Related Research Articles
An intermolecular force (IMF) is the force that mediates interaction between molecules, including the electromagnetic forces of attraction or repulsion which act between atoms and other types of neighbouring particles, e.g. atoms or ions. Intermolecular forces are weak relative to intramolecular forces – the forces which hold a molecule together. For example, the covalent bond, involving sharing electron pairs between atoms, is much stronger than the forces present between neighboring molecules. Both sets of forces are essential parts of force fields frequently used in molecular mechanics.

The enthalpy of vaporization, also known as the (latent) heat of vaporization or heat of evaporation, is the amount of energy (enthalpy) that must be added to a liquid substance to transform a quantity of that substance into a gas. The enthalpy of vaporization is a function of the pressure at which that transformation takes place.

The melting point of a substance is the temperature at which it changes state from solid to liquid. At the melting point the solid and liquid phase exist in equilibrium. The melting point of a substance depends on pressure and is usually specified at a standard pressure such as 1 atmosphere or 100 kPa.
The van der Waals radius, rw, of an atom is the radius of an imaginary hard sphere representing the distance of closest approach for another atom. It is named after Johannes Diderik van der Waals, winner of the 1910 Nobel Prize in Physics, as he was the first to recognise that atoms were not simply points and to demonstrate the physical consequences of their size through the van der Waals equation of state.

Magnesium oxide (MgO), or magnesia, is a white hygroscopic solid mineral that occurs naturally as periclase and is a source of magnesium (see also oxide). It has an empirical formula of MgO and consists of a lattice of Mg2+ ions and O2− ions held together by ionic bonding. Magnesium hydroxide forms in the presence of water (MgO + H2O → Mg(OH)2), but it can be reversed by heating it to remove moisture.

Spall are fragments of a material that are broken off a larger solid body. It can be produced by a variety of mechanisms, including as a result of projectile impact, corrosion, weathering, cavitation, or excessive rolling pressure. Spalling and spallation both describe the process of surface failure in which spall is shed.

Fused quartz,fused silica or quartz glass is a glass consisting of almost pure silica (silicon dioxide, SiO2) in amorphous (non-crystalline) form. This differs from all other commercial glasses in which other ingredients are added which change the glasses' optical and physical properties, such as lowering the melt temperature. Fused quartz, therefore, has high working and melting temperatures, making it less desirable for most common applications.

A chondrite is a stony (non-metallic) meteorite that has not been modified, by either melting or differentiation of the parent body. They are formed when various types of dust and small grains in the early Solar System accreted to form primitive asteroids. Some such bodies that are captured in the planet’s gravity well become the most common type of meteorite by arriving on a trajectory toward the planet’s surface. Estimates for their contribution to the total meteorite population vary between 85.7% and 86.2%.
Refractory metals are a class of metals that are extraordinarily resistant to heat and wear. The expression is mostly used in the context of materials science, metallurgy and engineering. The definition of which elements belong to this group differs. The most common definition includes five elements: two of the fifth period and three of the sixth period. They all share some properties, including a melting point above 2000 °C and high hardness at room temperature. They are chemically inert and have a relatively high density. Their high melting points make powder metallurgy the method of choice for fabricating components from these metals. Some of their applications include tools to work metals at high temperatures, wire filaments, casting molds, and chemical reaction vessels in corrosive environments. Partly due to the high melting point, refractory metals are stable against creep deformation to very high temperatures.

A refractory material or refractory is a material that is resistant to decomposition by heat, pressure, or chemical attack, and retains strength and form at high temperatures. Refractories are polycrystalline, polyphase, inorganic, non-metallic, porous, and heterogeneous. They are typically composed of oxides or carbides, nitrides etc. of the following materials: silicon, aluminium, magnesium, calcium, boron, chromium and zirconium.
A refractory is a heat-resistant material, such as:

Investment casting is an industrial process based on lost-wax casting, one of the oldest known metal-forming techniques. The term "lost-wax casting" can also refer to modern investment casting processes.
An intramolecular force is any force that binds together the atoms making up a molecule or compound, not to be confused with intermolecular forces, which are the forces present between molecules. The subtle difference in the name comes from the Latin roots of English with inter meaning between or among and intra meaning inside. Chemical bonds are considered to be intramolecular forces, for example. These forces are often stronger than intermolecular forces, which are present between atoms or molecules that are not bonded.

