Related Research Articles

Natural killer cells, also known as NK cells or large granular lymphocytes (LGL), are a type of cytotoxic lymphocyte critical to the innate immune system that belong to the rapidly expanding family of known innate lymphoid cells (ILC) and represent 5–20% of all circulating lymphocytes in humans. The role of NK cells is analogous to that of cytotoxic T cells in the vertebrate adaptive immune response. NK cells provide rapid responses to virus-infected cell and other intracellular pathogens acting at around 3 days after infection, and respond to tumor formation. Typically, immune cells detect the antigen presented on major histocompatibility complex (MHC) on infected cell surfaces, triggering cytokine release, causing the death of the infected cell by lysis or apoptosis. NK cells are unique, however, as they have the ability to recognize and kill stressed cells in the absence of antibodies and MHC, allowing for a much faster immune reaction. They were named "natural killers" because of the notion that they do not require activation to kill cells that are missing "self" markers of MHC class I. This role is especially important because harmful cells that are missing MHC I markers cannot be detected and destroyed by other immune cells, such as T lymphocyte cells.
Immunotherapy or biological therapy is the treatment of disease by activating or suppressing the immune system. Immunotherapies designed to elicit or amplify an immune response are classified as activation immunotherapies, while immunotherapies that reduce or suppress are classified as suppression immunotherapies. Immunotherapy is under preliminary research for its potential to treat various forms of cancer.
Cancer immunotherapy is the stimulation of the immune system to treat cancer, improving on the immune system's natural ability to fight the disease. It is an application of the fundamental research of cancer immunology and a growing subspecialty of oncology.

Cluster of differentiation 40, CD40 is a type I transmembrane protein found on antigen-presenting cells and is required for their activation. The binding of CD154 (CD40L) on TH cells to CD40 activates antigen presenting cells and induces a variety of downstream effects.

Integrin alpha M (ITGAM) is one protein subunit that forms heterodimeric integrin alpha-M beta-2 (αMβ2) molecule, also known as macrophage-1 antigen (Mac-1) or complement receptor 3 (CR3). ITGAM is also known as CR3A, and cluster of differentiation molecule 11B (CD11B). The second chain of αMβ2 is the common integrin β2 subunit known as CD18, and integrin αMβ2 thus belongs to the β2 subfamily integrins.

Monoclonal antibody therapy is a form of immunotherapy that uses monoclonal antibodies (mAbs) to bind monospecifically to certain cells or proteins. The objective is that this treatment will stimulate the patient's immune system to attack those cells. Alternatively, in radioimmunotherapy a radioactive dose localizes a target cell line, delivering lethal chemical doses. Antibodies are used to bind to molecules involved in T-cell regulation to remove inhibitory pathways that block T-cell responses. This is known as immune checkpoint therapy.

CD137, a member of the tumor necrosis factor (TNF) receptor family, is a type 1 transmembrane protein, expressed on surfaces of leukocytes and non-immune cells. Its alternative names are tumor necrosis factor receptor superfamily member 9 (TNFRSF9), 4-1BB, and induced by lymphocyte activation (ILA). It is of interest to immunologists as a co-stimulatory immune checkpoint molecule, and as a potential target in cancer immunotherapy.
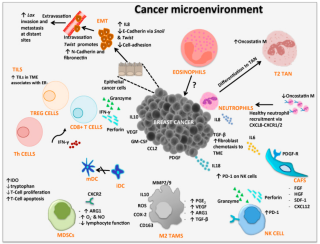
Cancer immunology is an interdisciplinary branch of biology that is concerned with understanding the role of the immune system in the progression and development of cancer; the most well known application is cancer immunotherapy, which utilises the immune system as a treatment for cancer. Cancer immunosurveillance and immunoediting are based on protection against development of tumors in animal systems and (ii) identification of targets for immune recognition of human cancer.

Programmed death-ligand 1 (PD-L1) also known as cluster of differentiation 274 (CD274) or B7 homolog 1 (B7-H1) is a protein that in humans is encoded by the CD274 gene.
Chemoimmunotherapy is chemotherapy combined with immunotherapy. Chemotherapy uses different drugs to kill or slow the growth of cancer cells; immunotherapy uses treatments to stimulate or restore the ability of the immune system to fight cancer. A common chemoimmunotherapy regimen is CHOP combined with rituximab (CHOP-R) for B-cell non-Hodgkin lymphomas.

Triggering receptor expressed on myeloid cells 1 (TREM1) an immunoglobulin (Ig) superfamily transmembrane protein that, in humans, is encoded by the TREM1 gene. TREM1 is constitutively expressed on the surface of peripheral blood monocytes and neutrophils, and upregulated by toll-like receptor (TLR) ligands; activation of TREM1 amplifies immune responses.

OX-2 membrane glycoprotein, also named CD200 is a human protein encoded by the CD200 gene. CD200 gene is in human located on chromosome 3 in proximity to genes encoding other B7 proteins CD80/CD86. In mice CD200 gene is on chromosome 16.

