Histocompatibility, or tissue compatibility, is the property of having the same, or sufficiently similar, alleles of a set of genes called human leukocyte antigens (HLA), or major histocompatibility complex (MHC). Each individual expresses many unique HLA proteins on the surface of their cells, which signal to the immune system whether a cell is part of the self or an invading organism. T cells recognize foreign HLA molecules and trigger an immune response to destroy the foreign cells. Histocompatibility testing is most relevant for topics related to whole organ, tissue, or stem cell transplants, where the similarity or difference between the donor's HLA alleles and the recipient's triggers the immune system to reject the transplant. The wide variety of potential HLA alleles lead to unique combinations in individuals and make matching difficult.
Organ transplantation is a medical procedure in which an organ is removed from one body and placed in the body of a recipient, to replace a damaged or missing organ. The donor and recipient may be at the same location, or organs may be transported from a donor site to another location. Organs and/or tissues that are transplanted within the same person's body are called autografts. Transplants that are recently performed between two subjects of the same species are called allografts. Allografts can either be from a living or cadaveric source.

Liver transplantation or hepatic transplantation is the replacement of a diseased liver with the healthy liver from another person (allograft). Liver transplantation is a treatment option for end-stage liver disease and acute liver failure, although availability of donor organs is a major limitation. The most common technique is orthotopic transplantation, in which the native liver is removed and replaced by the donor organ in the same anatomic position as the original liver. The surgical procedure is complex, requiring careful harvest of the donor organ and meticulous implantation into the recipient. Liver transplantation is highly regulated, and only performed at designated transplant medical centers by highly trained transplant physicians and supporting medical team. The duration of the surgery ranges from 4 to 18 hours depending on outcome. Favorable outcomes require careful screening for eligible recipient, as well as a well-calibrated live or cadaveric donor match.

Transplant rejection occurs when transplanted tissue is rejected by the recipient's immune system, which destroys the transplanted tissue. Transplant rejection can be lessened by determining the molecular similitude between donor and recipient and by use of immunosuppressant drugs after transplant.
Anti-thymocyte globulin (ATG) is an infusion of horse or rabbit-derived antibodies against human T cells and their precursors (thymocytes), which is used in the prevention and treatment of acute rejection in organ transplantation and therapy of aplastic anemia due to bone marrow insufficiency.

Xenotransplantation, or heterologous transplant, is the transplantation of living cells, tissues or organs from one species to another. Such cells, tissues or organs are called xenografts or xenotransplants. It is contrasted with allotransplantation, syngeneic transplantation or isotransplantation and autotransplantation. Xenotransplantation is an artificial method of creating an animal-human chimera, that is, a human with a subset of animal cells. In contrast, an individual where each cell contains genetic material from a human and an animal is called a human–animal hybrid.
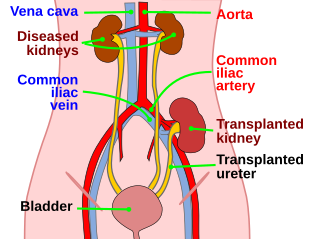
Kidney transplant or renal transplant is the organ transplant of a kidney into a patient with end-stage kidney disease (ESRD). Kidney transplant is typically classified as deceased-donor or living-donor transplantation depending on the source of the donor organ. Living-donor kidney transplants are further characterized as genetically related (living-related) or non-related (living-unrelated) transplants, depending on whether a biological relationship exists between the donor and recipient.
Alloimmunity is an immune response to nonself antigens from members of the same species, which are called alloantigens or isoantigens. Two major types of alloantigens are blood group antigens and histocompatibility antigens. In alloimmunity, the body creates antibodies against the alloantigens, attacking transfused blood, allotransplanted tissue, and even the fetus in some cases. Alloimmune (isoimmune) response results in graft rejection, which is manifested as deterioration or complete loss of graft function. In contrast, autoimmunity is an immune response to the self's own antigens. Alloimmunization (isoimmunization) is the process of becoming alloimmune, that is, developing the relevant antibodies for the first time.
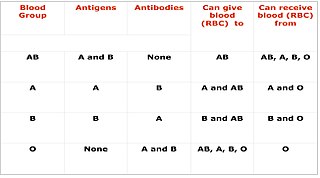
Cross-matching or crossmatching is a test performed before a blood transfusion as part of blood compatibility testing. Normally, this involves adding the recipient's blood plasma to a sample of the donor's red blood cells. If the blood is incompatible, the antibodies in the recipient's plasma will bind to antigens on the donor red blood cells. This antibody-antigen reaction can be detected through visible clumping or destruction of the red blood cells, or by reaction with anti-human globulin. Along with blood typing of the donor and recipient and screening for unexpected blood group antibodies, cross-matching is one of a series of steps in pre-transfusion testing. In some circumstances, an electronic cross-match can be performed by comparing records of the recipient's ABO and Rh blood type against that of the donor sample. In emergencies, blood may be issued before cross-matching is complete. Cross-matching is also used to determine compatibility between a donor and recipient in organ transplantation.

