Related Research Articles

Chemical vapor deposition (CVD) is a vacuum deposition method used to produce high-quality, and high-performance, solid materials. The process is often used in the semiconductor industry to produce thin films.
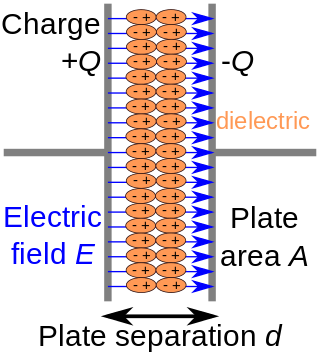
In electromagnetism, a dielectric is an electrical insulator that can be polarised by an applied electric field. When a dielectric material is placed in an electric field, electric charges do not flow through the material as they do in an electrical conductor, because they have no loosely bound, or free, electrons that may drift through the material, but instead they shift, only slightly, from their average equilibrium positions, causing dielectric polarisation. Because of dielectric polarisation, positive charges are displaced in the direction of the field and negative charges shift in the direction opposite to the field. This creates an internal electric field that reduces the overall field within the dielectric itself. If a dielectric is composed of weakly bonded molecules, those molecules not only become polarised, but also reorient so that their symmetry axes align to the field.

A microwave oven is an electric oven that heats and cooks food by exposing it to electromagnetic radiation in the microwave frequency range. This induces polar molecules in the food to rotate and produce thermal energy in a process known as dielectric heating. Microwave ovens heat foods quickly and efficiently because excitation is fairly uniform in the outer 25–38 mm(1–1.5 inches) of a homogeneous, high-water-content food item.

Sintering or frittage is the process of compacting and forming a solid mass of material by pressure or heat without melting it to the point of liquefaction. Sintering happens as part of a manufacturing process used with metals, ceramics, plastics, and other materials. The nanoparticles in the sintered material diffuse across the boundaries of the particles, fusing the particles together and creating a solid piece.

Rotational spectroscopy is concerned with the measurement of the energies of transitions between quantized rotational states of molecules in the gas phase. The spectra of polar molecules can be measured in absorption or emission by microwave spectroscopy or by far infrared spectroscopy. The rotational spectra of non-polar molecules cannot be observed by those methods, but can be observed and measured by Raman spectroscopy. Rotational spectroscopy is sometimes referred to as pure rotational spectroscopy to distinguish it from rotational-vibrational spectroscopy where changes in rotational energy occur together with changes in vibrational energy, and also from ro-vibronic spectroscopy where rotational, vibrational and electronic energy changes occur simultaneously.
Dynamic nuclear polarization (DNP) results from transferring spin polarization from electrons to nuclei, thereby aligning the nuclear spins to the extent that electron spins are aligned. Note that the alignment of electron spins at a given magnetic field and temperature is described by the Boltzmann distribution under the thermal equilibrium. It is also possible that those electrons are aligned to a higher degree of order by other preparations of electron spin order such as: chemical reactions, optical pumping and spin injection. DNP is considered one of several techniques for hyperpolarization. DNP can also be induced using unpaired electrons produced by radiation damage in solids.
Microwave chemistry is the science of applying microwave radiation to chemical reactions. Microwaves act as high frequency electric fields and will generally heat any material containing mobile electric charges, such as polar molecules in a solvent or conducting ions in a solid. Polar solvents are heated as their component molecules are forced to rotate with the field and lose energy in collisions. Semiconducting and conducting samples heat when ions or electrons within them form an electric current and energy is lost due to the electrical resistance of the material. Microwave heating in the laboratory began to gain wide acceptance following papers in 1986, although the use of microwave heating in chemical modification can be traced back to the 1950s. Although occasionally known by such acronyms as MAOS, MEC or MORE synthesis, these acronyms have had little acceptance outside a small number of groups.

A phase-change material (PCM) is a substance which releases/absorbs sufficient energy at phase transition to provide useful heat or cooling. Generally the transition will be from one of the first two fundamental states of matter - solid and liquid - to the other. The phase transition may also be between non-classical states of matter, such as the conformity of crystals, where the material goes from conforming to one crystalline structure to conforming to another, which may be a higher or lower energy state.
Microwave spectroscopy is the spectroscopy method that employs microwaves, i.e. electromagnetic radiation at GHz frequencies, for the study of matter.

Dielectric heating, also known as electronic heating, radio frequency heating, and high-frequency heating, is the process in which a radio frequency (RF) alternating electric field, or radio wave or microwave electromagnetic radiation heats a dielectric material. At higher frequencies, this heating is caused by molecular dipole rotation within the dielectric.

Pentacene is a polycyclic aromatic hydrocarbon consisting of five linearly-fused benzene rings. This highly conjugated compound is an organic semiconductor. The compound generates excitons upon absorption of ultra-violet (UV) or visible light; this makes it very sensitive to oxidation. For this reason, this compound, which is a purple powder, slowly degrades upon exposure to air and light.
In flow chemistry, also called reactor engineering, a chemical reaction is run in a continuously flowing stream rather than in batch production. In other words, pumps move fluid into a reactor, and where tubes join one another, the fluids contact one another. If these fluids are reactive, a reaction takes place. Flow chemistry is a well-established technique for use at a large scale when manufacturing large quantities of a given material. However, the term has only been coined recently for its application on a laboratory scale by chemists and describes small pilot plants, and lab-scale continuous plants. Often, microreactors are used.
A nonthermal plasma, cold plasma or non-equilibrium plasma is a plasma which is not in thermodynamic equilibrium, because the electron temperature is much hotter than the temperature of heavy species. As only electrons are thermalized, their Maxwell-Boltzmann velocity distribution is very different from the ion velocity distribution. When one of the velocities of a species does not follow a Maxwell-Boltzmann distribution, the plasma is said to be non-Maxwellian.