Macrophage receptor with collagenous structure (MARCO) is a protein that in humans is encoded by the MARCO gene. MARCO is a class A scavenger receptor that is found on particular subsets of macrophages. Scavenger receptors are pattern recognition receptors (PRRs) found most commonly on immune cells. Their defining feature is that they bind to polyanions and modified forms of a type of cholesterol called low-density lipoprotein (LDL). MARCO is able to bind and phagocytose these ligands and pathogen-associated molecular patterns (PAMPs), leading to the clearance of pathogens and cell signaling events that lead to inflammation. As part of the innate immune system, MARCO clears, or scavenges, pathogens, which leads to inflammatory responses. The scavenger receptor cysteine-rich (SRCR) domain at the end of the extracellular side of MARCO binds ligands to activate the subsequent immune responses. MARCO expression on macrophages has been associated with tumor development and also with Alzheimer's disease, via decreased responses of cells when ligands bind to MARCO.
H. Michael Shepard is an American cancer researcher who was awarded the 2019 Lasker Clinical research award, which he shared with Dennis Slamon and Axel Ullrich for their development of Herceptin

John Mendelsohn was a president of the University of Texas MD Anderson Cancer Center in Houston. He was an internationally recognized leader in cancer research.
Urelumab is a fully human, non‐ligand binding, CD137 agonist immunoglobulin‐γ 4 (IgG4) monoclonal antibody. It was developed utilizing Medarex's UltiMAb(R) technology by Bristol-Myers Squibb for the treatment of cancer and solid tumors. Urelumab promotes anti-tumor immunity, or an immune response against tumor cells, via CD137 activation. The application of Urelumab has been limited due to the fact that it can cause severe liver toxicity.

Immune checkpoints are regulators of the immune system. These pathways are crucial for self-tolerance, which prevents the immune system from attacking cells indiscriminately. However, some cancers can protect themselves from attack by stimulating immune checkpoint targets.
An immune checkpoint regulator is a modulator of the immune system, that allows initiation of a productive immune response and prevents the onset of autoimmunity. Examples of such a molecule are cytotoxic T-lymphocyte antigen 4, which is an inhibitory receptor found on immune cells and programmed cell death 1 (CD279), which has an important role in down-regulating the immune system by preventing the activation of T-cells.

Emactuzumab (RG-7155) is a humanized monoclonal antibody directed against colony stimulating factor 1 receptor (CSF-1R) expressed on macrophages and has demonstrated a profound antitumor effect through interference with the CSF-1/CSF-1R axis, along with a manageable safety profile in patients with diffuse-type tenosynovial giant cell tumors (d-TGCT).
APC Activators are a type of immunotherapy which leverages antigen-presenting cells (APCs) to drive an adaptive immune response. APC Activators are agonists to APC surface-expressed ligands that, when bound, induce the maturation and activation of APCs. Professional antigen-presenting cells – including dendritic cells, macrophages, and B cells – serve an indispensable role in the adaptive immune response through their unique ability to phagocytose, digest, and present exogenous (circulating) antigens to T cells, facilitating antigen-specific immune responses.
References
- ↑ "Study to Determine Safety and Dose of NJH395 in Non-breast HER2+ Advanced Cancer". Smart Patients. 2018. Retrieved 9 June 2019.
Immune stimulator antibody conjugate (ISAC), consisting of a monoclonal antibody which targets HER2 conjugated to an immune-stimulatory agent.
- ↑ Vinluan F (5 February 2019). "Bolt Bio Bags $54M for Drugs that Turn the Heat Up on "Cold" Tumors". Xconomy . Retrieved 9 June 2019.
The company's therapies consist of a tumor-targeting antibody that homes in on tumors and is paired with an immune stimulant that turns cold tumors into hot ones. In preclinical testing, the company says that these immune-stimulating antibody conjugates eliminated tumors.
- 1 2 Grover N (1 April 2019). "AACR: Bay Area biotech bets on antibodies armed with immuno-stimulant to fight checkpoint-resistant cancers". Endpoints News. Retrieved 9 June 2019.
The researcher, Stanford's Ed Engleman, has built on his research into dendritic cells — which are considered 'sentinels' of the immune system as they are responsible for inducing immune T-cell responses — to develop this Immune-Stimulating Antibody Conjugate (ISAC) technology, which was unveiled by exclusive licensee Bolt Biotherapeutics at the American Association for Cancer Research (AACR) Conference on Monday. "What Bolt has come up with is to wake up dendritic cells within the (tumor) microenvironment, and we were able to do this in a targeted way," said David Dornan, senior VP of research, in an interview with Endpoints News ahead of the conference. Many patients are refractory to checkpoint inhibitors because there are a number of immunosuppressive factors present in their tumor microenvironment, and so researchers have been trying to harness different molecules to stimulate the immune system, one of which are toll-like receptor (TLR) agonists — specialized proteins that initiate an immune response to foreign pathogens or, in this case, cancer cells. But the challenge of delivering these adjuvants is that they must be delivered intratumorally, because if they were administered systemically — say orally or intravenously — they can become toxic as immune cells across the body are activated and the impact is not targeted, Dornan emphasized.
- ↑ "Week In Review: China Resources And Charoen Pokphand Join To Launch $300 Million Life Science Fund". Seeking Alpha. 10 February 2019. Retrieved 9 June 2019.
Bolt is developing systemic immune system treatments to provide immunity-based therapies for cancer. The company will use the funds to advance its lead Boltbody™ Immune-Stimulating Antibody Conjugate (ISAC) into the clinic.
- ↑ Carmi Y, Spitzer MH, Linde IL, Burt BM, Prestwood TR, Perlman N, et al. (May 2015). "Allogeneic IgG combined with dendritic cell stimuli induce antitumour T-cell immunity". Nature. 521 (7550): 99–104. Bibcode:2015Natur.521...99C. doi:10.1038/nature14424. PMC 4877172 . PMID 25924063.
- ↑ Ackerman SE, Pearson CI, Gregorio JD, Gonzalez JC, Kenkel JA, Hartmann FJ, et al. (January 2021). "Immune-stimulating antibody conjugates elicit robust myeloid activation and durable antitumor immunity". Nature Cancer. 2 (1): 18–33. doi:10.1038/s43018-020-00136-x. PMC 9012298 . PMID 35121890.