
Pesticides are substances that are used to control pests. They include herbicides, insecticides, nematicides, fungicides, and many others. The most common of these are herbicides, which account for approximately 50% of all pesticide use globally. Most pesticides are used as plant protection products, which in general protect plants from weeds, fungi, or insects. In general, a pesticide is a chemical or biological agent that deters, incapacitates, kills, or otherwise discourages pests. Target pests can include insects, plant pathogens, weeds, molluscs, birds, mammals, fish, nematodes (roundworms), and microbes that destroy property, cause nuisance, or spread disease, or are disease vectors. Along with these benefits, pesticides also have drawbacks, such as potential toxicity to humans and other species.

Herbicides, also commonly known as weed killers, are substances used to control undesired plants, also known as weeds. Selective herbicides control specific weed species while leaving the desired crop relatively unharmed, while non-selective herbicides (sometimes called total weed killers kill plants indiscriminately. Due to herbicide resistance – a major concern in agriculture – a number of products combine herbicides with different means of action. Integrated pest management may use herbicides alongside other pest control methods.

Pesticide resistance describes the decreased susceptibility of a pest population to a pesticide that was previously effective at controlling the pest. Pest species evolve pesticide resistance via natural selection: the most resistant specimens survive and pass on their acquired heritable changes traits to their offspring. If a pest has resistance then that will reduce the pesticide's efficacy – efficacy and resistance are inversely related.

Glyphosate is a broad-spectrum systemic herbicide and crop desiccant. It is an organophosphorus compound, specifically a phosphonate, which acts by inhibiting the plant enzyme 5-enolpyruvylshikimate-3-phosphate synthase (EPSP). It is used to kill weeds, especially annual broadleaf weeds and grasses that compete with crops. Its herbicidal effectiveness was discovered by Monsanto chemist John E. Franz in 1970. Monsanto brought it to market for agricultural use in 1974 under the trade name Roundup. Monsanto's last commercially relevant United States patent expired in 2000.

Paraquat (trivial name; ), or N,N′-dimethyl-4,4′-bipyridinium dichloride (systematic name), also known as methyl viologen, is an organic compound with the chemical formula [(C6H7N)2]Cl2. It is classified as a viologen, a family of redox-active heterocycles of similar structure. This salt is one of the most widely used herbicides. It is quick-acting and non-selective, killing green plant tissue on contact. It is also toxic (lethal) to human beings and animals due to its redox activity, which produces superoxide anions. It has been linked to the development of Parkinson's disease and is banned in 58 countries.

Phenoxy herbicides are two families of chemicals that have been developed as commercially important herbicides, widely used in agriculture. They share the part structure of phenoxyacetic acid.

Glufosinate is a naturally occurring broad-spectrum herbicide produced by several species of Streptomyces soil bacteria. Glufosinate is a non-selective, contact herbicide, with some systemic action. Plants may also metabolize bialaphos and phosalacine, other naturally occurring herbicides, directly into glufosinate. The compound irreversibly inhibits glutamine synthetase, an enzyme necessary for the production of glutamine and for ammonia detoxification, giving it antibacterial, antifungal and herbicidal properties. Application of glufosinate to plants leads to reduced glutamine and elevated ammonia levels in tissues, halting photosynthesis and resulting in plant death.
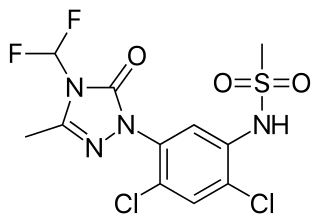
Sulfentrazone is the ISO common name for an organic compound used as a broad-spectrum herbicide. It acts by inhibiting the enzyme protoporphyrinogen oxidase. It was first marketed in the US in 1997 by FMC Corporation with the brand name Authority.
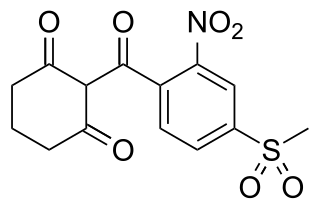
Mesotrione is a selective herbicide used mainly in maize crops. It is a synthetic compound inspired by the natural substance leptospermone found in the bottlebrush tree Callistemon citrinus. It inhibits the enzyme 4-hydroxyphenylpyruvate dioxygenase (HPPD) and is sold under brand names including Callisto and Tenacity. It was first marketed by Syngenta in 2001.

