Related Research Articles

Cyclic voltammetry (CV) is a type of potentiodynamic electrochemical measurement. In a cyclic voltammetry experiment, the working electrode potential is ramped linearly versus time. Unlike in linear sweep voltammetry, after the set potential is reached in a CV experiment, the working electrode's potential is ramped in the opposite direction to return to the initial potential. These cycles of ramps in potential may be repeated as many times as needed. The current at the working electrode is plotted versus the applied voltage to give the cyclic voltammogram trace. Cyclic voltammetry is generally used to study the electrochemical properties of an analyte in solution or of a molecule that is adsorbed onto the electrode.

Glass-like carbon, often called glassy carbon or vitreous carbon, is a non-graphitizing, or nongraphitizable, carbon which combines glassy and ceramic properties with those of graphite. The most important properties are high temperature resistance, hardness (7 Mohs), low density, low electrical resistance, low friction, low thermal resistance, extreme resistance to chemical attack, and impermeability to gases and liquids. Glassy carbon is widely used as an electrode material in electrochemistry, for high-temperature crucibles, and as a component of some prosthetic devices. It can be fabricated in different shapes, sizes and sections.

Voltammetry is a category of electroanalytical methods used in analytical chemistry and various industrial processes. In voltammetry, information about an analyte is obtained by measuring the current as the potential is varied. The analytical data for a voltammetric experiment comes in the form of a voltammogram which plots the current produced by the analyte versus the potential of the working electrode.
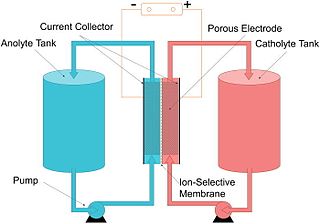
A flow battery, or redox flow battery, is a type of electrochemical cell where chemical energy is provided by two chemical components dissolved in liquids that are pumped through the system on separate sides of a membrane. Ion exchange occurs through the membrane while both liquids circulate in their own respective space. Cell voltage is chemically determined by the Nernst equation and ranges, in practical applications, from 1.0 to 2.43 volts.
A polymer-based battery uses organic materials instead of bulk metals to form a battery. Currently accepted metal-based batteries pose many challenges due to limited resources, negative environmental impact, and the approaching limit of progress. Redox active polymers are attractive options for electrodes in batteries due to their synthetic availability, high-capacity, flexibility, light weight, low cost, and low toxicity. Recent studies have explored how to increase efficiency and reduce challenges to push polymeric active materials further towards practicality in batteries. Many types of polymers are being explored, including conductive, non-conductive, and radical polymers. Batteries with a combination of electrodes are easier to test and compare to current metal-based batteries, however batteries with both a polymer cathode and anode are also a current research focus. Polymer-based batteries, including metal/polymer electrode combinations, should be distinguished from metal-polymer batteries, such as a lithium polymer battery, which most often involve a polymeric electrolyte, as opposed to polymeric active materials.
The electronic tongue is an instrument that measures and compares tastes. As per the IUPAC technical report, an “electronic tongue” as analytical instrument including an array of non-selective chemical sensors with partial specificity to different solution components and an appropriate pattern recognition instrument, capable to recognize quantitative and qualitative compositions of simple and complex solutions
Electroanalytical methods are a class of techniques in analytical chemistry which study an analyte by measuring the potential (volts) and/or current (amperes) in an electrochemical cell containing the analyte. These methods can be broken down into several categories depending on which aspects of the cell are controlled and which are measured. The three main categories are potentiometry, coulometry, and voltammetry.
The working electrode is the electrode in an electrochemical system on which the reaction of interest is occurring. The working electrode is often used in conjunction with an auxiliary electrode, and a reference electrode in a three electrode system. Depending on whether the reaction on the electrode is a reduction or an oxidation, the working electrode is called cathodic or anodic, respectively. Common working electrodes can consist of materials ranging from inert metals such as gold, silver or platinum, to inert carbon such as glassy carbon, boron doped diamond or pyrolytic carbon, and mercury drop and film electrodes. Chemically modified electrodes are employed for the analysis of both organic and inorganic samples.
Squarewave voltammetry (SWV) is a form of linear potential sweep voltammetry that uses a combined square wave and staircase potential applied to a stationary electrode. It has found numerous applications in various fields, including within medicinal and various sensing communities.

Electrochemiluminescence or electrogenerated chemiluminescence (ECL) is a kind of luminescence produced during electrochemical reactions in solutions. In electrogenerated chemiluminescence, electrochemically generated intermediates undergo a highly exergonic reaction to produce an electronically excited state that then emits light upon relaxation to a lower-level state. This wavelength of the emitted photon of light corresponds to the energy gap between these two states. ECL excitation can be caused by energetic electron transfer (redox) reactions of electrogenerated species. Such luminescence excitation is a form of chemiluminescence where one/all reactants are produced electrochemically on the electrodes.
Electrochemical gas sensors are gas detectors that measure the concentration of a target gas by oxidizing or reducing the target gas at an electrode and measuring the resulting current.