Hexafluorophosphate is an anion with chemical formula of [PF6]−. It is an octahedral species that imparts no color to its salts. [PF6]− is isoelectronic with sulfur hexafluoride, SF6, and the hexafluorosilicate dianion, [SiF6]2−, and hexafluoroantimonate [SbF6]−. In this anion, phosphorus has a valence of 5. Being poorly nucleophilic, hexafluorophosphate is classified as a non-coordinating anion.

Ethylene carbonate (sometimes abbreviated EC) is the organic compound with the formula (CH2O)2CO. It is classified as the cyclic carbonate ester of ethylene glycol and carbonic acid. At room temperature (25 °C) ethylene carbonate is a transparent crystalline solid, practically odorless and colorless, and somewhat soluble in water. In the liquid state (m.p. 34-37 °C) it is a colorless odorless liquid.

Superheated water is liquid water under pressure at temperatures between the usual boiling point, 100 °C (212 °F) and the critical temperature, 374 °C (705 °F). It is also known as "subcritical water" or "pressurized hot water". Superheated water is stable because of overpressure that raises the boiling point, or by heating it in a sealed vessel with a headspace, where the liquid water is in equilibrium with vapour at the saturated vapor pressure. This is distinct from the use of the term superheating to refer to water at atmospheric pressure above its normal boiling point, which has not boiled due to a lack of nucleation sites.

Ferroelectric polymers are a group of crystalline polar polymers that are also ferroelectric, meaning that they maintain a permanent electric polarization that can be reversed, or switched, in an external electric field.
In chemistry, solvent effects are the influence of a solvent on chemical reactivity or molecular associations. Solvents can have an effect on solubility, stability and reaction rates and choosing the appropriate solvent allows for thermodynamic and kinetic control over a chemical reaction.
Microwave volumetric heating (MVH) is a method of using microwaves to evenly heat the entire volume of a flowing liquid, suspension or semi-solid. The process is known as MVH because the microwaves penetrate uniformly throughout the volume of the product being heated, thus delivering energy evenly into the body of the material.
Radio-frequency welding, also known as dielectric welding and high-frequency welding, is a plastic welding process that utilizes high-frequency electric fields to induce heating and melting of thermoplastic base materials. The electric field is applied by a pair of electrodes after the parts being joined are clamped together. The clamping force is maintained until the joint solidifies. Advantages of this process are fast cycle times, automation, repeatability, and good weld appearance. Only plastics which have dipoles can be heated using radio waves and therefore not all plastics are able to be welded using this process. Also, this process is not well suited for thick or overly complex joints. The most common use of this process is lap joints or seals on thin plastic sheets or parts.
References
- ↑ Stuerga, D.; Gaillard, P. Journal of Microwave Power and Electromagnetic Energy , 1996, 31, 101-113. http://jmpee.org/JMPEE_temp/31-2_bl/JMPEEA-31-2-Pg101.htm
- ↑ Stuerga, D.; Gaillard, P. Journal of Microwave Power and Electromagnetic Energy, 1996, 31, 87-99. http://jmpee.org/JMPEE_temp/31-2_bl/JMPEEA-31-2-Pg87.htm
- ↑ Microwaves in organic synthesis. Thermal and non-thermal microwave effects, Antonio de la Hoz, Angel Diaz-Ortiz, Andres Moreno, Chem. Soc. Rev., 2005, 164-178. doi : 10.1039/B411438H
- ↑ Strong Acceleration of Chemical Reactions Occurring Through the Effects of Rotational Excitation on Collisional Geometry, Adolf Miklavc, ChemPhysChem, 2001, 552-555. doi : 10.1002/1439-7641(20010917)2:8/9<552::AID-CPHC552>3.0.CO;2-5
- ↑ Whittaker, A.G., Chem. Mater., 17 (13), 3426 -3432, 2005.
- ↑ Booske, J. H.; Cooper, R. F.; Dobson, I. Journal of Materials Research 1992, 7, 495-501.
- ↑ Booske, J. H.; Cooper, R. F.; Freeman, S. A. Materials Research Innovations 1997, 1, 77-84.
- ↑ Freeman, S. A.; Booske, J. H.; Cooper, R. F. J. Appl. Phys., 1998, 83, 5761.
- ↑ Robb, G.; Harrison, A.; Whittaker, A. G. Phys. Chem. Comm., 2002, 135-137
- ↑ Kappe, C. O.; Pieber, B.; Dallinger, D. (2013). "Microwave Effects in Organic Synthesis: Myth or Reality?". Angew. Chem. Int. Ed. 52 (4): 1088–1094. doi:10.1002/anie.201204103.
- ↑ Dudley, G. B.; Richert, R.; Stiegman, A. E. (2015). "On the existence of and mechanism for microwave-specific reaction rate enhancement". Chem. Sci. 6 (4): 2144. doi:10.1039/c4sc03372h. PMC 5639434 . PMID 29308138.