Related Research Articles
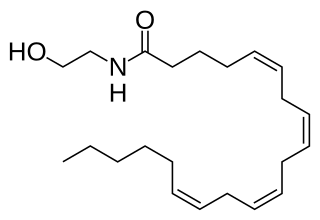
Anandamide (ANA), also known as N-arachidonoylethanolamine (AEA), an N-acylethanolamine (NAE), is a fatty acid neurotransmitter. Anandamide was the first endocannabinoid to be discovered: it participates in the body's endocannabinoid system by binding to cannabinoid receptors, the same receptors that the psychoactive compound THC in cannabis acts on. Anandamide is found in nearly all tissues in a wide range of animals. Anandamide has also been found in plants, including small amounts in chocolate. The name 'anandamide' is taken from the Sanskrit word ananda, which means "joy, bliss, delight", plus amide.

Cannabinoids are several structural classes of compounds found in the cannabis plant primarily and most animal organisms or as synthetic compounds. The most notable cannabinoid is the phytocannabinoid tetrahydrocannabinol (THC) (delta-9-THC), the primary psychoactive compound in cannabis. Cannabidiol (CBD) is also a major constituent of temperate cannabis plants and a minor constituent in tropical varieties. At least 113 distinct phytocannabinoids have been isolated from cannabis, although only four have been demonstrated to have a biogenetic origin. It was reported in 2020 that phytocannabinoids can be found in other plants such as rhododendron, licorice and liverwort, and earlier in Echinacea.

Cannabinoid receptors, located throughout the body, are part of the endocannabinoid system of vertebrates– a class of cell membrane receptors in the G protein-coupled receptor superfamily. As is typical of G protein-coupled receptors, the cannabinoid receptors contain seven transmembrane spanning domains. Cannabinoid receptors are activated by three major groups of ligands:

CP 55,940 is a synthetic cannabinoid which mimics the effects of naturally occurring THC. CP 55,940 was created by Pfizer in 1974 but was never marketed. It is currently used as a research tool to study the endocannabinoid system.
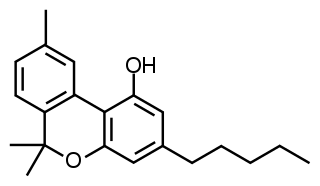
Cannabinol (CBN) is a mildly psychoactive cannabinoid that acts as a low affinity partial agonist at both CB1 and CB2 receptors. This activity at CB1 and CB2 receptors constitutes interaction of CBN with the endocannabinoid system (ECS).

Cannabinoid receptor 1 (CB1), is a G protein-coupled cannabinoid receptor that in humans is encoded by the CNR1 gene. The human CB1 receptor is expressed in the peripheral nervous system and central nervous system. It is activated by endogenous cannabinoids called endocannabinoids, a group of retrograde neurotransmitters that include lipids, such as anandamide and 2-arachidonoylglycerol (2-AG); plant phytocannabinoids, such as docosatetraenoylethanolamide found in wild daga, the compound THC which is an active constituent of the psychoactive drug cannabis; and synthetic analogs of THC. CB1 is antagonized by the phytocannabinoid tetrahydrocannabivarin (THCV).

The cannabinoid receptor 2(CB2), is a G protein-coupled receptor from the cannabinoid receptor family that in humans is encoded by the CNR2 gene. It is closely related to the cannabinoid receptor 1 (CB1), which is largely responsible for the efficacy of endocannabinoid-mediated presynaptic-inhibition, the psychoactive properties of tetrahydrocannabinol (THC), the active agent in cannabis, and other phytocannabinoids. The principal endogenous ligand for the CB2 receptor is 2-Arachidonoylglycerol (2-AG).
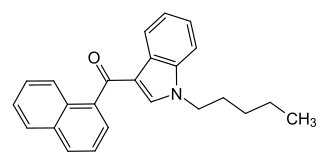
JWH-018 (1-pentyl-3-(1-naphthoyl)indole, NA-PIMO or AM-678) is an analgesic chemical from the naphthoylindole family that acts as a full agonist at both the CB1 and CB2 cannabinoid receptors, with some selectivity for CB2. It produces effects in animals similar to those of tetrahydrocannabinol (THC), a cannabinoid naturally present in cannabis, leading to its use in synthetic cannabis products that in some countries are sold legally as "incense blends".

Synthetic cannabinoids are a class of designer drug molecules that bind to the same receptors to which cannabinoids in cannabis plants attach. These novel psychoactive substances should not be confused with synthetic phytocannabinoids or synthetic endocannabinoids from which they are in many aspects distinct.
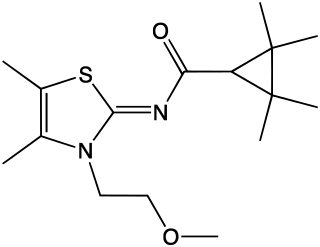
A-836,339 is a drug developed by Abbott Laboratories that acts as a potent cannabinoid receptor full agonist. It is selective for CB2, with Ki values of 0.64 nM at CB2 vs 270 nM at the psychoactive CB1 receptor, but while it exhibits selective analgesic, anti-inflammatory and anti-hyperalgesic effects at low doses, its high efficacy at both targets results in typical cannabis-like effects appearing at higher doses, despite its low binding affinity for CB1. In 2012 A-836,339 was detected via X-ray crystallography in a "dubious product" sold in Japan, though the product was described as a white powder, not herbal incense, it was suggested to be for human consumption.

