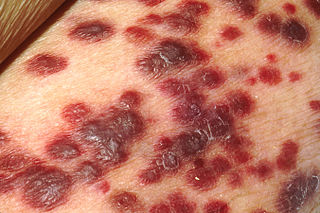
Kaposi's sarcoma-associated herpesvirus (KSHV) is the ninth known human herpesvirus; its formal name according to the International Committee on Taxonomy of Viruses (ICTV) is Human gammaherpesvirus 8, or HHV-8 in short. Like other herpesviruses, its informal names are used interchangeably with its formal ICTV name. This virus causes Kaposi's sarcoma, a cancer commonly occurring in AIDS patients, as well as primary effusion lymphoma, HHV-8-associated multicentric Castleman's disease and KSHV inflammatory cytokine syndrome. It is one of seven currently known human cancer viruses, or oncoviruses. Even after so many years of discovery of KSHV/HHV8, there is no known cure for KSHV associated tumorigenesis.
An oncovirus or oncogenic virus is a virus that can cause cancer. This term originated from studies of acutely transforming retroviruses in the 1950–60s, when the term "oncornaviruses" was used to denote their RNA virus origin. With the letters "RNA" removed, it now refers to any virus with a DNA or RNA genome causing cancer and is synonymous with "tumor virus" or "cancer virus". The vast majority of human and animal viruses do not cause cancer, probably because of longstanding co-evolution between the virus and its host. Oncoviruses have been important not only in epidemiology, but also in investigations of cell cycle control mechanisms such as the retinoblastoma protein.

Nelfinavir, sold under the brand name Viracept, is an antiretroviral medication used in the treatment of HIV/AIDS. Nelfinavir belongs to the class of drugs known as protease inhibitors (PIs) and like other PIs is almost always used in combination with other antiretroviral drugs.
Cancer immunotherapy is the stimulation of the immune system to treat cancer, improving on the immune system's natural ability to fight the disease. It is an application of the fundamental research of cancer immunology and a growing subspeciality of oncology.

Azacitidine, sold under the brand name Vidaza among others, is used for the treatment of myelodysplastic syndrome, myeloid leukemia, and juvenile myelomonocytic leukemia. It is a chemical analog of cytidine, a nucleoside in DNA and RNA. Azacitidine and its deoxy derivative, decitabine were first synthesized in Czechoslovakia as potential chemotherapeutic agents for cancer.

Transcription factor Sp1, also known as specificity protein 1* is a protein that in humans is encoded by the SP1 gene.

Murine respirovirus, formerly Sendai virus (SeV) and previously also known as murine parainfluenza virus type 1 or hemagglutinating virus of Japan (HVJ), is an enveloped,150-200 nm in diameter, a negative sense, single-stranded RNA virus of the family Paramyxoviridae. It typically infects rodents and it is not pathogenic for humans or domestic animals. Sendai virus (SeV) is a member of genus Respirovirus. The virus was isolated in the city of Sendai in Japan in the early 1950s. Since then, it has been actively used in research as a model pathogen. The virus is infectious for many cancer cell lines, has oncolytic properties demonstrated in animal models and in naturally-occurring cancers in animals. SeV's ability to fuse eukaryotic cells and to form syncytium was used to produce hybridoma cells capable of manufacturing monoclonal antibodies in large quantities. Recent applications of SeV-based vectors include the reprogramming of somatic cells into induced pluripotent stem cells and vaccines creation. For vaccination purpose the Sendai virus-based constructs could be delivered in a form of nasal drops, which may be beneficial in inducing a mucosal immune response. SeV has several features that are important in a vector for a successful vaccine: the virus does not integrate into the host genome, it does not undergo genetic recombination, it replicates only in the cytoplasm without DNA intermediates or a nuclear phase and it is not causing any disease in humans or domestic animals. Sendai virus is used as a backbone for vaccine development against Mycobacterium tuberculosis that causes tuberculosis, against HIV-1 that causes AIDS and against other viruses, including those that cause severe respiratory infections in children. The latter include Human Respiratory Syncytial Virus (HRSV), Human Metapneumovirus (HMPV) and Human Parainfluenza Viruses (HPIV). The vaccine studies against Mycobacterium tuberculosis, HMPV, HPIV1 and, HPIV2 are in pre-clinical stage, against HRSV phase I clinical trail has been completed. The phase I clinical studies of SeV-based vaccination were also completed for HPIV1. They were done in adults and in 3- to 6-year-old children. As a result of vaccination against HPIV1 the significant boost in virus-specific neutralizing antibodies was observed. The SeV-based vaccine development against HIV-1 have reached phase II clinical trial. Fudan University in collaboration with ID Pharma Co. Ltd. is engaged in development of the vaccine for COVID-19 prevention. SeV serves as a vaccine backbone vector in the project.
Env is a viral gene that encodes the protein forming the viral envelope. The expression of the env gene enables retroviruses to target and attach to specific cell types, and to infiltrate the target cell membrane.

