| Look up permeability , impermeability , impermeable , permeable , or semipermeable in Wiktionary, the free dictionary. |
Permeability, permeable, and semipermeable may refer to:
| Look up permeability , impermeability , impermeable , permeable , or semipermeable in Wiktionary, the free dictionary. |
Permeability, permeable, and semipermeable may refer to:
| | This disambiguation page lists articles associated with the title Permeability. If an internal link led you here, you may wish to change the link to point directly to the intended article. |

Osmotic pressure is the minimum pressure which needs to be applied to a solution to prevent the inward flow of its pure solvent across a semipermeable membrane. It is also defined as the measure of the tendency of a solution to take in pure solvent by osmosis. Potential osmotic pressure is the maximum osmotic pressure that could develop in a solution if it were separated from its pure solvent by a semipermeable membrane.

A vacuum is space devoid of matter. The word stems from the Latin adjective vacuus for "vacant" or "void". An approximation to such vacuum is a region with a gaseous pressure much less than atmospheric pressure. Physicists often discuss ideal test results that would occur in a perfect vacuum, which they sometimes simply call "vacuum" or free space, and use the term partial vacuum to refer to an actual imperfect vacuum as one might have in a laboratory or in space. In engineering and applied physics on the other hand, vacuum refers to any space in which the pressure is considerably lower than atmospheric pressure. The Latin term in vacuo is used to describe an object that is surrounded by a vacuum.

In medicine, dialysis (from Greek is the process of removing excess water, solutes, and toxins from the blood in people whose kidneys can no longer perform these functions naturally. This is referred to as renal replacement therapy. The first successful dialysis was performed in 1943.

A capillary is a small blood vessel from 5 to 10 micrometres (μm) in diameter, and having a wall one endothelial cell thick. They are the smallest blood vessels in the body: they convey blood between the arterioles and venules. These microvessels are the site of exchange of many substances with the interstitial fluid surrounding them. Substances which exit include water, oxygen, and glucose; substances which enter include water, carbon dioxide, uric acid, lactic acid, urea and creatinine. Lymph capillaries connect with larger lymph vessels to drain lymphatic fluid collected in the microcirculation.

Semipermeable membrane is a type of biological or synthetic, polymeric membrane that will allow certain molecules or ions to pass through it by osmosis—or occasionally by more specialized processes of facilitated diffusion, passive transport or active transport. The rate of passage depends on the pressure, concentration, and temperature of the molecules or solutes on either side, as well as the permeability of the membrane to each solute. Depending on the membrane and the solute, permeability may depend on solute size, solubility, properties, or chemistry. How the membrane is constructed to be selective in its permeability will determine the rate and the permeability. Many natural and synthetic materials thicker than a membrane are also semipermeable. One example of this is the thin film on the inside of the egg. Note that a semipermeable membrane is not the same as a selectively permeable membrane. Semipermeable membrane describes a membrane that allows some particles to pass through, whereas the selectively permeable membrane will choose what passes through.
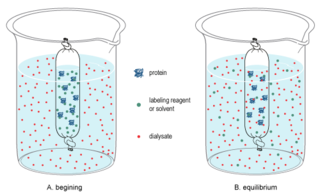
In biochemistry, dialysis is the process of separating molecules in solution by the difference in their rates of diffusion through a semipermeable membrane, such as dialysis tubing.
In cellular biology, membrane transport refers to the collection of mechanisms that regulate the passage of solutes such as ions and small molecules through biological membranes, which are lipid bilayers that contain proteins embedded in them. The regulation of passage through the membrane is due to selective membrane permeability - a characteristic of biological membranes which allows them to separate substances of distinct chemical nature. In other words, they can be permeable to certain substances but not to others.
Permeability in fluid mechanics and the Earth sciences is a measure of the ability of a porous material to allow fluids to pass through it.
In physics and engineering, a constitutive equation or constitutive relation is a relation between two physical quantities that is specific to a material or substance, and approximates the response of that material to external stimuli, usually as applied fields or forces. They are combined with other equations governing physical laws to solve physical problems; for example in fluid mechanics the flow of a fluid in a pipe, in solid state physics the response of a crystal to an electric field, or in structural analysis, the connection between applied stresses or forces to strains or deformations.
Membrane-introduction mass spectrometry (MIMS) is a method of introducing analytes into the mass spectrometer's vacuum chamber via a semipermeable membrane. Usually a thin, gas-permeable, hydrophobic membrane is used, for example polydimethylsiloxane. Samples can be almost any fluid including water, air or sometimes even solvents. The great advantage of the method of sample introduction is its simplicity. MIMS can be used to measure a variety of analytes in real-time, with little or no sample preparation. MIMS is most useful for the measurement of small, non-polar molecules, since molecules of this type have a greater affinity for the membrane material than the sample.
Vacuum permeability is the magnetic permeability in a classical vacuum. Vacuum permeability is derived from production of a magnetic field by an electric current or by a moving electric charge and in all other formulas for magnetic-field production in a vacuum. Since the redefinition of SI units in 2019, the vacuum permeability μ0 is no longer a defined constant, but rather needs to be determined experimentally.
In physics and engineering, permeation is the penetration of a permeate through a solid. It is directly related to the concentration gradient of the permeate, a material's intrinsic permeability, and the materials' mass diffusivity. Permeation is modeled by equations such as Fick's laws of diffusion, and can be measured using tools such as a minipermeameter.