2,4-Dichlorophenoxyacetic acid is an organic compound with the chemical formula Cl2C6H3OCH2CO2H. It is usually referred to by its ISO common name 2,4-D. It is a systemic herbicide that kills most broadleaf weeds by causing uncontrolled growth, but most grasses such as cereals, lawn turf, and grassland are relatively unaffected.

Acifluorfen is the ISO common name for an organic compound used as an herbicide. It acts by inhibiting the enzyme protoporphyrinogen oxidase which is necessary for chlorophyll synthesis. Soybeans naturally have a high tolerance to acifluorfen and its salts, via metabolic disposal by glutathione S-transferase. It is effective against broadleaf weeds and grasses and is used agriculturally on fields growing soybeans, peanuts, peas, and rice.
5-enolpyruvylshikimate-3-phosphate (EPSP) synthase is an enzyme produced by plants and microorganisms. EPSPS catalyzes the chemical reaction:
4-Hydroxyphenylpyruvate dioxygenase (HPPD) inhibitors are a class of herbicides that prevent growth in plants by blocking 4-Hydroxyphenylpyruvate dioxygenase, an enzyme in plants that breaks down the amino acid tyrosine into molecules that are then used by plants to create other molecules that plants need. This process of breakdown, or catabolism, and making new molecules from the results, or biosynthesis, is something all living things do. HPPD inhibitors were first brought to market in 1980, although their mechanism of action was not understood until the late 1990s. They were originally used primarily in Japan in rice production, but since the late 1990s have been used in Europe and North America for corn, soybeans, and cereals, and since the 2000s have become more important as weeds have become resistant to glyphosate and other herbicides. Genetically modified crops are under development that include resistance to HPPD inhibitors. There is a pharmaceutical drug on the market, nitisinone, that was originally under development as an herbicide as a member of this class, and is used to treat an orphan disease, type I tyrosinemia.
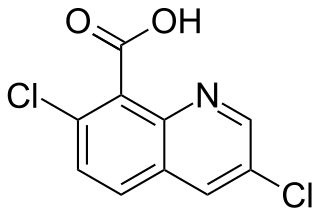
Quinclorac is an organic compound with the formula {C9NH4Cl2CO2H. A colorless solid, it is soluble in hydrocarbons and alcohols. The compound is a derivative of quinoline.
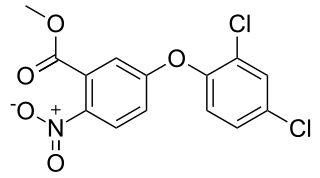
Bifenox is the ISO common name for an organic compound used as an herbicide. It acts by inhibiting the enzyme protoporphyrinogen oxidase which is necessary for chlorophyll synthesis.
Evolutionary toxicology is an emerging field of science focusing on shifts in population genetics caused by the introduction of contaminants to the environment. Research in evolutionary toxicology combines aspects of ecotoxicology, population genetics, evolutionary biology, and conservation genetics to form a unified field investigating genome and population wide changes in genetic diversity, allelic frequency, gene flow, and mutation rates. Each of these areas of investigation is characterized as one of four central tenets to the field, proposed and described in detail by John Bickham in 2011.

Fomesafen is the ISO common name for an organic compound used as an herbicide. It acts by inhibiting the enzyme protoporphyrinogen oxidase (PPO) which is necessary for chlorophyll synthesis. Soybeans naturally have a high tolerance to fomesafen, via metabolic disposal by glutathione S-transferase. As a result, soy is the most common crop treated with fomesafen, followed by other beans and a few other crop types. It is not safe for maize/corn or other Poaceae.

Fluazifop is the common name used by the ISO for an organic compound that is used as a selective herbicide. The active ingredient is the 2R enantiomer at its chiral centre and this material is known as fluazifop-P when used in that form. More commonly, it is sold as its butyl ester, fluazifop-P butyl with the brand name Fusilade.

Indaziflam is a preemergent herbicide especially for grass control in tree and bush crops.

Aclonifen is a diphenyl ether herbicide which has been used in agriculture since the 1980s. Its mode of action has been uncertain, with evidence suggesting it might interfere with carotenoid biosynthesis or inhibit the enzyme protoporphyrinogen oxidase (PPO). Both mechanisms could result in the observed whole-plant effect of bleaching and the compound includes chemical features that are known to result in PPO effects, as seen with acifluorfen, for example. In 2020, further research revealed that aclonifen has a different and novel mode of action, targeting solanesyl diphosphate synthase which would also cause bleaching.


