The BK virus is a member of the polyomavirus family. Past infection with the BK virus is widespread, but significant consequences of infection are uncommon, with the exception of the immunocompromised and the immunosuppressed. BK virus is an abbreviation of the name of the first patient, from whom the virus was isolated in 1971.
Tissue typing is a procedure in which the tissues of a prospective donor and recipient are tested for compatibility prior to transplantation. Mismatched donor and recipient tissues can lead to rejection of the tissues. There are multiple methods of tissue typing.
A panel-reactive antibody (PRA) is a group of antibodies in a test serum that are reactive against any of several known specific antigens in a panel of test leukocytes or purified HLA antigens from cells. It is an immunologic metric routinely performed by clinical laboratories on the blood of people awaiting organ transplantation.

Paul Ichiro Terasaki was an American scientist in the field of human organ transplant technology, and professor emeritus of surgery at UCLA School of Medicine.
Human leukocyte antigens (HLA) began as a list of antigens identified as a result of transplant rejection. The antigens were initially identified by categorizing and performing massive statistical analyses on interactions between blood types. This process is based upon the principle of serotypes. HLA are not typical antigens, like those found on surface of infectious agents. HLAs are alloantigens, they vary from individual to individual as a result of genetic differences. An organ called the thymus is responsible for ensuring that any T-cells that attack self proteins are not allowed to live. In essence, every individual's immune system is tuned to the specific set of HLA and self proteins produced by that individual; where this goes awry is when tissues are transferred to another person. Since individuals almost always have different "banks" of HLAs, the immune system of the recipient recognizes the transplanted tissue as non-self and destroys the foreign tissue, leading to transplant rejection. It was through the realization of this that HLAs were discovered.
Bernd Schröppel, M.D., is a former transplant nephrologist at the Mount Sinai Medical Center and the former Medical Director of the Kidney Pancreas Transplant Program at the Recanati/Miller Transplantation Institute at the Mount Sinai Medical Center in New York City. He is also a former Assistant Professor of Nephrology at the Mount Sinai School of Medicine.

Intestine transplantation is the surgical replacement of the small intestine for chronic and acute cases of intestinal failure. While intestinal failure can oftentimes be treated with alternative therapies such as parenteral nutrition (PN), complications such as PN-associated liver disease and short bowel syndrome may make transplantation the only viable option. One of the rarest type of organ transplantation performed, intestine transplantation is becoming increasingly prevalent as a therapeutic option due to improvements in immunosuppressive regimens, surgical technique, PN, and the clinical management of pre and post-transplant patients.
Complement-dependent cytotoxicity (CDC) is an effector function of IgG and IgM antibodies. When they are bound to surface antigen on target cell, the classical complement pathway is triggered by bonding protein C1q to these antibodies, resulting in formation of a membrane attack complex (MAC) and target cell lysis.
Kidney paired donation (KPD), or paired exchange, is an approach to living donor kidney transplantation where patients with incompatible donors swap kidneys to receive a compatible kidney. KPD is used in situations where a potential donor is incompatible. Because better donor HLA and age matching are correlated with lower lifetime mortality and longer lasting kidney transplants, many compatible pairs are also participating in swaps to find better matched kidneys. In the United States, the National Kidney Registry organizes the majority of U.S. KPD transplants, including the largest swaps. The first large swap was a 60 participant chain in 2012 that appeared on the front page of the New York Times and the second, even larger swap, included 70 participants and was completed in 2014. Other KPD programs in the U.S. include the UNOS program, which was launched in 2010 and completed its 100th KPD transplant in 2014, and the Alliance for Paired Donation.

William James Dempster was a British surgeon and researcher in organ transplantation at St Mary’s Hospital, London. He published more than 100 scientific reviews and papers on kidney transplant rejection in dogs, confirming that rejection was an example of immune response, mediated by serum antibodies.
Alexandre Loupy is a French nephrologist, a university professor and hospital practitioner at the Necker Hospital of the Assistance Publique - Hôpitaux de Paris, in the kidney transplant department. He is known for his discoveries on the topic of graft rejection.,, Its approach proposing innovative methodological tools has led to a better understanding but has also led to important changes in the international classification of graft rejection., These discoveries allow to improve the performance of clinical trials and to consider new therapeutic innovations in transplantation.









