
Ultraviolet (UV) light is electromagnetic radiation in a range of energies higher than that of visible light, but less than X-rays. UV radiation is present in sunlight, and constitutes about 10% of the total electromagnetic radiation output from the Sun. It is also produced by electric arcs; Cherenkov radiation; and specialized lights, such as mercury-vapor lamps, tanning lamps, and black lights.

Melanocytes are melanin-producing neural crest-derived cells located in the bottom layer of the skin's epidermis, the middle layer of the eye, the inner ear, vaginal epithelium, meninges, bones, and heart. Melanin is a dark pigment primarily responsible for skin color. Once synthesized, melanin is contained in special organelles called melanosomes which can be transported to nearby keratinocytes to induce pigmentation. Thus darker skin tones have more melanosomes present than lighter skin tones. Functionally, melanin serves as protection against UV radiation. Melanocytes also have a role in the immune system.

Skin cancers are cancers that arise from the skin. They are due to the development of abnormal cells that have the ability to invade or spread to other parts of the body. Skin cancer is the most commonly diagnosed form of cancer in humans. There are three main types of skin cancers: basal-cell skin cancer (BCC), squamous-cell skin cancer (SCC) and melanoma. The first two, along with a number of less common skin cancers, are known as nonmelanoma skin cancer (NMSC). Basal-cell cancer grows slowly and can damage the tissue around it but is unlikely to spread to distant areas or result in death. It often appears as a painless raised area of skin that may be shiny with small blood vessels running over it or may present as a raised area with an ulcer. Squamous-cell skin cancer is more likely to spread. It usually presents as a hard lump with a scaly top but may also form an ulcer. Melanomas are the most aggressive. Signs include a mole that has changed in size, shape, color, has irregular edges, has more than one color, is itchy or bleeds.

Sunscreen, also known as sunblock or sun cream, is a photoprotective topical product for the skin that helps protect against sunburn and most importantly prevent skin cancer. Sunscreens come as lotions, sprays, gels, foams, sticks, powders and other topical products. Sunscreens are common supplements to clothing, particularly sunglasses, sunhats and special sun protective clothing, and other forms of photoprotection.

Sun tanning or tanning is the process whereby skin color is darkened or tanned. It is most often a result of exposure to ultraviolet (UV) radiation from sunlight or from artificial sources, such as a tanning lamp found in indoor tanning beds. People who deliberately tan their skin by exposure to the sun engage in a passive recreational activity of sun bathing. Some people use chemical products which can produce a tanning effect without exposure to ultraviolet radiation, known as sunless tanning.

Indoor tanning involves using a device that emits ultraviolet radiation to produce a cosmetic tan. Typically found in tanning salons, gyms, spas, hotels, and sporting facilities, and less often in private residences, the most common device is a horizontal tanning bed, also known as a sunbed or solarium. Vertical devices are known as tanning booths or stand-up sunbeds.

Sunless tanning, also known as UV filled tanning, self tanning, spray tanning, or fake tanning, refers to the effect of a suntan without exposure to the Sun. Sunless tanning involves the use of oral agents (carotenids), or creams, lotions or sprays applied to the skin. Skin-applied products may be skin-reactive agents or temporary bronzers (colorants).

