
Bacillus cereus is a Gram-positive, rod-shaped, facultatively anaerobic, motile, beta-hemolytic, spore forming bacterium commonly found in soil and food. The specific name, cereus, meaning "waxy" in Latin, refers to the appearance of colonies grown on blood agar. Some strains are harmful to humans and cause foodborne illness, while other strains can be beneficial as probiotics for animals. The bacteria is classically contracted from fried rice dishes that have been sitting at room temperature for hours. B. cereus bacteria are facultative anaerobes, and like other members of the genus Bacillus, can produce protective endospores. Its virulence factors include cereolysin and phospholipase C.

Shiga toxins are a family of related toxins with two major groups, Stx1 and Stx2, expressed by genes considered to be part of the genome of lambdoid prophages. The toxins are named after Kiyoshi Shiga, who first described the bacterial origin of dysentery caused by Shigella dysenteriae. Shiga-like toxin (SLT) is a historical term for similar or identical toxins produced by Escherichia coli. The most common sources for Shiga toxin are the bacteria S. dysenteriae and some serotypes of Escherichia coli (STEC), which includes serotypes O157:H7, and O104:H4.
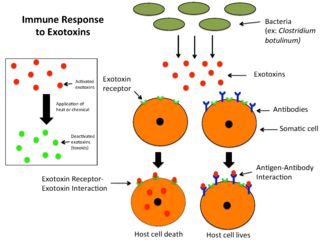
An exotoxin is a toxin secreted by bacteria. An exotoxin can cause damage to the host by destroying cells or disrupting normal cellular metabolism. They are highly potent and can cause major damage to the host. Exotoxins may be secreted, or, similar to endotoxins, may be released during lysis of the cell. Gram negative pathogens may secrete outer membrane vesicles containing lipopolysaccharide endotoxin and some virulence proteins in the bounding membrane along with some other toxins as intra-vesicular contents, thus adding a previously unforeseen dimension to the well-known eukaryote process of membrane vesicle trafficking, which is quite active at the host-pathogen interface.
Pathogenicity islands (PAIs), as termed in 1990, are a distinct class of genomic islands acquired by microorganisms through horizontal gene transfer. Pathogenicity islands are found in both animal and plant pathogens. Additionally, PAIs are found in both gram-positive and gram-negative bacteria. They are transferred through horizontal gene transfer events such as transfer by a plasmid, phage, or conjugative transposon. Therefore, PAIs contribute to microorganisms' ability to evolve.
Adenylate cyclase toxin is a virulence factor produced by some members of the genus Bordetella. Together with the pertussis toxin it is the most important virulence factor of the causative agent of whooping cough, Bordetella pertussis. Bordetella bronchiseptica and Bordetella parapertussis, also able to cause pertussis-like symptoms, also produce adenylate cyclase toxin. It is a toxin secreted by the bacteria to influence the host immune system.
Cytolysin refers to the substance secreted by microorganisms, plants or animals that is specifically toxic to individual cells, in many cases causing their dissolution through lysis. Cytolysins that have a specific action for certain cells are named accordingly. For instance, the cytolysins responsible for the destruction of red blood cells, thereby liberating hemoglobins, are named hemolysins, and so on. Cytolysins may be involved in immunity as well as in venoms.

Pore-forming proteins are usually produced by bacteria, and include a number of protein exotoxins but may also be produced by other organisms such as earthworms, who produce lysenin. They are frequently cytotoxic, as they create unregulated pores in the membrane of targeted cells.
The AB5 toxins are six-component protein complexes secreted by certain pathogenic bacteria known to cause human diseases such as cholera, dysentery, and hemolytic–uremic syndrome. One component is known as the A subunit, and the remaining five components are B subunits. All of these toxins share a similar structure and mechanism for entering targeted host cells. The B subunit is responsible for binding to receptors to open up a pathway for the A subunit to enter the cell. The A subunit is then able to use its catalytic machinery to take over the host cell's regular functions.

Hemolysins or haemolysins are lipids and proteins that cause lysis of red blood cells by disrupting the cell membrane. Although the lytic activity of some microbe-derived hemolysins on red blood cells may be of great importance for nutrient acquisition, many hemolysins produced by pathogens do not cause significant destruction of red blood cells during infection. However, hemolysins are often capable of lysing red blood cells in vitro.
Listeriolysin O (LLO) is a hemolysin produced by the bacterium Listeria monocytogenes, the pathogen responsible for causing listeriosis. The toxin may be considered a virulence factor, since it is crucial for the virulence of L. monocytogenes.

