Related Research Articles
Molecular biology is a branch of biology that seeks to understand the molecular basis of biological activity in and between cells, including biomolecular synthesis, modification, mechanisms, and interactions.

Electroporation, or electropermeabilization, is a technique in which an electrical field is applied to cells in order to increase the permeability of the cell membrane. This may allow chemicals, drugs, electrode arrays or DNA to be introduced into the cell.

In molecular biology and genetics, transformation is the genetic alteration of a cell resulting from the direct uptake and incorporation of exogenous genetic material from its surroundings through the cell membrane(s). For transformation to take place, the recipient bacterium must be in a state of competence, which might occur in nature as a time-limited response to environmental conditions such as starvation and cell density, and may also be induced in a laboratory.
Transfection is the process of deliberately introducing naked or purified nucleic acids into eukaryotic cells. It may also refer to other methods and cell types, although other terms are often preferred: "transformation" is typically used to describe non-viral DNA transfer in bacteria and non-animal eukaryotic cells, including plant cells. In animal cells, transfection is the preferred term as transformation is also used to refer to progression to a cancerous state (carcinogenesis) in these cells. Transduction is often used to describe virus-mediated gene transfer into eukaryotic cells.

A Morpholino, also known as a Morpholino oligomer and as a phosphorodiamidate Morpholino oligomer (PMO), is a type of oligomer molecule used in molecular biology to modify gene expression. Its molecular structure contains DNA bases attached to a backbone of methylenemorpholine rings linked through phosphorodiamidate groups. Morpholinos block access of other molecules to small specific sequences of the base-pairing surfaces of ribonucleic acid (RNA). Morpholinos are used as research tools for reverse genetics by knocking down gene function.
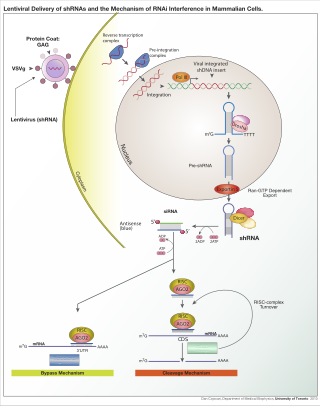
A short hairpin RNA or small hairpin RNA is an artificial RNA molecule with a tight hairpin turn that can be used to silence target gene expression via RNA interference (RNAi). Expression of shRNA in cells is typically accomplished by delivery of plasmids or through viral or bacterial vectors. shRNA is an advantageous mediator of RNAi in that it has a relatively low rate of degradation and turnover. However, it requires use of an expression vector, which has the potential to cause side effects in medicinal applications.
Viral vectors are tools commonly used by molecular biologists to deliver genetic material into cells. This process can be performed inside a living organism or in cell culture. Viruses have evolved specialized molecular mechanisms to efficiently transport their genomes inside the cells they infect. Delivery of genes or other genetic material by a vector is termed transduction and the infected cells are described as transduced. Molecular biologists first harnessed this machinery in the 1970s. Paul Berg used a modified SV40 virus containing DNA from the bacteriophage λ to infect monkey kidney cells maintained in culture.

Exogenous DNA is DNA originating outside the organism of concern or study. Exogenous DNA can be found naturally in the form of partially degraded fragments left over from dead cells. These DNA fragments may then become integrated into the chromosomes of nearby bacterial cells to undergo mutagenesis. This process of altering bacteria is known as transformation. Bacteria may also undergo artificial transformation through chemical and biological processes. The introduction of exogenous DNA into eukaryotic cells is known as transfection. Exogenous DNA can also be artificially inserted into the genome, which revolutionized the process of genetic modification in animals. By microinjecting an artificial transgene into the nucleus of an animal embryo, the exogenous DNA is allowed to merge the cell's existing DNA to create a genetically modified, transgenic animal. The creation of transgenic animals also leads into the study of altering sperm cells with exogenous DNA.

