil. S. ureae is distinguished by its ability to grow in relatively high concentrations of urea through production of at least one exourease, an enzyme that converts urea to ammonia. S. ureae has also been found to sporulate when environmental conditions become unfavorable, and can remain viable for up to a year.

Nitrapyrin is an organic compound with the formula ClC5H3NCCl3, and is described as a white crystalline solid with a sweet odor. It is used as a nitrification inhibitor and bactericide, which is applied to soils for the growing of agricultural crops since 1974. Nitrapyrin was put up for review by the EPA and deemed safe for use in 2005. Nitrapyrin is an effective nitrification inhibitor to the bacteria Nitrosomonas and has been shown to drastically the reduce the amount of N2O emissions from the soil.
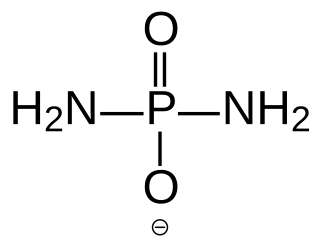
Diamidophosphate (DAP) is the simplest phosphorodiamidate ion, with formula PO2(NH2)2−. It is a phosphorylating ion and was first used for the phosphorylation of sugars in aqueous medium. DAP has attracted interest in the area of primordial chemistry.

N-(n-Butyl)thiophosphoric triamide (NBPT) is the organophosphorus compound with the formula SP(NH2)2(NHC4H9). It is an amide of thiophosphoric acid. A white solid, NBPT is an "enhanced efficiency fertilizer", intended to limit the release of nitrogen-containing gases following fertilization. Regarding its chemical structure, the molecule features tetrahedral phosphorus bonded to sulfur and three amido groups.
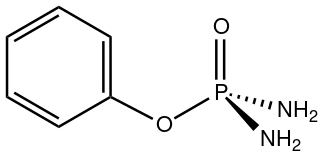
Phenyl phosphorodiamidate is an organophosphorus compound with the formula C6H5OP(O)(NH2)2. A white solid, it is used as an inhibitor of urease, an enzyme that accelerates the hydrolysis of urea. In this way, phenyl phosphorodiamidate enhances the effectiveness of urea-based fertilizers. It is a component of the technology of controlled release fertilizers.















