Related Research Articles

Calciseptine (CaS) is a natural neurotoxin isolated from the black mamba Dendroaspis p. polylepis venom. This toxin consists of 60 amino acids with four disulfide bonds. Calciseptine specifically blocks L-type calcium channels, but not other voltage-dependent Ca2+ channels such as N-type and T-type channels.
Phoneutria nigriventer toxin-3 is more commonly referred to as PhTx3.
Birtoxin is a neurotoxin from the venom of the South African Spitting scorpion. By changing sodium channel activation, the toxin promotes spontaneous and repetitive firing much like pyrethroid insecticides do
Bestoxin is a neurotoxin from the venom of the South African spitting scorpion Parabuthus transvaalicus. Most likely, it targets sodium channel function, thus promoting spontaneous and repetitive neuronal firing. Following injection into mice, it causes non-lethal writhing behaviour.
BmKAEP is a neurotoxin from the venom of the Manchurian scorpion (Mesobuthus martensii). It is a β-toxin, which shift the activation voltage of sodium channels towards more negative potentials.
Ikitoxin is a neurotoxin from the venom of the South African Spitting scorpion that targets voltage-sensitive sodium channels. It causes unprovoked jumps in mice following intracerebroventricular injections.
Butantoxin (BuTX) is a compound of the venom of three Brazilian and an Argentinean scorpion species of the genus Tityus. Butantoxin reversibly blocks the voltage-gated K+ channels Shaker B and Kv1.2, and the Ca2+-activated K+ channelsKCa 1.1 and KCa 3.1.
BotIT2 is a neurotoxin from the scorpion Buthus occitanus tunetanus, which modifies activation and slows down the deactivation of voltage gated sodium channels.

Bactridines are a group of six antibacterial peptides from the venom of the Tityusdiscrepans scorpion. They exclusively target sodium channels. Bactridines are unique in that this scorpion toxin acts on sodium channels of both bacteria and eukaryotes.
Tamulotoxin is a venomous neurotoxin from the Indian Red Scorpion.
TsIV is a toxin from the venom of the Brazilian scorpion Tityus serrulatus which slows the inactivation of sodium channels.
Ts15 is produced by the Brazilian yellow scorpion Tityus serrulatus. It targets voltage-gated potassium channels, primarily the subtypes Kv1.2 and Kv1.3.
Tityustoxin peptide 2 (TsPep2) is a peptide isolated from the venom of the Tityus serrulatus. It belongs to a class of short peptides, together with Tityustoxin peptide 1 and Tityustoxin peptide 3.

Noxiustoxin (NTX) is a toxin from the venom of the Mexican scorpion Centruroides noxius Hoffmann which block voltage-dependent potassium channels and calcium-activated potassium channels.
Beta-mammal toxin Cn2, also known as Cn2 toxin, is a single chain β-scorpion neurotoxic peptide and the primary toxin in the venom of the Centruroides noxius Hoffmann scorpion. The toxin specifically targets mammalian Nav1.6 voltage-gated sodium channels (VGSC).
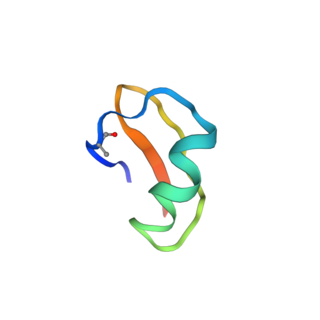
The Tst26 toxin is a voltage-gated potassium channel blocker present in the venom of Tityus stigmurus, a species of Brazilian scorpion. Tst26 selectively blocks Kv1.2 and Kv1.3 channels.
Ts8 is a neurotoxin present in the venom of the Brazilian yellow scorpion, Tityus serrulatus. Ts8 is a selective inhibitor of the voltage-gated potassium channel Kv4.2