Alpha-toxin, also known as alpha-hemolysin (Hla), is the major cytotoxic agent released by bacterium Staphylococcus aureus and the first identified member of the pore forming beta-barrel toxin family. This toxin consists mostly of beta-sheets (68%) with only about 10% alpha-helices. The hly gene on the S. aureus chromosome encodes the 293 residue protein monomer, which forms heptameric units on the cellular membrane to form a complete beta-barrel pore. This structure allows the toxin to perform its major function, development of pores in the cellular membrane, eventually causing cell death.
'Staphylococcus aureus delta toxin is a toxin produced by Staphylococcus aureus. It has a wide spectrum of cytolytic activity.
Clostridium novyi (oedematiens) a Gram-positive, endospore- forming, obligate anaerobic bacteria of the class clostridia. It is ubiquitous, being found in the soil and faeces. It is pathogenic, causing a wide variety of diseases in man and animals. It comes in three types, labelled A, B, and a non-pathogenic type C distinguished by the range of toxins they produce. Some authors include Clostridium haemolyticum as Clostridium novyi type D. C novyi is closely related to Clostridium botulinum types C and D as Yoshimasa Sasaki et al. have demonstrated by 16S rDNA sequence analysis.
John Hagan Pryce Bayley FRS is a British scientist, who holds the position of Professor of Chemical Biology at the University of Oxford.
The RTX toxin superfamily is a group of cytolysins and cytotoxins produced by bacteria. There are over 1000 known members with a variety of functions. The RTX family is defined by two common features: characteristic repeats in the toxin protein sequences, and extracellular secretion by the type I secretion systems (T1SS). The name RTX refers to the glycine and aspartate-rich repeats located at the C-terminus of the toxin proteins, which facilitate export by a dedicated T1SS encoded within the rtx operon.
Adenylate cyclase toxin (CyaA) is released or secreted from bacterium Bordetella pertussis by the T1SS in the host’s respiratory tract in order to suppress its early innate and subsequent adaptive immune defense.
In molecular biology, the Bacillus haemolytic enterotoxin family of proteins consists of several Bacillus haemolytic enterotoxins, which can cause food poisoning in humans. Haemolysin BL and non-haemolytic enterotoxin, represent the major enterotoxins produced by Bacillus cereus. Most of the cytotoxic activity of B. cereus isolates has been attributed to the level of Nhe, which may indicate a highly diarrheic potential. The exact mechanism by which B. cereus causes diarrhoea is unknown. Hbl, cytotoxin K (CytK) and Nhe are all putative causes.

In molecular biology, the haemolysin expression modulating protein family is a family of proteins. This family consists of haemolysin expression modulating protein (Hha) from Escherichia coli and its enterobacterial homologues, such as YmoA from Yersinia enterocolitica, and RmoA encoded on the R100 plasmid. These proteins act as modulators of bacterial gene expression. Members of this family act in conjunction with members of the H-NS family, participating in the thermoregulation of different virulence factors and in plasmid transfer. Hha, along with the chromatin-associated protein H-NS, is involved in the regulation of expression of the toxin alpha-haemolysin in response to osmolarity and temperature. YmoA modulates the expression of various virulence factors, such as Yop proteins and YadA adhesin, in response to temperature. RmoA is a plasmid R100 modulator involved in plasmid transfer. The HHA family of proteins display striking similarity to the oligomerisation domain of the H-NS proteins.
The Lambda Holin S Family is a group of integral membrane transporter proteins belonging to the Holin Superfamily III. Members of this family generally consist of the characteristic three transmembrane segments (TMSs) and are of 110 amino acyl residues (aas) in length, on average. A representative list of members belonging to this family can be found in the Transporter Classification Database.

Cry6Aa is a toxic crystal protein generated by the bacterial family Bacillus thuringiensis during sporulation. This protein is a member of the alpha pore forming toxins family, which gives it insecticidal qualities advantageous in agricultural pest control. Each Cry protein has some level of target specificity; Cry6Aa has specific toxic action against coleopteran insects and nematodes. The corresponding B. thuringiensis gene, cry6aa, is located on bacterial plasmids. Along with several other Cry protein genes, cry6aa can be genetically recombined in Bt corn and Bt cotton so the plants produce specific toxins. Insects are developing resistance to the most commonly inserted proteins like Cry1Ac. Since Cry6Aa proteins function differently than other Cry proteins, they are combined with other proteins to decrease the development of pest resistance. Recent studies suggest this protein functions better in combination with other virulence factors such as other Cry proteins and metalloproteinases.






