Related Research Articles

Ion channels are pore-forming membrane proteins that allow ions to pass through the channel pore. Their functions include establishing a resting membrane potential, shaping action potentials and other electrical signals by gating the flow of ions across the cell membrane, controlling the flow of ions across secretory and epithelial cells, and regulating cell volume. Ion channels are present in the membranes of all cells. Ion channels are one of the two classes of ionophoric proteins, the other being ion transporters.
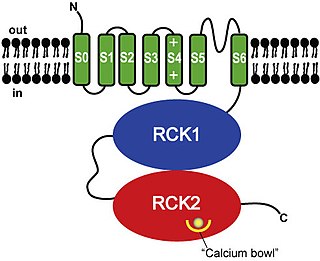
BK channels (big potassium), are large conductance calcium-activated potassium channels, also known as Maxi-K, slo1, or Kca1.1. BK channels are voltage-gated potassium channels that conduct large amounts of potassium ions (K+) across the cell membrane, hence their name, big potassium. These channels can be activated (opened) by either electrical means, or by increasing Ca2+ concentrations in the cell. BK channels help regulate physiological processes, such as circadian behavioral rhythms and neuronal excitability. BK channels are also involved in many processes in the body, as it is a ubiquitous channel. They have a tetrameric structure that is composed of a transmembrane domain, voltage sensing domain, potassium channel domain, and a cytoplasmic C-terminal domain, with many X-ray structures for reference. Their function is to repolarize the membrane potential by allowing for potassium to flow outward, in response to a depolarization or increase in calcium levels.
Sodium channels are integral membrane proteins that form ion channels, conducting sodium ions (Na+) through a cell's membrane. They belong to the superfamily of cation channels and can be classified according to the trigger that opens the channel for such ions, i.e. either a voltage-change ("voltage-gated", "voltage-sensitive", or "voltage-dependent" sodium channel; also called "VGSCs" or "Nav channel") or a binding of a substance (a ligand) to the channel (ligand-gated sodium channels).

Phoneutria nigriventer is a species of medically significant spider in the family Ctenidae, found in South America. Along with other members of the genus, they are often referred to as Brazilian wandering spiders.

Delta atracotoxin is a low-molecular-weight neurotoxic polypeptide found in the venom of the Sydney funnel-web spider.
The P-type calcium channel is a type of voltage-dependent calcium channel. Similar to many other high-voltage-gated calcium channels, the α1 subunit determines most of the channel's properties. The 'P' signifies cerebellar Purkinje cells, referring to the channel's initial site of discovery. P-type calcium channels play a similar role to the N-type calcium channel in neurotransmitter release at the presynaptic terminal and in neuronal integration in many neuronal types.

Agatoxins are a class of chemically diverse polyamine and peptide toxins which are isolated from the venom of various spiders. Their mechanism of action includes blockade of glutamate-gated ion channels, voltage-gated sodium channels, or voltage-dependent calcium channels. Agatoxin is named after the funnel web spider which produces a venom containing several agatoxins. There are different agatoxins. The ω-agatoxins are approximately 100 amino acids in length and are antagonists of voltage-sensitive calcium channels and also block the release of neurotransmitters. For instance, the ω-agatoxin 1A is a selective blocker and will block L-type calcium channels whereas the ω-agatoxin 4B will inhibit voltage sensitive P-type calcium channels. The μ-agatoxins only act on insect voltage-gated sodium channels.