Gene delivery is the process of introducing foreign genetic material, such as DNA or RNA, into host cells. Gene delivery must reach the genome of the host cell to induce gene expression. Successful gene delivery requires the foreign gene delivery to remain stable within the host cell and can either integrate into the genome or replicate independently of it. This requires foreign DNA to be synthesized as part of a vector, which is designed to enter the desired host cell and deliver the transgene to that cell's genome. Vectors utilized as the method for gene delivery can be divided into two categories, recombinant viruses and synthetic vectors.
Lipofectamine or Lipofectamine 2000 is a common transfection reagent, produced and sold by Invitrogen, used in molecular and cellular biology. It is used to increase the transfection efficiency of RNA or plasmid DNA into in vitro cell cultures by lipofection. Lipofectamine contains lipid subunits that can form liposomes in an aqueous environment, which entrap the transfection payload, e.g. DNA plasmids.
Magnetofection is a transfection method that uses magnetic fields to concentrate particles containing vectors to target cells in the body. Magnetofection has been adapted to a variety of vectors, including nucleic acids, non-viral transfection systems, and viruses. This method offers advantages such as high transfection efficiency and biocompatibility which are balanced with limitations.
Reverse transfection is a technique for the transfer of genetic material into cells. As DNA is printed on a glass slide for the transfection process to occur before the addition of adherent cells, the order of addition of DNA and adherent cells is reverse that of conventional transfection. Hence, the word “reverse” is used.
Magnet-assisted transfection is a transfection method which uses magnetic interactions to deliver DNA into target cells. Nucleic acids are associated with magnetic nanoparticles, and magnetic fields drive the nucleic acid-particle complexes into target cells, where the nucleic acids are released.
Mirus Bio LLC, develops and manufactures transfection reagents, electroporation solutions and related products for life science research.

Gene therapy utilizes the delivery of DNA into cells, which can be accomplished by several methods, summarized below. The two major classes of methods are those that use recombinant viruses and those that use naked DNA or DNA complexes.

Genetic engineering techniques allow the modification of animal and plant genomes. Techniques have been devised to insert, delete, and modify DNA at multiple levels, ranging from a specific base pair in a specific gene to entire genes. There are a number of steps that are followed before a genetically modified organism (GMO) is created. Genetic engineers must first choose what gene they wish to insert, modify, or delete. The gene must then be isolated and incorporated, along with other genetic elements, into a suitable vector. This vector is then used to insert the gene into the host genome, creating a transgenic or edited organism.
Tissue nanotransfection (TNT) is an electroporation-based technique capable of gene and drug cargo delivery or transfection at the nanoscale. Furthermore, TNT is a scaffold-less tissue engineering (TE) technique that can be considered cell-only or tissue inducing depending on cellular or tissue level applications. The transfection method makes use of nanochannels to deliver cargo to tissues topically.
Transient expression, more frequently referred to "transient gene expression", is the temporary expression of genes that are expressed for a short time after nucleic acid, most frequently plasmid DNA encoding an expression cassette, has been introduced into eukaryotic cells with a chemical delivery agent like calcium phosphate (CaPi) or polyethyleneimine (PEI). However, unlike "stable expression," the foreign DNA does not fuse with the host cell DNA, resulting in the inevitable loss of the vector after several cell replication cycles. The majority of transient gene expressions are done with cultivated animal cells. The technique is also used in plant cells; however, the transfer of nucleic acids into these cells requires different methods than those with animal cells. In both plants and animals, transient expression should result in a time-limited use of transferred nucleic acids, since any long-term expression would be called "stable expression."
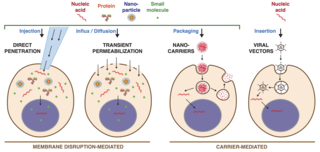
Intracellular delivery is the process of introducing external materials into living cells. Materials that are delivered into cells include nucleic acids, proteins, peptides, impermeable small molecules, synthetic nanomaterials, organelles, and micron-scale tracers, devices and objects. Such molecules and materials can be used to investigate cellular behavior, engineer cell operations or correct a pathological function.

Hydrodynamic Delivery (HD) is a method of DNA insertion in rodent models. Genes are delivered via injection into the bloodstream of the animal, and are expressed in the liver. This protocol is helpful to determine gene function, regulate gene expression, and develop pharmaceuticals in vivo.
References
- Kerima Maasho; Alina Marusina; Nicole M. Reynolds; John E. Coligan; Francisco Borrego (January 2004). "Efficient gene transfer into the human natural killer cell line, NKL, using the amaxa nucleofection system". Journal of Immunological Methods. 284 (1–2): 133–140. doi:10.1016/j.jim.2003.10.010. PMID 14736423.
- Pascal G. Leclere; Aliza Panjwani; Reginald Docherty; Martin Berry; John Pizzey; David A. Tonge (15 Mar 2005). "Effective gene delivery to adult neurons by a modified form of electroporation". Journal of Neuroscience Methods. 142 (1): 137–143. doi:10.1016/j.jneumeth.2004.08.012. PMID 15652627. S2CID 38463232.
- Michela Aluigi; Miriam Fogli; Antonio Curti; Alessandro Isidori; Elisa Gruppioni; Claudia Chiodoni; Mario P. Colombo; Piera Versura; Antonia D'Errico-Grigioni; Elisa Ferri; Michele Baccarani; Roberto M. Lemoli (February 2006). "Nucleofection is an efficient non-viral transfection technique for human bone marrow-derived mesenchymal stem cells". Stem Cells. 24 (2): 454–461. doi: 10.1634/stemcells.2005-0198 . PMID 16099993.