Material is a substance or mixture of substances that constitutes an object. Materials can be pure or impure, living or non-living matter. Materials can be classified on the basis of their physical and chemical properties, or on their geological origin or biological function. Materials science is the study of materials and their applications.

A molecular solid is a solid consisting of discrete molecules. The cohesive forces that bind the molecules together are van der Waals forces, dipole-dipole interactions, quadrupole interactions, π-π interactions, hydrogen bonding, halogen bonding, London dispersion forces, and in some molecular solids, coulombic interactions. Van der Waals, dipole interactions, quadrupole interactions, π-π interactions, hydrogen bonding, and halogen bonding are typically much weaker than the forces holding together other solids: metallic, ionic, and network solids. Intermolecular interactions, typically do not involve delocalized electrons, unlike metallic and certain covalent bonds. Exceptions are charge-transfer complexes such as the tetrathiafulvane-tetracyanoquinodimethane (TTF-TCNQ), a radical ion salt. These differences in the strength of force and electronic characteristics from other types of solids give rise to the unique mechanical, electronic, and thermal properties of molecular solids.
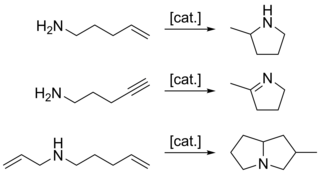
Hydroamination is the addition of an N-H bond of an amine across a carbon-carbon multiple bond of an alkene, alkyne, diene, or allene. In the ideal case, hydroamination is atom economical and green. Amines are common in fine-chemical, pharmaceutical, and agricultural industries. Hydroamination can be used intramolecularly to create heterocycles or intermolecularly with a separate amine and unsaturated compound. The development of catalysts for hydroamination remains an active area, especially for alkenes. Although practical hydroamination reactions can be effected for dienes and electrophilic alkenes, the term hydroamination often implies reactions metal-catalyzed processes.
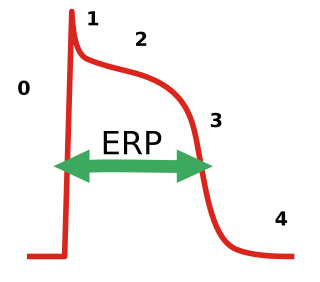
In electrocardiography, during a cardiac cycle, once an action potential is initiated, there is a period of time that a new action potential cannot be initiated. This is termed the effective refractory period (ERP) of the tissue. This period is approximately equal to the absolute refractory period (ARP), it occurs because the fast sodium channels remain closed until the cell fully repolarizes. During this period, depolarization on adjacent cardiac muscles does not produce a new depolarization in the current cell as it has to refract back to phase 4 of the action potential before a new action potential can activate it. ERP acts as a protective mechanism and keeps the heart rate in check and prevents arrhythmias, and it helps coordinates muscle contraction. Anti-arrhythmic agents used for arrhythmias usually prolong the ERP. For the treatment of atrial fibrillation, it is a problem that the prolongation of the ERP by these agents also affects the ventricles, which can induce other types of arrhythmias.
Permanent mold casting is a metal casting process that employs reusable molds, usually made from metal. The most common process uses gravity to fill the mold, however gas pressure or a vacuum are also used. A variation on the typical gravity casting process, called slush casting, produces hollow castings. Common casting metals are aluminium, magnesium, and copper alloys. Other materials include tin, zinc, and lead alloys and iron and steel are also cast in graphite molds.
In chemistry, a metallophilic interaction is defined as a type of non-covalent attraction between heavy metal atoms. The atoms are often within Van der Waals distance of each other and are about as strong as hydrogen bonds. The effect can be intramolecular or intermolecular. Intermolecular metallophilic interactions can lead to formation of supramolecular assemblies whose properties vary with the choice of element and oxidation states of the metal atoms and the attachment of various ligands to them.

Gas is one of the four fundamental states of matter.