Related Research Articles

Adhesive, also known as glue, cement, mucilage, or paste, is any non-metallic substance applied to one or both surfaces of two separate items that binds them together and resists their separation.
SMP may refer to:

In organosilicon and polymer chemistry, a silicone or polysiloxane is a polymer composed of repeating units of siloxane. They are typically colorless oils or rubber-like substances. Silicones are used in sealants, adhesives, lubricants, medicine, cooking utensils, thermal insulation, and electrical insulation. Some common forms include silicone oil, grease, rubber, resin, and caulk.

Polyethylene or polythene (abbreviated PE; IUPAC name polyethene or poly(methylene)) is the most commonly produced plastic. It is a polymer, primarily used for packaging (plastic bags, plastic films, geomembranes and containers including bottles, cups, jars, etc.). As of 2017, over 100 million tonnes of polyethylene resins are being produced annually, accounting for 34% of the total plastics market.

Epoxy is the family of basic components or cured end products of epoxy resins. Epoxy resins, also known as polyepoxides, are a class of reactive prepolymers and polymers which contain epoxide groups. The epoxide functional group is also collectively called epoxy. The IUPAC name for an epoxide group is an oxirane.

Hot-melt adhesive (HMA), also known as hot glue, is a form of thermoplastic adhesive that is commonly sold as solid cylindrical sticks of various diameters designed to be applied using a hot glue gun. The gun uses a continuous-duty heating element to melt the plastic glue, which the user pushes through the gun either with a mechanical trigger mechanism on the gun, or with direct finger pressure. The glue squeezed out of the heated nozzle is initially hot enough to burn and even blister skin. The glue is sticky when hot, and solidifies in a few seconds to one minute. Hot-melt adhesives can also be applied by dipping or spraying, and are popular with hobbyists and crafters both for affixing and as an inexpensive alternative to resin casting.
Silicone rubber is an elastomer composed of silicone—itself a polymer—containing silicon together with carbon, hydrogen, and oxygen. Silicone rubbers are widely used in industry, and there are multiple formulations. Silicone rubbers are often one- or two-part polymers, and may contain fillers to improve properties or reduce cost. Silicone rubber is generally non-reactive, stable, and resistant to extreme environments and temperatures from −55 to 300 °C while still maintaining its useful properties. Due to these properties and its ease of manufacturing and shaping, silicone rubber can be found in a wide variety of products, including voltage line insulators; automotive applications; cooking, baking, and food storage products; apparel such as undergarments, sportswear, and footwear; electronics; medical devices and implants; and in home repair and hardware, in products such as silicone sealants.

