Related Research Articles
A biocide is defined in the European legislation as a chemical substance or microorganism intended to destroy, deter, render harmless, or exert a controlling effect on any harmful organism. The US Environmental Protection Agency (EPA) uses a slightly different definition for biocides as "a diverse group of poisonous substances including preservatives, insecticides, disinfectants, and pesticides used for the control of organisms that are harmful to human or animal health or that cause damage to natural or manufactured products". When compared, the two definitions roughly imply the same, although the US EPA definition includes plant protection products and some veterinary medicines.

A safety data sheet (SDS), material safety data sheet (MSDS), or product safety data sheet (PSDS) is a document that lists information relating to occupational safety and health for the use of various substances and products. SDSs are a widely used system for cataloguing information on chemicals, chemical compounds, and chemical mixtures. SDS information may include instructions for the safe use and potential hazards associated with a particular material or product, along with spill-handling procedures. The older MSDS formats could vary from source to source within a country depending on national requirements; however, the newer SDS format is internationally standardized.

Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) is a European Union regulation dating from 18 December 2006. REACH addresses the production and use of chemical substances, and their potential impacts on both human health and the environment. Its 849 pages took seven years to pass, and it has been described as the most complex legislation in the Union's history and the most important in 20 years. It is the strictest law to date regulating chemical substances and will affect industries throughout the world. REACH entered into force on 1 June 2007, with a phased implementation over the next decade. The regulation also established the European Chemicals Agency, which manages the technical, scientific and administrative aspects of REACH.

The Dangerous Substances Directive was one of the main European Union laws concerning chemical safety, until its full replacement by the new regulation CLP Regulation (2008), starting in 2016. It was made under Article 100 of the Treaty of Rome. By agreement, it is also applicable in the EEA, and compliance with the directive will ensure compliance with the relevant Swiss laws. The Directive ceased to be in force on 31 May 2015 and was repealed by Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006.

The European Chemicals Agency is an agency of the European Union which manages the technical and administrative aspects of the implementation of the European Union regulation called Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH). ECHA is the driving force among regulatory authorities in implementing the EU's chemicals legislation. ECHA has to ascertain that companies comply with the legislation, advances the safe use of chemicals, provides information on chemicals and addresses chemicals of concern. It is located in Helsinki, Finland. ECHA is an independent and mature regulatory agency established by REACH. It is not a subsidiary entity of the European Commission.
IUCLID is a software application to capture, store, maintain and exchange data on intrinsic and hazard properties of chemical substances. Distributed free of charge, the software is especially useful to chemical industry companies and to government authorities. It is the key tool for chemical industry to fulfill data submission obligations under REACH, the most important European Union legal document covering the production and use of chemical substances. The software is maintained by the European Chemicals Agency, ECHA. The latest version, version 6, was made available on 29 April 2016.

