Related Research Articles

Polysaccharides, or polycarbohydrates, are the most abundant carbohydrates found in food. They are long chain polymeric carbohydrates composed of monosaccharide units bound together by glycosidic linkages. This carbohydrate can react with water (hydrolysis) using amylase enzymes as catalyst, which produces constituent sugars. They range in structure from linear to highly branched. Examples include storage polysaccharides such as starch, glycogen and galactogen and structural polysaccharides such as cellulose and chitin.
Glycomics is the comprehensive study of glycomes, including genetic, physiologic, pathologic, and other aspects. Glycomics "is the systematic study of all glycan structures of a given cell type or organism" and is a subset of glycobiology. The term glycomics is derived from the chemical prefix for sweetness or a sugar, "glyco-", and was formed to follow the omics naming convention established by genomics and proteomics.

Glycoproteins are proteins which contain oligosaccharide chains (glycans) covalently attached to amino acid side-chains. The carbohydrate is attached to the protein in a cotranslational or posttranslational modification. This process is known as glycosylation. Secreted extracellular proteins are often glycosylated.
A congenital disorder of glycosylation is one of several rare inborn errors of metabolism in which glycosylation of a variety of tissue proteins and/or lipids is deficient or defective. Congenital disorders of glycosylation are sometimes known as CDG syndromes. They often cause serious, sometimes fatal, malfunction of several different organ systems in affected infants. The most common sub-type is PMM2-CDG where the genetic defect leads to the loss of phosphomannomutase 2 (PMM2), the enzyme responsible for the conversion of mannose-6-phosphate into mannose-1-phosphate.
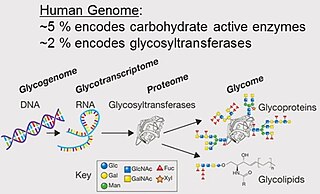
The glycome is the entire complement of sugars, whether free or present in more complex molecules, of an organism. An alternative definition is the entirety of carbohydrates in a cell. The glycome may in fact be one of the most complex entities in nature. "Glycomics, analogous to genomics and proteomics, is the systematic study of all glycan structures of a given cell type or organism" and is a subset of glycobiology.

