Related Research Articles
A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements as members of its ring(s). Heterocyclic organic chemistry is the branch of organic chemistry dealing with the synthesis, properties, and applications of organic heterocycles.
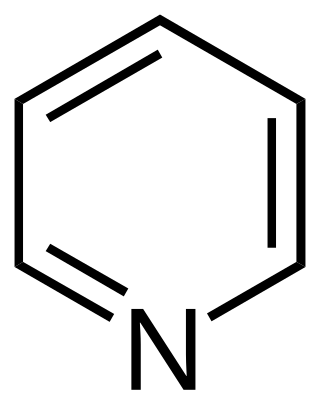
Simple aromatic rings, also known as simple arenes or simple aromatics, are aromatic organic compounds that consist only of a conjugated planar ring system. Many simple aromatic rings have trivial names. They are usually found as substructures of more complex molecules. Typical simple aromatic compounds are benzene, indole, and pyridine.

Triazines are a class of nitrogen-containing heterocycles. The parent molecules' molecular formula is C3H3N3. They exist in three isomeric forms, 1,3,5-triazines being common.
Azoles are a class of five-membered heterocyclic compounds containing a nitrogen atom and at least one other non-carbon atom as part of the ring. Their names originate from the Hantzsch–Widman nomenclature. The parent compounds are aromatic and have two double bonds; there are successively reduced analogs with fewer. One, and only one, lone pair of electrons from each heteroatom in the ring is part of the aromatic bonding in an azole. Names of azoles maintain the prefix upon reduction. The numbering of ring atoms in azoles starts with the heteroatom that is not part of a double bond, and then proceeds towards the other heteroatom.
Pyrazole is an organic compound of azole group with the formula C3H3N2H. It is a heterocycle characterized by a 5-membered ring of three carbon atoms and two adjacent nitrogen atoms, which are in ortho-substitution. Pyrazole is a weak base, with pKb 11.5 (pKa of the conjugate acid 2.49 at 25 °C). Pyrazoles are also a class of compounds that have the ring C3N2 with adjacent nitrogen atoms. Notable drugs containing a pyrazole ring are celecoxib (celebrex) and the anabolic steroid stanozolol.

Benzoxazole is an aromatic organic compound with a molecular formula C7H5NO, a benzene-fused oxazole ring structure, and an odor similar to pyridine. Although benzoxazole itself is of little practical value, many derivatives of benzoxazoles are commercially important.

Pyridazine is an aromatic, heterocyclic, organic compound with the molecular formula C4H4N2. It contains a six-membered ring with two adjacent nitrogen atoms. It is a colorless liquid with a boiling point of 208 °C. It is isomeric with two other diazine rings, pyrimidine and pyrazine.
1,2,3-Triazole is one of a pair of isomeric chemical compounds with molecular formula C2H3N3, called triazoles, which have a five-membered ring of two carbon atoms and three nitrogen atoms. 1,2,3-Triazole is a basic aromatic heterocycle.
Azirines are three-membered heterocyclic unsaturated compounds containing a nitrogen atom and related to the saturated analogue aziridine. They are highly reactive yet have been reported in a few natural products such as Dysidazirine. There are two isomers of azirine: 1H-Azirines with a carbon-carbon double bond are not stable and rearrange to the tautomeric 2H-azirine, a compound with a carbon-nitrogen double bond. 2H-Azirines can be considered strained imines and are isolable.

A cyclic compound is a term for a compound in the field of chemistry in which one or more series of atoms in the compound is connected to form a ring. Rings may vary in size from three to many atoms, and include examples where all the atoms are carbon, none of the atoms are carbon, or where both carbon and non-carbon atoms are present. Depending on the ring size, the bond order of the individual links between ring atoms, and their arrangements within the rings, carbocyclic and heterocyclic compounds may be aromatic or non-aromatic; in the latter case, they may vary from being fully saturated to having varying numbers of multiple bonds between the ring atoms. Because of the tremendous diversity allowed, in combination, by the valences of common atoms and their ability to form rings, the number of possible cyclic structures, even of small size numbers in the many billions.

Azepines are unsaturated heterocycles of seven atoms, with a nitrogen replacing a carbon at one position.
In chemistry, mesoionic compounds are one in which a heterocyclic structure is dipolar and where both the negative and the positive charges are delocalized. A completely uncharged structure cannot be written and mesoionic compounds cannot be represented satisfactorily by any one mesomeric structure. Mesoionic compounds are a subclass of betaines. Examples are sydnones and sydnone imines, münchnones, and mesoionic carbenes.

Oxazepines are a family of unsaturated heterocycles containing seven atoms, with a nitrogen replacing a carbon at one position and with an oxygen replacing a carbon at one position.
1,4,2-Dithiazole is a heterocyclic compound consisting of an unsaturated five-membered ring containing two carbon atoms, one nitrogen atom, and two sulfur atoms. 1,4,2-Dithiazole compounds may be formed by the reaction of nitrile sulfide with various reactive species; for instance thiocarbonyls via a 1,3-dipolar cycloaddition reaction. These compounds may be protonated by strong acids to give synthetically useful aromatic cations.

Bismole is a theoretical heterocyclic organic compound, a five-membered ring with the formula C4H4BiH. It is classified as a metallole. It can be viewed as a structural analog of pyrrole, with bismuth replacing the nitrogen atom of pyrrole. The unsubstituted compound has not been isolated due to the high energy of the Bi-H bond. Substituted derivatives, which have been synthesized, are called bismoles.
The Minisci reaction is a named reaction in organic chemistry. It is a nucleophilic radical substitution to an electron deficient aromatic compound, most commonly the introduction of an alkyl group to a nitrogen containing heterocycle. The reaction was published in 1971 by F. Minisci. In the case of N-Heterocycles, the conditions must be acidic to ensure protonation of said heterocycle. A typical reaction is that between pyridine and pivalic acid with silver nitrate, sulfuric acid and ammonium persulfate to form 2-tert-butylpyridine. The reaction resembles Friedel-Crafts alkylation but with opposite reactivity and selectivity.
Mesomeric betaines are dipolar heterocyclic compounds in which both the negative and the positive charges are delocalized.
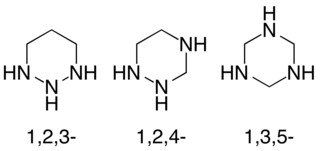
Triazinanes are a class of nitrogen-containing heterocycles. The parent molecules' molecular formula is (CH2)3(NH)3. They exist in three isomeric forms, 1,3,5-triazinanes being common. The triazinanes have six-membered cyclohexane-like ring but with three carbons replaced by nitrogens. Most commonly, the amines are tertiary.

Telluropyrylium is an aromatic heterocyclic compound consisting of a six member ring with five carbon atoms, and a positively charged tellurium atom. Derivatives of telluropyrylium are important in research of infrared dyes.
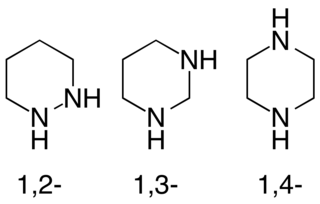
Diazinanes or hexahydrodiazines are a class of nitrogen-containing heterocycles consisting of a saturated four-carbon, two-nitrogen ring. They exist in three isomeric forms depending on the relative position of the two nitrogen atoms, with 1,4-diazinanes being common.
References
- ↑ Kawase, Masami (2009). "The Chemistry of Bioactive Mesoionic Heterocycles". Bioactive Heterocycles VII. Topics in Heterocyclic Chemistry. Vol. 16. Springer. pp. 135–152. doi:10.1007/7081_2007_096. ISBN 978-3-642-00335-6.