The CLP Regulation is a European Union regulation from 2008, which aligns the European Union system of classification, labelling and packaging of chemical substances and mixtures to the Globally Harmonised System (GHS). It is expected to facilitate global trade and the harmonised communication of hazard information of chemicals and to promote regulatory efficiency. It complements the 2006 Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) Regulation and replaces an older system contained in the Dangerous Substances Directive (67/548/EEC) and the Dangerous Preparations Directive (1999/45/EC).
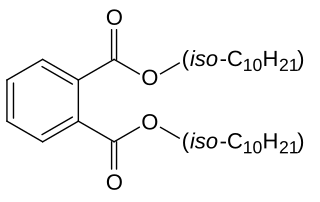
Diisodecyl phthalate (DIDP) is a commonly used plasticizer used in the production of plastic and plastic coating to increase flexibility. It is a mixture of compounds derived from the esterification of phthalic acid and isomeric decyl alcohols.
Hazard statements form part of the Globally Harmonized System of Classification and Labelling of Chemicals (GHS). They are intended to form a set of standardized phrases about the hazards of chemical substances and mixtures that can be translated into different languages. As such, they serve the same purpose as the well-known R-phrases, which they are intended to replace.
Hazard pictograms form part of the international Globally Harmonized System of Classification and Labelling of Chemicals (GHS). Two sets of pictograms are included within the GHS: one for the labelling of containers and for workplace hazard warnings, and a second for use during the transport of dangerous goods. Either one or the other is chosen, depending on the target audience, but the two are not used together. The two sets of pictograms use the same symbols for the same hazards, although certain symbols are not required for transport pictograms. Transport pictograms come in wider variety of colors and may contain additional information such as a subcategory number.
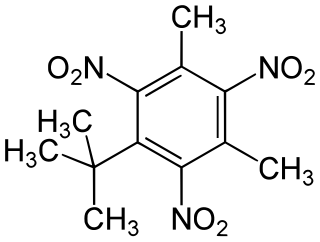
Musk xylene is a synthetic musk fragrance which mimics natural musk. It has been used as a perfume fixative in a wide variety of consumer products, and is still used in some cosmetics and fragrances.
A substance of very high concern (SVHC) is a chemical substance concerning which it has been proposed that use within the European Union be subject to authorisation under the REACH Regulation. Indeed, listing of a substance as an SVHC by the European Chemicals Agency (ECHA) is the first step in the procedure for authorisation or restriction of use of a chemical. The first list of SVHCs was published on 28 October 2008 and the list has been updated many times to include new candidates. The most recent update occurred in January 2022 to include a total of 223 SVHC.
The European chemical Substances Information System (ESIS) was a chemoinformatics database that stored information system on chemicals of the European Union. It was created by the former European Chemicals Bureau, which completed its mandate in 2008, in the year 2003. ESIS was set up by the Joint Research Centre of the European Commission in order to make data on the safety of chemicals more readily accessible to the public, offering a single search tool on chemicals and the legislation under which they are presently covered. By October 3, 2013, ESIS contained 14,897 substance records.
The Biocidal Products Directive (BPD) also known as the Biocides Directive is European Union Directive, (98/8/EC), which concerns biocides. It is officially known as Directive 98/8/EC of the European Parliament and of the Council of 16 February 1998 concerning the placing of biocidal products on the market. In 2013 the Biocidal Products Directive was superseded by The Biocidal Products Regulation.
European hazard symbols for chemicals are pictograms defined by the European Union for labelling chemical packaging and containers. They are standardised currently by the CLP/GHS classification.
The Substitute It Now! List is a database developed by the International Chemical Secretariat (ChemSec) of chemicals the uses of which are likely to become legally restricted under EU REACH regulation. The list is being used by public interest groups as a campaign tool to advocate for increasing the pace of implementation of REACH and by commercial interests to identify substances for control in chemicals management programmes.
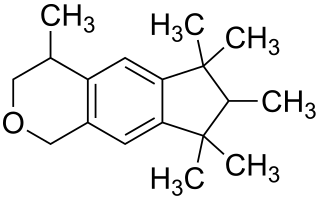
Galaxolide is a synthetic musk with a clean sweet musky floral woody odor used in fragrances. It is one of the musk components that perfume and cologne manufacturers use to add a musk odor to their products. Galaxolide was first synthesized in 1965, and used in the late 1960s in some fabric softeners and detergents. High concentrations were also incorporated in fine fragrances.

The European Partnership for Alternative Approaches to Animal Testing (EPAA) promotes the application of the 3Rs principles in meeting regulatory requirements for products such as pharmaceuticals, chemicals, soaps, detergents and cosmetics. The EPAA covers research and development, as well as the use of 3Rs approaches in regulatory compliance and communication and dissemination.

A pesticide, also called Plant Protection Product (PPP), which is a term used in regulatory documents, consists of several different components. The active ingredient in a pesticide is called “active substance” and these active substances either consist of chemicals or micro-organisms. The aims of these active substances are to specifically take action against organisms that are harmful to plants. In other words, active substances are the active components against pests and plant diseases.
References
- ↑ JRC-IHCP "former ECB", September 2011. Retrieved on 27 September 2011.
- ↑ Eisenreich, Steven; Allanou, Remi; Aschberger, Karin; Baraibar, FENTANES Joaquin; Berggren, Katarina; Cole, Thomas; Christensen, Frans; Langezaal, Ingrid; Macchi, Chiara (23 July 2008). "The European Chemicals Bureau: an Overview of 15 Years Experience in EU Chemicals Legislation". JRC Publications Repository. Retrieved 11 December 2022.