Potassium voltage-gated channel, Shab-related subfamily, member 1, also known as KCNB1 or Kv2.1, is a protein that, in humans, is encoded by the KCNB1 gene.
Philanthotoxins are components of the venom of the Egyptian solitary wasp Philanthus triangulum, commonly known as the European beewolf. Philanthotoxins are polyamine toxins, a group of toxins isolated from the venom of wasps and spiders which immediately but reversibly paralyze their prey. δ-philanthotoxin, also known as PhTX-433, is the most active philanthotoxin that can be refined from the venom. PhTX-433 functions by non-selectively blocking excitatory neurotransmitter ion channels, including nicotinic acetylcholine receptors (nAChRs) and ionotropic glutamate receptors (iGluRs). Synthetic analogues, including PhTX-343 and PhTX-12, have been developed to improve selectivity. While the IC50 values of philanthotoxins varies between analogues and receptor subunit composition, the IC50 value of PhTX-433 at the iGluR AMPA receptor naturally expressed in locust leg muscle is 18 μM and the IC50 value at rat nAChRs is 1 μM.
Phoneutria nigriventer toxin-3 is more commonly referred to as PhTx3.
Phaiodotoxin (PhTx1) is a toxin from the venom of Anuroctonus phaiodactylus, also known as the Mafia scorpion. It affects voltage-gated sodium ion channels leading to an increased duration of its opening.
Jingzhaotoxin proteins are part of a venom secreted by Chilobrachys jingzhao, the Chinese tarantula. and act as neurotoxins. There are several subtypes of jingzhaotoxin, which differ in terms of channel selectivity and modification characteristics. All subspecies act as gating modifiers of sodium channels and/or, to a lesser extent, potassium channels.

Guangxitoxin, also known as GxTX, is a peptide toxin found in the venom of the tarantula Plesiophrictus guangxiensis. It primarily inhibits outward voltage-gated Kv2.1 potassium channel currents, which are prominently expressed in pancreatic β-cells, thus increasing insulin secretion.
Hanatoxin is a toxin found in the venom of the Grammostola spatulata tarantula. The toxin is mostly known for inhibiting the activation of voltage-gated potassium channels, most specifically Kv4.2 and Kv2.1, by raising its activation threshold.
Hainantoxins (HNTX) are neurotoxins from the venom of the Chinese bird spider Haplopelma hainanum. Hainantoxins specifically inhibit tetrodotoxin-sensitive Voltage-gated sodium channels, thereby causing blockage of neuromuscular transmission and paralysis. Currently, 13 different hainantoxins are known, but only HNTX-I, -II, -III, -IV and -V have been investigated in detail.
Huwentoxins (HWTX) are a group of neurotoxic peptides found in the venom of the Chinese bird spider Haplopelma schmidti. The species was formerly known as Haplopelma huwenum, Ornithoctonus huwena and Selenocosmia huwena. While structural similarity can be found among several of these toxins, HWTX as a group possess high functional diversity.
HsTx1 is a toxin from the venom of the scorpion Heterometrus spinifer. HsTx1 is a very potent inhibitor of the rat Kv1.3 voltage-gated potassium channel.
GTx1-15 is a toxin from the Chilean tarantula venom that acts as both a voltage-gated calcium channel blocker and a voltage-gated sodium channel blocker.
μ-THTX-Cl6a, also known as Cl6a, is a 33-residue peptide toxin extracted from the venom of the spider Cyriopagopus longipes. The toxin acts as an inhibitor of the tetrodotoxin-sensitive (TTX-S) voltage-gated sodium channel (NaV1.7), thereby causing sustained reduction of NaV1.7 currents.
Protoxin-I, also known as ProTx-I, or Beta/omega-theraphotoxin-Tp1a, is a 35-amino-acid peptide neurotoxin extracted from the venom of the tarantula Thrixopelma pruriens. Protoxin-I belongs to the inhibitory cystine knot (ICK) family of peptide toxins, which have been known to potently inhibit voltage-gated ion channels. Protoxin-I selectively blocks low voltage threshold T-type calcium channels., voltage gated sodium channels and the nociceptor cation channel TRPA1. Due to its unique ability to bind to TRPA1, Protoxin-I has been implicated as a valuable pharmacological reagent with potential applications in clinical contexts with regards to pain and inflammation
References
- ↑ Fernanber (2013-04-30). TOXINOLOGIA CLINICA. LESIONES POR PICADURAS Y MORDEDURAS DE ANIMALES (in Spanish). Bubok. ISBN 978-84-686-3691-7.
- ↑ Weiss, Marcelo Barros; Paiva, Jorge Wilson Souza (2018-10-10). Acidentes com Animais Peçonhentos (in Brazilian Portuguese). Thieme Revinter Publicações LTDA. ISBN 978-85-5465-084-1.