Psoralen is the parent compound in a family of naturally occurring organic compounds known as the linear furanocoumarins. It is structurally related to coumarin by the addition of a fused furan ring, and may be considered as a derivative of umbelliferone. Psoralen occurs naturally in the seeds of Psoralea corylifolia, as well as in the common fig, celery, parsley, West Indian satinwood, and in all citrus fruits. It is widely used in PUVA treatment for psoriasis, eczema, vitiligo, and cutaneous T-cell lymphoma; these applications are typically through the use of medications such as Methoxsalen. Many furanocoumarins are extremely toxic to fish, and some are deposited in streams in Indonesia to catch fish.
Photolyases are DNA repair enzymes that repair damage caused by exposure to ultraviolet light. These enzymes require visible light both for their own activation and for the actual DNA repair. The DNA repair mechanism involving photolyases is called photoreactivation. They mainly convert pyrimidine dimers into a normal pair of pyrimidine bases.
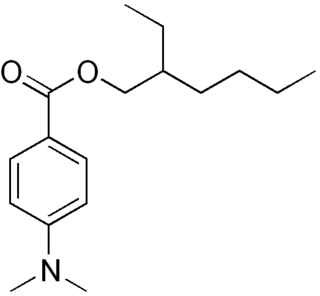
Padimate O is an organic compound related to the water-soluble compound PABA that is used as an ingredient in some sunscreens. This yellowish water-insoluble oily liquid is an ester formed by the condensation of 2-ethylhexanol with dimethylaminobenzoic acid. Other names for padimate O include 2-ethylhexyl 4-dimethylaminobenzoate, Escalol 507, octyldimethyl PABA, and OD-PABA.
UV-induced apoptosis UV-induced apoptosis is an adequate (physiological) reaction of a cell damaged by UV radiation (UVR) in a sufficiently large (lethal) dose and it prevents the disordered destruction of UV damaged cells by help necrosis. Cell elimination by apoptosis occurs when UV-induced cell damage which cannot be repaired by the intracellular repair system exceeds at it certain limit. Through apoptosis, the cells are self-disassembled into compartments with their subsequent utilization. The first time sign of the beginning of the apoptosis system is working in a UV damaged cell is the activation of restriction enzymes, which divide cell DNA into fragments convenient for utilization. But too large a dose of UVR can lead to breakdown (inactivation) of the energy-dependent mechanism of apoptosis. In this case, cell destruction occurs randomly, not orderly, and during a significantly longer time interval. UV-irradiated cells do not change their appearance for a long time [1, 6], as a result of which the researchers may make the erroneous conclusion that “revealed an unexpected response to a dose at which a higher dose of UV increased the viability of keratinocytes” [2]. The fact that UV-induced apoptosis at high doses of UVR begins to be replaced by necrosis was established in 2000 [3]. For keratinocytes, the proportion of cells that have elimination by help apoptosis, with an increase in UVR dose can reach to achieve 45%, but with a further increase in the dose of UVR, destruction of damaged cells by help necrosis and the part of cells that eliminated by apoptosis begins to decrease [4, 11]. In the dose range of UVR from “lethal” to “super-lethal”, “pro-inflammatory” apoptosis can be manifested, which was experimentally discovered in 2003 [5]. This may be the result of partial damage to the apoptosis mechanism by UV radiation [1]. If at moderate doses “pure” apoptosis does not cause an inflammatory reaction, then at sufficiently large doses, an inflammatory reaction arises due to pro-inflammatory apoptosis, which leads to the appearance of “fast” erythema for UV irradiated skin keratinocytes. Kinetic of “fast” erythema is much faster by the time of development of UV erythema caused by necrosis of UV damaged keratinocytes [6]. The most erythemogenic is UVB the spectral range of UVR, since radiation in this range is less absorbed by the outer layers of the skin, which allows UVB radiation, in contrast to UVC, to reach more deep layers skin and act on keratinocytes of the deep-lying basal layer of the epidermis of the skin. The ability to induce apoptosis for UVB and UVC radiation is due to the fact that the DNA of the nucleus [7] and / or mitochondria [8] of the cell absorbs UVR well in the UVC and UVB spectral range. Keratinocytes of the skin are in a state of programmed apoptosis, during which the keratinocytes of the basal layer are removed from it and during the transition through all layers of the epidermis within 28 days turn into flakes of the outer stratum corneum, which are subsequently desquamated. It is clear that the keratinocyte response to UV exposure will depend on what phase of programmed apoptosis the keratinocyte experienced UV exposure, and this is the main reason for the difference of the UV effect for UVC and UVB on the skin. There are also differences in the initiation of mitochondrial (internal) and caspase-dependent (external) apoptosis for the UVC and UVB spectral ranges [9]. Sunburn cells (SBS) are the keratinocytes in the process of UV-induced apoptosis. The appearance of SBC may be not associated with an inflammatory reaction, but the role of UV-induced apoptosis of skin keratinocytes in the development of UV erythema of the skin has been established, which allowed the development of a patent-protected METHOD FOR QUANTITATIVE ASSESSMENT OF APOPTOSIS SYSTEM [10], in which “the brightest lamp of skin display "(photoerythema) is used to diagnose the state of the body systems involved in the elimination of UV-induced damage. Such systems include the immune system, the intracellular repair system, the microcirculation system and not only.

Pyrimidine dimers are molecular lesions formed from thymine or cytosine bases in DNA via photochemical reactions, commonly associated with direct DNA damage. Ultraviolet light induces the formation of covalent linkages between consecutive bases along the nucleotide chain in the vicinity of their carbon–carbon double bonds. The photo-coupled dimers are fluorescent. The dimerization reaction can also occur among pyrimidine bases in dsRNA —uracil or cytosine. Two common UV products are cyclobutane pyrimidine dimers (CPDs) and 6–4 photoproducts. These premutagenic lesions alter the structure of the DNA helix and cause non-canonical base pairing. Specifically, adjacent thymines or cytosines in DNA will form a cyclobutane ring when joined together and cause a distortion in the DNA. This distortion prevents replication or transcription machinery beyond the site of the dimerization. Up to 50–100 such reactions per second might occur in a skin cell during exposure to sunlight, but are usually corrected within seconds by photolyase reactivation or nucleotide excision repair. In humans, the most common form of DNA repair is nucleotide excision repair (NER). In contrast, organisms such as bacteria can counterintuitively harvest energy from the sun to fix DNA damage from pyrimidine dimers via photolyase activity. If these lesions are not fixed, polymerase machinery may misread or add in the incorrect nucleotide to the strand. If the damage to the DNA is overwhelming, mutations can arise within the genome of an organism and may lead to the production of cancer cells. Uncorrected lesions can inhibit polymerases, cause misreading during transcription or replication, or lead to arrest of replication. It causes sunburn and it triggers the production of melanin. Pyrimidine dimers are the primary cause of melanomas in humans.