DNA-directed RNA polymerases I, II, and III subunit RPABC1 is a protein that in humans is encoded by the POLR2E gene.

Nuclear factor of activated T-cells, cytoplasmic 2 is a protein that in humans is encoded by the NFATC2 gene.

Programmed death-ligand 1 (PD-L1) also known as cluster of differentiation 274 (CD274) or B7 homolog 1 (B7-H1) is a protein that in humans is encoded by the CD274 gene.

Y box binding protein 1 also known as Y-box transcription factor or nuclease-sensitive element-binding protein 1 is a protein that in humans is encoded by the YBX1 gene.

Squamous cell carcinoma antigen recognized by T-cells 3 is a protein that in humans is encoded by the SART3 gene.

CREB-regulated transcription coactivator 1 (CRTC1), previously referred to as TORC1, is a protein that in humans is encoded by the CRTC1 gene. It is expressed in a limited number of tissues that include fetal brain and liver and adult heart, skeletal muscles, liver and salivary glands and various regions of the adult central nervous system.

Programmed cell death 1 ligand 2 is a protein that in humans is encoded by the PDCD1LG2 gene. PDCD1LG2 has also been designated as CD273. PDCD1LG2 is an immune checkpoint receptor ligand which plays a role in negative regulation of the adaptive immune response. PD-L2 is one of two known ligands for Programmed cell death protein 1 (PD-1).

Transcription factor IIIA is a protein that in humans is encoded by the GTF3A gene. It was first isolated and characterized by Wolffe and Brown in 1988.
Elliott D. Kieff is the Harriett Ryan Albee Professor of Microbiology and Immunobiology at Harvard Medical School and Brigham and Women’s Hospital. He had previously served as Chair of the Virology Program at Harvard Medical School from 1991 to 2004.
Julianna Lisziewicz is a Hungarian immunologist. Lisziewicz headed many research teams that have discovered and produced immunotheraputic drugs to treat diseases like cancer and chronic infections like HIV/AIDS. Some of these drugs have been successfully used in clinical trials.

Soma Sengupta is a British-American and a specialty board certified neuro-oncologist board certified Neurologist. She is an Associate Professor of Neurology and Rehabilitation Medicine at the University of Cincinnati, with appointments in Cancer Biology and Neurosurgery. She is also affiliated with Cincinnati Children's Hospital Medical Center.
Richard B. Gaynor is an American physician specializing in hematology-oncology, educator, drug developer, and business executive. He served as an Associate Professor of Medicine at UCLA School of Medicine for nearly a decade, and subsequently as an endowed Professor of Medicine and Microbiology at the University of Texas Southwestern Medical School prior to joining the pharmaceutical industry in 2002. His research on NF-κB, IκB kinase, and other mechanisms regulating viral and cellular gene expression has been covered in leading subject reviews. He has been a top executive at several pharmaceutical companies, with respect to the development and clinical testing of novel anticancer drugs and cell therapies. For over a decade and a half, he worked at Eli Lilly and Company, where he became the Senior Vice President of Oncology Clinical Development and Medical Affairs in 2013. Gaynor was President of R&D at Neon Therapeutics from 2016 to 2020, when he became the President of BioNTech US, both pharmaceutical companies headquartered in Cambridge, MA. His honors include being elected a member of the American Society for Clinical Investigation, and the Association of American Physicians.