Polyester is a category of polymers that contain one or two ester linkages in every repeat unit of their main chain. As a specific material, it most commonly refers to a type called polyethylene terephthalate (PET). Polyesters include some naturally occurring chemicals, such as those found in plants and insects. Natural polyesters and a few synthetic ones are biodegradable, but most synthetic polyesters are not. Synthetic polyesters are used extensively in clothing.
Disilane is a chemical compound with general chemical formula Si2R6 that was first identified in 1902 by Henri Moissan and Samuel Smiles (1877–1953) where R = H. Moissan and Smiles reported disilane as being among the products formed by the action of dilute acids on metal silicides. Although these reactions had been previously investigated by Friedrich Woehler and Heinrich Buff between 1857 and 1858, Moissan and Smiles were the first to explicitly identify disilane. They referred to disilane as silicoethane. Higher members of the homologous series SinH2n+2 formed in these reactions were subsequently identified by Carl Somiesky (sometimes spelled "Karl Somieski") and Alfred Stock.
Sealant is a substance used to block the passage of fluids through openings in materials, a type of mechanical seal. In building construction sealant is sometimes synonymous with caulk and also serve the purposes of blocking dust, sound and heat transmission. Sealants may be weak or strong, flexible or rigid, permanent or temporary. Sealants are not adhesives but some have adhesive qualities and are called adhesive-sealants or structural sealants.
Silanization is the attachment of an organosilyl group to some chemical species. Almost always, silanization is the conversion of a silanol-terminated surface to a alkylsiloxy-terminated surface. This conversion confers hydrophobicity to a previously hydrophilic surface. This process is often used to modify the surface properties of glass, silicon, alumina, quartz, and metal oxide substrates, which all have an abundance of hydroxyl groups. Silanization differs from silylation, which usually refers to attachment of organosilicon groups to molecular substrates.
Dimethyldichlorosilane is a tetrahedral organosilicon compound with the formula Si(CH3)2Cl2. At room temperature it is a colorless liquid that readily reacts with water to form both linear and cyclic Si-O chains. Dimethyldichlorosilane is made on an industrial scale as the principal precursor to dimethylsilicone and polysilane compounds.
RTV silicone is a type of silicone rubber that cures at room temperature. It is available as a one-component product, or mixed from two components. Manufacturers provide it in a range of hardnesses from very soft to medium—usually from 15 to 40 Shore A. RTV silicones can be cured with a catalyst consisting of either platinum or a tin compound such as dibutyltin dilaurate. Applications include low-temperature over-molding, making molds for reproducing, and lens applications for some optically clear grades. It is also used widely in the automotive industry as an adhesive and sealant, for example to create gaskets in place.
Adhesive bonding is a joining technique used in the manufacture and repair of a wide range of products. Along with welding and soldering, adhesive bonding is one of the basic joining processes. In this technique, components are bonded together using adhesives. The broad range of types of adhesives available allows numerous materials to be bonded together in products as diverse as vehicles, mobile phones, personal care products, buildings, computers and medical devices.
Waterborne resins are sometimes called water-based resins. They are resins or polymeric resins that use water as the carrying medium as opposed to solvent or solvent-less. Resins are used in the production of coatings, adhesives, sealants, elastomers and composite materials. When the phrase waterborne resin is used, it usually describes all resins which have water as the main carrying solvent. The resin could be water-soluble, water reducible or water dispersed.
Hydrogenated MDI (H12MDI or 4,4′-diisocyanato dicyclohexylmethane) is an organic compound in the class known as isocyanates. More specifically, it is an aliphatic diisocyanate. It is a water white liquid at room temperature and is manufactured in relatively small quantities. It is also known as 4,4'-methylenedi(cyclohexyl isocyanate) or methylene bis(4-cyclohexylisocyanate) and has the formula CH2[(C6H10)NCO]2.

Castor oil glycidyl ether is a liquid organic chemical in the glycidyl ether family. It is sometimes called castor oil triglycidyl ether. It has the theoretical formula C66H116O12. There are two CAS numbers in use, 14228-73-0 and 74398-71-3. The IUPAC name is 2,3-bis[[(E)-12-(oxiran-2-ylmethoxy)octadec-9-enoyl]oxy]propyl (E)-12-(oxiran-2-ylmethoxy)octadec-9-enoate. A key use is acting as a modifier for epoxy resins as a reactive diluent that adds flexibility and improved mechanical properties.

Trimethylolethane triglycidyl ether (TMETGE) is an organic chemical in the glycidyl ether family. It has the formula C14H24O6 and the IUPAC name is 2-({2-methyl-3-[(oxiran-2-yl)methoxy]-2-{[(oxiran-2-yl)methoxy]methyl}propoxy}methyl)oxirane. The CAS number is 68460-21-9. A key use is as a modifier for epoxy resins as a reactive diluent.
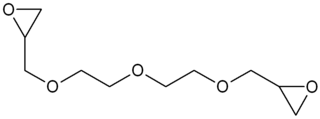
Diethylene glycol diglycidyl ether (DEGDGE) is an organic chemical in the glycidyl ether family with the formula C10H18O5.. The oxirane functionality makes it useful as a reactive diluent for epoxy resin viscosity reduction.
Negative hyperconjugation is a theorized phenomenon in organosilicon compounds, in which hyperconjugation stabilizes or destabilizes certain accumulations of positive charge. The phenomenon explains corresponding peculiarities in the stereochemistry and rate of hydrolysis.
References
- ↑ Jeffrey D. Umpleby "Polymer composition" U.S. patent 4574133. 1986.
- ↑ Michael Ravers. "MS Polymer™-Based PSA technology". Afera.
- ↑ Michael Ravers (2017-12-10). "MS Polymer based PSA Technology (slides)" (PDF). Afera.