The Marchywka effect refers to electrochemical cleaning of diamond using an electric field induced with remote electrodes.
A chemically modified electrode is an electrical conductor that has its surface modified for different electrochemical functions. Chemically modified electrodes are made using advanced approaches to electrode systems by adding a thin film or layer of certain chemicals to change properties of the conductor according to its targeted function.

Smart inorganic polymers (SIPs) are hybrid or fully inorganic polymers with tunable (smart) properties such as stimuli responsive physical properties. While organic polymers are often petrol-based, the backbones of SIPs are made from elements other than carbon which can lessen the burden on scarce non-renewable resources and provide more sustainable alternatives. Common backbones utilized in SIPs include polysiloxanes, polyphosphates, and polyphosphazenes, to name a few.
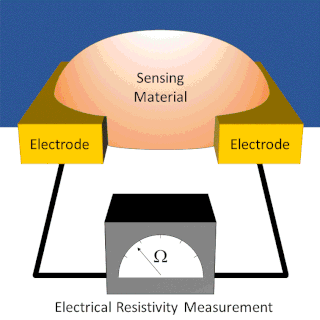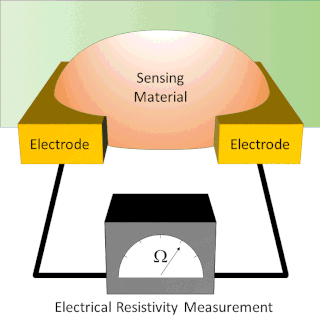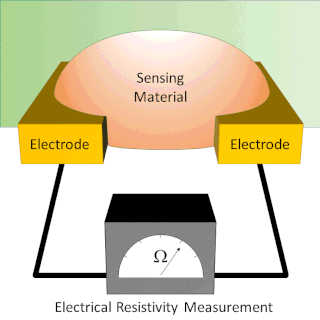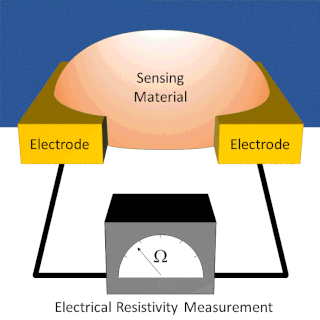
A chemiresistor is a material that changes its electrical resistance in response to changes in the nearby chemical environment. Chemiresistors are a class of chemical sensors that rely on the direct chemical interaction between the sensing material and the analyte. The sensing material and the analyte can interact by covalent bonding, hydrogen bonding, or molecular recognition. Several different materials have chemiresistor properties: metal-oxide semiconductors, some conductive polymers, and nanomaterials like graphene, carbon nanotubes and nanoparticles. Typically these materials are used as partially selective sensors in devices like electronic tongues or electronic noses.
There are many water purifiers available in the market which use different techniques like boiling, filtration, distillation, chlorination, sedimentation and oxidation. Currently nanotechnology plays a vital role in water purification techniques. Nanotechnology is the process of manipulating atoms on a nanoscale. In nanotechnology, nano membranes are used with the purpose of softening the water and removal of contaminants such as physical, biological and chemical contaminants. There are variety of techniques in nanotechnology which uses nano particles for providing safe drinking water with a high level of effectiveness. Some techniques have become commercialized.

Electrochemical stripping analysis is a set of analytical chemistry methods based on voltammetry or potentiometry that are used for quantitative determination of ions in solution. Stripping voltammetry have been employed for analysis of organic molecules as well as metal ions. Carbon paste, glassy carbon paste, and glassy carbon electrodes when modified are termed as chemically modified electrodes and have been employed for the analysis of organic and inorganic compounds.

Spectroelectrochemistry (SEC) is a set of multi-response analytical techniques in which complementary chemical information is obtained in a single experiment. Spectroelectrochemistry provides a whole vision of the phenomena that take place in the electrode process. The first spectroelectrochemical experiment was carried out by Theodore Kuwana, PhD, in 1964.
Screen-printed electrodes (SPEs) are electrochemical measurement devices that are manufactured by printing different types of ink on plastic or ceramic substrates, allowing quick in-situ analysis with high reproducibility, sensitivity and accuracy. The composition of the different inks used in the manufacture of the electrode determines its selectivity and sensitivity. This fact allows the analyst to design the most optimal device according to its purpose.
A chemical sensor array is a sensor architecture with multiple sensor components that create a pattern for analyte detection from the additive responses of individual sensor components. There exist several types of chemical sensor arrays including electronic, optical, acoustic wave, and potentiometric devices, which are described below. These chemical sensor arrays can employ multiple sensor types that are cross-reactive or tuned to sense specific analytes.
References
- ↑ Grygar, Tomáš; Marken, Frank; Schröder, Uwe; Scholz, Fritz (2002), "Electrochemical Analysis of Solids. A Review", Collection of Czechoslovak Chemical Communications, 67 (2): 163–208, doi:10.1135/cccc20020163
- ↑ Sanghavi, Bankim; Srivastava, Ashwini (2010). "Simultaneous voltammetric determination of acetaminophen, aspirin and caffeine using an in situ surfactant-modified multiwalled carbon nanotube paste electrode". Electrochimica Acta. 55 (28): 8638–8648. doi:10.1016/j.electacta.2010.07.093.