Trioxsalen (trimethylpsoralen (TMP), trioxysalen (INN) or Trisoralen) is a furanocoumarin and a psoralen derivative. It is obtained from several plants, mainly Psoralea corylifolia. Like other psoralens it causes photosensitization of the skin. It is administered either topically or orally in conjunction with UV-A (the least damaging form of ultraviolet light) for phototherapy treatment of vitiligo and hand eczema. After photoactivation it creates interstrand cross-links in DNA, which can cause programmed cell death unless repaired by cellular mechanisms. In research it can be conjugated to dyes for confocal microscopy and used to visualize sites of DNA damage. The compound is also being explored for development of antisense oligonucleotides that can be cross-linked specifically to a mutant mRNA sequence without affecting normal transcripts differing at even a single base pair.
A photocarcinogen is a substance which causes cancer when an organism is exposed to it, then illuminated. Many chemicals that are not carcinogenic can be photocarcinogenic when combined with exposure to light, especially UV. This can easily be understood from a photochemical perspective: The reactivity of a chemical substance itself might be low, but after illumination it transitions to an excited state, which is chemically much more reactive and therefore potentially harmful to biological tissue and DNA. Light can also split photocarcinogens, releasing free radicals, whose unpaired electrons cause them to be extremely reactive.

Sunburn is a form of radiation burn that affects living tissue, such as skin, that results from an overexposure to ultraviolet (UV) radiation, usually from the Sun. Common symptoms in humans and other animals include red or reddish skin that is hot to the touch or painful, general fatigue, and mild dizziness. Other symptoms include blistering, peeling skin, swelling, itching, and nausea. Excessive UV radiation is the leading cause of (primarily) non-malignant skin tumors, which in extreme cases can be life-threatening. Sunburn is an inflammatory response in the tissue triggered by direct DNA damage by UV radiation. When the cells' DNA is overly damaged by UV radiation, type I cell-death is triggered and the tissue is replaced.

Photoaging or photoageing is a term used for the characteristic changes to skin induced by chronic UVA and UVB exposure. Tretinoin is the best studied retinoid in the treatment of photoaging.

Exposing skin to the ultraviolet radiation in sunlight has both positive and negative health effects. On the positive side, exposure allows for the synthesis of vitamin D3. Vitamin D has been suggested as having a wide range of positive health effects, which include strengthening bones and possibly inhibiting the growth of some cancers. A dietary supplement can also supply vitamin D, but there are also benefits to exposure not obtainable through Vitamin D supplementation. Long-term sun exposure is associated with reduced all-cause mortality and reduced mortality risk from cardiovascular disease (CVD), some forms of cancer, and non-CVD/noncancer related disease, with indications in these studies that Vitamin D is not the mediator. Supplementation offers limited bioavailability and no synthesis of subdermal nitric oxide. UV exposure also has positive effects for endorphin levels, and possibly for protection against multiple sclerosis. Abundant visible light to the eyes gives health benefits through its association with the timing of melatonin synthesis, maintenance of normal and robust circadian rhythms, and reduced risk of seasonal affective disorder.
Animal-type melanoma is a rare subtype of melanoma that is characterized by heavily pigmented dermal epithelioid and spindled melanocytes. Animal-type melanoma is also known to be called equine-type melanoma, pigment synthesizing melanoma, and pigmented epithelioid melanocytoma (PEM). While melanoma is known as the most aggressive skin cancer, the mortality for PEM is lower than in other melanoma types. Animal-type melanoma earned its name due to the resemblance of melanocytic tumors in grey horses.

Angelicin is the parent compound in a family of naturally occurring organic compounds known as the angular furanocoumarins. Structurally, it can be considered as benzapyra-2-one fused with a furan moiety in the 7,8-position. Angelicin is commonly found in certain Apiaceae and Fabaceae plant species such as Bituminaria bituminosa. It has a skin permeability coefficient (LogKp) of -2.46. The maximum absorption is observed at 300 nm. The 1HNMR spectrum is available; the infrared and mass spectra of angelicin can be found in this database. The sublimation of angelicin occurs at 120 °C and the pressure of 0.13 Pa. Angelicin is a coumarin.

The Sunscreen Innovation Act is a 2014 law that amended the Federal Food, Drug, and Cosmetic Act to establish an expedited process for the review and approval of over-the-counter (OTC) sunscreens. The US Food and Drug Administration (FDA) had not approved a new active ingredient in sunscreen since 1999, despite some sunscreens having been approved and used overseas for a decade. The new law gave the FDA one year to respond to the existing backlog of sunscreen ingredient approval requests, and then 18 months to reply to any future applications.
