Makatoxin-3 is an α-like scorpion neurotoxin found in the venom of Olivierus martensii. Makatoxin-3 both enhances the activation and slows down the inactivation of voltage-gated NaV1.7 channels, resulting in hyperexcitability of the neurons involved in pain perception.
Tb1 is a neurotoxin that is naturally found in the venom of the Brazilian scorpion Tityus bahiensis. Presumably by acting on voltage-gated sodium channels, it triggers excessive glutamate release, which can lead to both behavioral and electrographic epileptiform alterations, as well as neuronal injury.
Tityus stigmurus toxin 1 (Tst1) is a neurotoxin found in the venom of the Brazilian scorpion, Tityus stigmurus. It acts on voltage-gated sodium channels (Navs), altering opening and inactivation voltages, recovery from inactivation, and overall current flow.
References
- 1 2 3 4 5 Corrêa, M.M.; Sampaio, S.V.; Lopes, R.A.; Mancuso, L.C.; Cunha, O.A.B.; Franco, J.J.; Giglio, J.R. (1997). "Biochemical and histopathological alterations induced in rats by Tityus serrulatus scorpion venom and its major neurotoxin tityustoxin-I". Toxicon. 35 (7): 1053–1067. Bibcode:1997Txcn...35.1053C. doi:10.1016/s0041-0101(96)00219-x. PMID 9248004.
- 1 2 3 4 5 Novello, José C; Arantes, Eliane C; Varanda, Wamberto A; Oliveira, Benedito; Giglio, José R; Marangoni, Sérgio (1999). "TsTX-IV, a short chain four-disulfide-bridged neurotoxin from Tityus serrulatus venom which acts on Ca2+-activated K+ channels". Toxicon. 37 (4): 651–660. doi:10.1016/s0041-0101(98)00206-2. PMID 10082164.
- 1 2 3 Clemente, G.T; Rossoni, R.B; Safe, J.M.M; Freire-Maia, L (1999). "Effects of crude venom, tityustoxin and toxin Ts-γ from Tityus serrulatus scorpion on secretion and structure of the rat submandibular gland". Archives of Oral Biology. 44 (2): 103–110. doi:10.1016/s0003-9969(98)00109-5. PMID 10206328.
- 1 2 3 Mesquita, Michel B.S; Moraes-Santos, Tasso; Moraes, M.árcio F.D (2003). "Centrally injected tityustoxin produces the systemic manifestations observed in severe scorpion poisoning". Toxicology and Applied Pharmacology. 187 (1): 58–66. Bibcode:2003ToxAP.187...58M. doi:10.1016/s0041-008x(02)00036-4. PMID 12628585.
- ↑ Werkman, T. R.; Gustafson, T. A.; Rogowski, R. S.; Blaustein, M. P.; Rogawski, M. A. (August 1993). "Tityustoxin-K alpha, a structurally novel and highly potent K+ channel peptide toxin, interacts with the alpha-dendrotoxin binding site on the cloned Kv1.2 K+ channel". Molecular Pharmacology. 44 (2): 430–436. ISSN 0026-895X. PMID 8355670.
- ↑ Legros, Christian; Oughuideni, Razika; Darbon, Hervé; Rochat, Hervé; Bougis, Pierre E.; Martin-Eauclaire, Marie-France (1996-07-15). "Characterization of a new peptide from Tityus serrulatus scorpion venom which is a ligand of the apamin-binding site". FEBS Letters. 390 (1): 81–84. Bibcode:1996FEBSL.390...81L. doi: 10.1016/0014-5793(96)00616-3 . ISSN 1873-3468. PMID 8706835.
- ↑ Conceicao, I. M.; Lebrun, I.; Cano-Abad, M.; Gandia, L.; Hernandez-Guijo, J. M.; Lopez, M. G.; Villarroya, M.; Jurkiewicz, A.; Garcia, A. G. (1998). "Synergism between toxin-gamma from Brazilian scorpion Tityus serrulatus and veratridine in chromaffin cells". The American Journal of Physiology. 274 (6 Pt 1): C1745–1754. doi:10.1152/ajpcell.1998.274.6.C1745. ISSN 0002-9513. PMID 9611141.
- 1 2 Casali, T.A.A.; Gomez, R.S.; Moraes-santos, T.; Gomez, M.V. (1995). "Differential effects of calcium channel antagonists on tityustoxin and ouabain-induced release of [3H]acetylcholine from brain cortical slices". Neuropharmacology. 34 (6): 599–603. doi:10.1016/0028-3908(95)00019-3. PMID 7566495. S2CID 29615707.
- ↑ Pessini, Andréa C; Takao, Tânia T; Cavalheiro, Elisângela C; Vichnewski, Walter; Sampaio, Suely V; Giglio, José R; Arantes, Eliane C (2001). "A hyaluronidase from Tityus serrulatus scorpion venom: isolation, characterization and inhibition by flavonoids". Toxicon. 39 (10): 1495–1504. Bibcode:2001Txcn...39.1495P. doi:10.1016/s0041-0101(01)00122-2. PMID 11478957.
- ↑ Rogowski, R. S.; Krueger, B. K.; Collins, J. H.; Blaustein, M. P. (1994-02-15). "Tityustoxin K alpha blocks voltage-gated noninactivating K+ channels and unblocks inactivating K+ channels blocked by alpha-dendrotoxin in synaptosomes". Proceedings of the National Academy of Sciences. 91 (4): 1475–1479. Bibcode:1994PNAS...91.1475R. doi: 10.1073/pnas.91.4.1475 . ISSN 0027-8424. PMC 43182 . PMID 7509073.
- ↑ Lima, E.G.; Freire-Maia, L. (1977). "Cardiovascular and respiratory effects induced by intracerebroventricular injection of scorpion toxin (tityustoxin) in the rat". Toxicon. 15 (3): 225–234. Bibcode:1977Txcn...15..225L. doi:10.1016/0041-0101(77)90048-4. PMID 867438.
- ↑ Mesquita, Michel Bernanos Soares; Moraes-Santos, Tasso; Moraes, Márcio Flávio Dutra (2002). "Phenobarbital blocks the lung edema induced by centrally injected tityustoxin in adult Wistar rats". Neuroscience Letters. 332 (2): 119–122. doi:10.1016/s0304-3940(02)00932-1. PMID 12384225. S2CID 8617029.
- ↑ Guatimosim, Silvia C.F.; Kalapothakis, Evanguedes; Diniz, Carlos R.; Chávez-Olórtegui, Carlos (2000). "Induction of neutralizing antibodies against Tityus serrulatus toxins by immunization with a recombinant nontoxic protein". Toxicon. 38 (1): 113–121. Bibcode:2000Txcn...38..113G. doi:10.1016/s0041-0101(99)00138-5. PMID 10669016.