Cellulase is any of several enzymes produced chiefly by fungi, bacteria, and protozoans that catalyze cellulolysis, the decomposition of cellulose and of some related polysaccharides. The name is also used for any naturally occurring mixture or complex of various such enzymes, that act serially or synergistically to decompose cellulosic material.
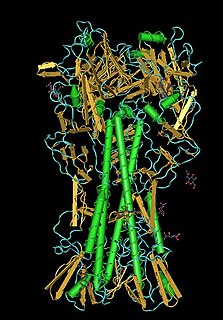
Lectins are carbohydrate-binding proteins that are highly specific for sugar groups that are part of other molecules, so cause agglutination of particular cells or precipitation of glycoconjugates and polysaccharides. Lectins have a role in recognition at the cellular and molecular level and play numerous roles in biological recognition phenomena involving cells, carbohydrates, and proteins. Lectins also mediate attachment and binding of bacteria, viruses, and fungi to their intended targets.
An oligosaccharide is a saccharide polymer containing a small number of monosaccharides. Oligosaccharides can have many functions including cell recognition and cell adhesion.
An Endoglycosidase is an enzyme that releases oligosaccharides from glycoproteins or glycolipids. It may also cleave polysaccharide chains between residues that are not the terminal residue, although releasing oligosaccharides from conjugated protein and lipid molecules is more common.
The terms glycans and polysaccharides are defined by IUPAC as synonyms meaning "compounds consisting of a large number of monosaccharides linked glycosidically". However, in practice the term glycan may also be used to refer to the carbohydrate portion of a glycoconjugate, such as a glycoprotein, glycolipid, or a proteoglycan, even if the carbohydrate is only an oligosaccharide. Glycans usually consist solely of O-glycosidic linkages of monosaccharides. For example, cellulose is a glycan composed of β-1,4-linked D-glucose, and chitin is a glycan composed of β-1,4-linked N-acetyl-D-glucosamine. Glycans can be homo- or heteropolymers of monosaccharide residues, and can be linear or branched.
Cyanines, also referred to as tetramethylindo(di)-carbocyanines are a synthetic dye family belonging to the polymethine group. Although the name derives etymologically from terms for shades of blue, the cyanine family covers the electromagnetic spectrum from near IR to UV.
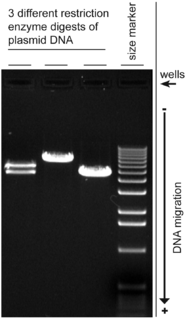
A molecular-weight size marker, also referred to as a protein ladder, DNA ladder, or RNA ladder, is a set of standards that are used to identify the approximate size of a molecule run on a gel during electrophoresis, using the principle that molecular weight is inversely proportional to migration rate through a gel matrix. Therefore, when used in gel electrophoresis, markers effectively provide a logarithmic scale by which to estimate the size of the other fragments.
The enzyme Endoglycosidase H is an enzyme with systematic name glycopeptide-D-mannosyl-N4-(N-acetyl-D-glucosaminyl)2-asparagine 1,4-N-acetyl-beta-glucosaminohydrolase. It is a highly specific endoglycosidase which cleaves asparagine-linked mannose rich oligosaccharides, but not highly processed complex oligosaccharides from glycoproteins. It is used for research purposes to deglycosylate glycoproteins.
Oligosaccharides and polysaccharides are an important class of polymeric carbohydrates found in virtually all living entities. Their structural features make their nomenclature challenging and their roles in living systems make their nomenclature important.
Carbohydrate NMR Spectroscopy is the application of nuclear magnetic resonance (NMR) spectroscopy to structural and conformational analysis of carbohydrates. This method allows the scientists to elucidate structure of monosaccharides, oligosaccharides, polysaccharides, glycoconjugates and other carbohydrate derivatives from synthetic and natural sources. Among structural properties that could be determined by NMR are primary structure, saccharide conformation, stoichiometry of substituents, and ratio of individual saccharides in a mixture. Modern high field NMR instruments used for carbohydrate samples, typically 500 MHz or higher, are able to run a suite of 1D, 2D, and 3D experiments to determine a structure of carbohydrate compounds.
Carbohydrate synthesis is a sub-field of organic chemistry concerned specifically with the generation of natural and unnatural carbohydrate structures. This can include the synthesis of monosaccharide residues or structures containing more than one monosaccharide, known as oligosaccharides.
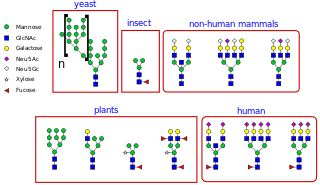
N-linked glycosylation, is the attachment of an oligosaccharide, a carbohydrate consisting of several sugar molecules, sometimes also referred to as glycan, to a nitrogen atom, in a process called N-glycosylation, studied in biochemistry. This type of linkage is important for both the structure and function of many eukaryotic proteins. The N-linked glycosylation process occurs in eukaryotes and widely in archaea, but very rarely in bacteria. The nature of N-linked glycans attached to a glycoprotein is determined by the protein and the cell in which it is expressed. It also varies across species. Different species synthesize different types of N-linked glycan.

Peptide:N-glycosidase F, commonly referred to as PNGase F, is an amidase of the peptide-N4-(N-acetyl-beta-glucosaminyl)asparagine amidase class. PNGase F works by cleaving between the innermost GlcNAc and asparagine residues of high mannose, hybrid, and complex oligosaccharides from N-linked glycoproteins and glycopeptides. This results in a deaminated protein or peptide and a free glycan.
Ten Feizi is a British molecular biologist who is Professor and Director of the Glycosciences Laboratory at Imperial College London. Her research considers the structure and function of glycans. She was awarded the Society for Glycobiology Rosalind Kornfeld award in 2014. She was also awarded the Fellowship of the Academy of Medical Sciences in 2021.
Rosalind Hauk Kornfeld (1935–2007) was a scientist at Washington University in St. Louis known for her research determining the structure and formation of oligosaccharides. The Society of Glycobiology annually awards a lifetime achievement award in her honor.
References
- ↑ Harish, P. M. Kumar (July 23, 1999). "Use of Fluorophore-Assisted Carbohydrate Electrophoresis (FACE®) in the Elucidation of N-Linked Oligosaccharide Structures". Use of Fluorophore-Assisted Carbohydrate Electrophoresis (FACE) in the Elucidation of N-Linked Oligosaccharide Structures. Methods in Biotechnology. Vol. 10. pp. 221–234. doi:10.1007/978-1-59259-261-6_18. ISBN 978-0-89603-563-8.