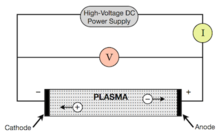
An electric current is a flow of charged particles, such as electrons or ions, moving through an electrical conductor or space. It is defined as the net rate of flow of electric charge through a surface. The moving particles are called charge carriers, which may be one of several types of particles, depending on the conductor. In electric circuits the charge carriers are often electrons moving through a wire. In semiconductors they can be electrons or holes. In an electrolyte the charge carriers are ions, while in plasma, an ionized gas, they are ions and electrons.

A fusor is a device that uses an electric field to heat ions to a temperature at which they undergo nuclear fusion. The machine induces a potential difference between two metal cages, inside a vacuum. Positive ions fall down this voltage drop, building up speed. If they collide in the center, they can fuse. This is one kind of an inertial electrostatic confinement device – a branch of fusion research.
A Langmuir probe is a device used to determine the electron temperature, electron density, and electric potential of a plasma. It works by inserting one or more electrodes into a plasma, with a constant or time-varying electric potential between the various electrodes or between them and the surrounding vessel. The measured currents and potentials in this system allow the determination of the physical properties of the plasma.

A corona discharge is an electrical discharge caused by the ionization of a fluid such as air surrounding a conductor carrying a high voltage. It represents a local region where the air has undergone electrical breakdown and become conductive, allowing charge to continuously leak off the conductor into the air. A corona discharge occurs at locations where the strength of the electric field around a conductor exceeds the dielectric strength of the air. It is often seen as a bluish glow in the air adjacent to pointed metal conductors carrying high voltages, and emits light by the same mechanism as a gas discharge lamp, chemiluminescence. Corona discharges can also happen in weather, such as thunderstorms, where objects like ship masts or airplane wings have a charge significantly different from the air around them.

An ion source is a device that creates atomic and molecular ions. Ion sources are used to form ions for mass spectrometers, optical emission spectrometers, particle accelerators, ion implanters and ion engines.

Inertial electrostatic confinement, or IEC, is a class of fusion power devices that use electric fields to confine the plasma rather than the more common approach using magnetic fields found in magnetic confinement fusion (MCF) designs. Most IEC devices directly accelerate their fuel to fusion conditions, thereby avoiding energy losses seen during the longer heating stages of MCF devices. In theory, this makes them more suitable for using alternative aneutronic fusion fuels, which offer a number of major practical benefits and makes IEC devices one of the more widely studied approaches to fusion.
Plasma diagnostics are a pool of methods, instruments, and experimental techniques used to measure properties of a plasma, such as plasma components' density, distribution function over energy (temperature), their spatial profiles and dynamics, which enable to derive plasma parameters.

An inductively coupled plasma (ICP) or transformer coupled plasma (TCP) is a type of plasma source in which the energy is supplied by electric currents which are produced by electromagnetic induction, that is, by time-varying magnetic fields.

An electric arc is an electrical breakdown of a gas that produces a prolonged electrical discharge. The current through a normally nonconductive medium such as air produces a plasma, which may produce visible light. An arc discharge is initiated either by thermionic emission or by field emission. After initiation, the arc relies on thermionic emission of electrons from the electrodes supporting the arc. An arc discharge is characterized by a lower voltage than a glow discharge. An archaic term is voltaic arc, as used in the phrase "voltaic arc lamp".
In physics, the Saha ionization equation is an expression that relates the ionization state of a gas in thermal equilibrium to the temperature and pressure. The equation is a result of combining ideas of quantum mechanics and statistical mechanics and is used to explain the spectral classification of stars. The expression was developed by physicist Meghnad Saha in 1920. It is discussed in many textbooks on statistical physics and plasma physics.

The Madison Symmetric Torus (MST) is a reversed field pinch (RFP) physics experiment with applications to both fusion energy research and astrophysical plasmas.
The diffusion of plasma across a magnetic field was conjectured to follow the Bohm diffusion scaling as indicated from the early plasma experiments of very lossy machines. This predicted that the rate of diffusion was linear with temperature and inversely linear with the strength of the confining magnetic field.
Neutral-beam injection (NBI) is one method used to heat plasma inside a fusion device consisting in a beam of high-energy neutral particles that can enter the magnetic confinement field. When these neutral particles are ionized by collision with the plasma particles, they are kept in the plasma by the confining magnetic field and can transfer most of their energy by further collisions with the plasma. By tangential injection in the torus, neutral beams also provide momentum to the plasma and current drive, one essential feature for long pulses of burning plasmas. Neutral-beam injection is a flexible and reliable technique, which has been the main heating system on a large variety of fusion devices. To date, all NBI systems were based on positive precursor ion beams. In the 1990s there has been impressive progress in negative ion sources and accelerators with the construction of multi-megawatt negative-ion-based NBI systems at LHD (H0, 180 keV) and JT-60U (D0, 500 keV). The NBI designed for ITER is a substantial challenge (D0, 1 MeV, 40 A) and a prototype is being constructed to optimize its performance in view of the ITER future operations. Other ways to heat plasma for nuclear fusion include RF heating, electron cyclotron resonance heating (ECRH), ion cyclotron resonance heating (ICRH), and lower hybrid resonance heating (LH).
The polywell is a proposed design for a fusion reactor using an electric and magnetic field to heat ions to fusion conditions.
In plasma physics, the Hasegawa–Mima equation, named after Akira Hasegawa and Kunioki Mima, is an equation that describes a certain regime of plasma, where the time scales are very fast, and the distance scale in the direction of the magnetic field is long. In particular the equation is useful for describing turbulence in some tokamaks. The equation was introduced in Hasegawa and Mima's paper submitted in 1977 to Physics of Fluids, where they compared it to the results of the ATC tokamak.
A double layer is a structure in a plasma consisting of two parallel layers of opposite electrical charge. The sheets of charge, which are not necessarily planar, produce localised excursions of electric potential, resulting in a relatively strong electric field between the layers and weaker but more extensive compensating fields outside, which restore the global potential. Ions and electrons within the double layer are accelerated, decelerated, or deflected by the electric field, depending on their direction of motion.
A microplasma is a plasma of small dimensions, ranging from tens to thousands of micrometers. Microplasmas can be generated at a variety of temperatures and pressures, existing as either thermal or non-thermal plasmas. Non-thermal microplasmas that can maintain their state at standard temperatures and pressures are readily available and accessible to scientists as they can be easily sustained and manipulated under standard conditions. Therefore, they can be employed for commercial, industrial, and medical applications, giving rise to the evolving field of microplasmas.
The electrothermal instability is a magnetohydrodynamic (MHD) instability appearing in magnetized non-thermal plasmas used in MHD converters. It was first theoretically discovered in 1962 and experimentally measured into a MHD generator in 1963 by Evgeny Velikhov.
"This paper shows that it is possible to assert sufficiently specifically that the ionization instability is the number one problem for the utilization of a plasma with hot electrons."

In electromagnetism, a streamer discharge, also known as filamentary discharge, is a type of transient electric discharge which forms at the surface of a conductive electrode carrying a high voltage in an insulating medium such as air. Streamers are luminous writhing branching sparks, plasma channels composed of ionized air molecules, which repeatedly strike out from the electrode into the air.
Plasmalysis is a electrochemical process that requires a voltage source. On the one hand, it describes the plasma-chemical dissociation of organic and inorganic compounds in interaction with a thermal/non-thermal plasma between two electrodes. On the other hand, it describes the synthesis, i.e. the combination of two or more elements to form a new molecule. Plasmalysis is an artificial word made of plasma and lysis.