AB-001 (1-pentyl-3-(1-adamantoyl)indole) is a designer drug that was found as an ingredient in synthetic cannabis smoking blends in Ireland in 2010 and Hungary and Germany in 2011. It is unclear who AB-001 was originally developed by, but it is structurally related to compounds such as AM-1248 and its corresponding 1-(tetrahydropyran-4-ylmethyl) analogue, which are known to be potent cannabinoid agonists with moderate to a high selectivity for CB2 over CB1. The first published synthesis and pharmacological evaluation of AB-001 revealed that it acts as a full agonist at CB1 (EC50 = 35 nM) and CB2 receptors (EC50 = 48 nM). However, AB-001 was found to possess only weak cannabimimetic effects in rats at doses up to 30 mg/kg, making it less potent than the carboxamide analogue APICA, which possesses potent cannabimimetic activity at doses of 3 mg/kg.

UR-144 (TMCP-018, KM-X1, MN-001, YX-17) is a drug invented by Abbott Laboratories, that acts as a selective full agonist of the peripheral cannabinoid receptor CB2, but with much lower affinity for the psychoactive CB1 receptor.

APICA is an indole based drug that acts as a potent agonist for the cannabinoid receptors.

ADBICA (also known as ADB-PICA) is a designer drug identified in synthetic cannabis blends in Japan in 2013. ADBICA had not previously been reported in the scientific literature prior to its sale as a component of synthetic cannabis blends. ADBICA features a carboxamide group at the 3-indole position, like SDB-001 and STS-135. The stereochemistry of the tert-butyl side-chain in the product is unresolved, though in a large series of indazole derivatives structurally similar to ADBICA that are disclosed in Pfizer patent WO 2009/106980, activity resides exclusively in the (S) enantiomers. ADBICA is a potent agonist of the CB1 receptor and CB2 receptor with an EC50 value of 0.69 nM and 1.8 nM respectively.

SDB-006 is a drug that acts as a potent agonist for the cannabinoid receptors, with an EC50 of 19 nM for human CB2 receptors, and 134 nM for human CB1 receptors. It was discovered during research into the related compound SDB-001 which had been sold illicitly as "2NE1". SDB-006 metabolism has been described in literature.

AMB-FUBINACA (also known as FUB-AMB and MMB-FUBINACA) is an indazole-based synthetic cannabinoid that is a potent agonist for the cannabinoid receptors, with Ki values of 10.04 nM at CB1 and 0.786 nM at CB2 and EC50 values of 0.5433 nM at CB1 and 0.1278 nM at CB2, and has been sold online as a designer drug. It was originally developed by Pfizer which described the compound in a patent in 2009, but was later abandoned and never tested on humans. AMB-FUBINACA was the most common synthetic cannabinoid identified in drug seizures by the Drug Enforcement Administration in 2017 and the first half of 2018.

MDMB-4en-PINACA is an indazole-based synthetic cannabinoid that has been sold online as a designer drug. MDMB-4en-PINACA was first identified in Europe in 2017. In 2021, MDMB-4en-PINACA was the most common synthetic cannabinoid identified by the Drug Enforcement Administration in the United States. MDMB-4en-PINACA differs from 5F-MDMB-PINACA due to replacement of 5-fluoropentyl with a pent-4-ene moiety (4-en).

CUMYL-FUBINACA (SGT-149) is an indazole-3-carboxamide based synthetic cannabinoid receptor agonist, with an EC50 of 1.8nM for human CB1 receptors and 23.7nM for human CB2 receptors, giving it around 13x selectivity for CB1. It has been sold online as a designer drug.

ADB-BINACA (also known as ADMB-BZINACA using EMCDDA naming standards) is a cannabinoid designer drug that has been found as an ingredient in some synthetic cannabis products. It was originally developed by Pfizer as a potential analgesic, and is a potent agonist of the CB1 receptor with a binding affinity (Ki) of 0.33 nM and an EC50 of 14.7 nM.
Synthetic drugs refer to substances that are artificially modified from naturally-occurring drugs and are capable of exhibiting both therapeutic and psychoactive effects.
References
- 1 2 "Staff profile at the Department of Pharmacology & Toxicology".
- ↑ "Professor Michelle Glass - The University of Auckland". unidirectory.auckland.ac.nz.
- ↑ "Researchers split $53m fund". Stuff. 5 October 2011.
- ↑ "$157,000 to study cannabinoids' effect on brain cancer". 20 July 2016.
- ↑ "Demystifying synthetic cannabis". 10 August 2018.
- ↑ "Killer synthetic drug was never tested on humans". Newshub. 16 September 2017.
- ↑ "Why is synthetic cannabis killing Kiwis more than anyone else in the world?". TVNZ.
- ↑ simon.collins@nzherald.co.nz, Simon Collins Education reporter, NZ Herald (29 July 2017). "Minister and drug expert: 'Tell us what's in killer synthetic cannabis'" – via www.nzherald.co.nz.
{{cite web}}: CS1 maint: multiple names: authors list (link) - ↑ "Med student's heartfelt medicinal marijuana documentary Mum, Cannabis and Me premieres". Newshub. 8 September 2017.
- ↑ "Mum, Cannabis and Me documentary premier challenges medicinal cannabis use". Stuff. 7 September 2017.
- ↑ "Mum, cannabis and me to screen tomorrow".
- ↑ "Educational Docco World Premiere - 'Mum, Cannabis, And Me' - Scoop News". www.scoop.co.nz.
- ↑ "Royal Society Te Apārangi - Saving kauri with a human drug discovery approach". royalsociety.org.nz. Retrieved 18 January 2019.