In renal physiology, ultrafiltration occurs at the barrier between the blood and the filtrate in the glomerular capsule in the kidneys. As in nonbiological examples of ultrafiltration, pressure and concentration gradients lead to a separation through a semipermeable membrane. The Bowman's capsule contains a dense capillary network called the glomerulus. Blood flows into these capillaries through the afferent arterioles and leaves through the efferent arterioles.

The following outline is provided as an overview of and topical guide to cell biology:

Osmosis is the spontaneous net movement of solvent molecules through a selectively permeable membrane into a region of higher solute concentration, in the direction that tends to equalize the solute concentrations on the two sides. It may also be used to describe a physical process in which any solvent moves across a selectively permeable membrane separating two solutions of different concentrations. Osmosis can be made to do work. Osmotic pressure is defined as the external pressure required to be applied so that there is no net movement of solvent across the membrane. Osmotic pressure is a colligative property, meaning that the osmotic pressure depends on the molar concentration of the solute but not on its identity.
Cell theory has its origins in seventeenth century microscopy observations, but it was nearly two hundred years before a complete cell membrane theory was developed to explain what separates cells from the outside world. By the 19th century it was accepted that some form of semi-permeable barrier must exist around a cell. Studies of the action of anesthetic molecules led to the theory that this barrier might be made of some sort of fat (lipid), but the structure was still unknown. A series of pioneering experiments in 1925 indicated that this barrier membrane consisted of two molecular layers of lipids—a lipid bilayer. New tools over the next few decades confirmed this theory, but controversy remained regarding the role of proteins in the cell membrane. Eventually the fluid mosaic model was composed in which proteins “float” in a fluid lipid bilayer "sea". Although simplistic and incomplete, this model is still widely referenced today.
Nuclear magnetic resonance (NMR) in porous materials covers the application of using NMR as a tool to study the structure of porous media and various processes occurring in them. This technique allows the determination of characteristics such as the porosity and pore size distribution, the permeability, the water saturation, the wettability, etc.

Permeability is a property of foundry sand with respect to how well the sand can vent, i.e. how well gases pass through the sand. And in other words, permeability is the property by which we can know the ability of material to transmit fluid/gases. The permeability is commonly tested to see if it is correct for the casting conditions.

The cell membrane is a biological membrane that separates the interior of all cells from the outside environment which protects the cell from its environment. The cell membrane consists of a lipid bilayer, including cholesterols that sit between phospholipids to maintain their fluidity at various temperatures. The membrane also contains membrane proteins, including integral proteins that go across the membrane serving as membrane transporters, and peripheral proteins that loosely attach to the outer (peripheral) side of the cell membrane, acting as enzymes shaping the cell. The cell membrane controls the movement of substances in and out of cells and organelles. In this way, it is selectively permeable to ions and organic molecules. In addition, cell membranes are involved in a variety of cellular processes such as cell adhesion, ion conductivity and cell signalling and serve as the attachment surface for several extracellular structures, including the cell wall, the carbohydrate layer called the glycocalyx, and the intracellular network of protein fibers called the cytoskeleton. In the field of synthetic biology, cell membranes can be artificially reassembled.

A membrane osmometer is a device used to indirectly measure the number average molecular weight of a polymer sample. One chamber contains pure solvent and the other chamber contains a solution in which the solute is a polymer with an unknown . The osmotic pressure of the solvent across the semipermeable membrane is measured by the membrane osmometer. This osmotic pressure measurement is used to calculate for the sample.