
gamma-Hydroxybutyric acid is a naturally occurring neurotransmitter and a depressant drug. It is a precursor to GABA, glutamate, and glycine in certain brain areas. It acts on the GHB receptor and is a weak agonist at the GABAB receptor. GHB has been used in the medical setting as a general anesthetic and as treatment for cataplexy, narcolepsy, and alcoholism. The substance is also used illicitly for various reasons, including as a performance-enhancing drug, date rape drug, and as a recreational drug.

γ-Aminobutyric acid, or GABA, is the chief inhibitory neurotransmitter in the developmentally mature mammalian central nervous system. Its principal role is reducing neuronal excitability throughout the nervous system.
Depressants, or central depressants, are drugs that lower neurotransmission levels, or depress or reduce arousal or stimulation in various areas of the brain. Depressants are also colloquially referred to as "downers" as they lower the level of arousal when taken. Depressants do not change the mood or mental state of others. Stimulants, or "uppers," increase mental or physical function, hence the opposite drug class from depressants are stimulants, not antidepressants.

Butane-1,4-diol, not to be confused with 1,3 butanediol, is a primary alcohol, and an organic compound, with the formula HOCH2CH2CH2CH2OH. It is a colorless viscous liquid. It is one of four stable isomers of butanediol.

The GABAA receptor (GABAAR) is an ionotropic receptor and ligand-gated ion channel. Its endogenous ligand is γ-aminobutyric acid (GABA), the major inhibitory neurotransmitter in the central nervous system. Accurate regulation of GABAergic transmission through appropriate developmental processes, specificity to neural cell types, and responsiveness to activity is crucial for the proper functioning of nearly all aspects of the central nervous system (CNS). Upon opening, the GABAA receptor on the postsynaptic cell is selectively permeable to chloride ions (Cl−) and, to a lesser extent, bicarbonate ions (HCO3−).

Gamma-butyrolactone (GBL) or γ-butyrolactone, is a hygroscopic colorless, water-miscible liquid with a weak characteristic odor. It is the simplest 4-carbon lactone. It is mainly used as an intermediate in the production of other chemicals, such as methyl-2-pyrrolidone.
GABAB receptors (GABABR) are G-protein coupled receptors for gamma-aminobutyric acid (GABA), therefore making them metabotropic receptors, that are linked via G-proteins to potassium channels. The changing potassium concentrations hyperpolarize the cell at the end of an action potential. The reversal potential of the GABAB-mediated IPSP is –100 mV, which is much more hyperpolarized than the GABAA IPSP. GABAB receptors are found in the central nervous system and the autonomic division of the peripheral nervous system.

Gabazine (SR-95531) is a drug that acts as an antagonist at GABAA receptors. It is used in scientific research and has no role in medicine, as it would be expected to produce convulsions if used in humans.

Succinic semialdehyde dehydrogenase deficiency (SSADHD) is a rare autosomal recessive disorder of the degradation pathway of the inhibitory neurotransmitter γ-aminobutyric acid, or GABA. The disorder has been identified in approximately 350 families, with a significant proportion being consanguineous families. The first case was identified in 1981 and published in a Dutch clinical chemistry journal that highlighted a number of neurological conditions such as delayed intellectual, motor, speech, and language as the most common manifestations. Later cases reported in the early 1990s began to show that hypotonia, hyporeflexia, seizures, and a nonprogressive ataxia were frequent clinical features as well.

A GABA receptor agonist is a drug that is an agonist for one or more of the GABA receptors, producing typically sedative effects, and may also cause other effects such as anxiolytic, anticonvulsant, and muscle relaxant effects. There are three receptors of the gamma-aminobutyric acid. The two receptors GABA-α and GABA-ρ are ion channels that are permeable to chloride ions which reduces neuronal excitability. The GABA-β receptor belongs to the class of G-Protein coupled receptors that inhibit adenylyl cyclase, therefore leading to decreased cyclic adenosine monophosphate (cAMP). GABA-α and GABA-ρ receptors produce sedative and hypnotic effects and have anti-convulsion properties. GABA-β receptors also produce sedative effects. Furthermore, they lead to changes in gene transcription.

γ-Amino-β-hydroxybutyric acid (GABOB), also known as β-hydroxy-γ-aminobutyric acid (β-hydroxy-GABA), and sold under the brand name Gamibetal among others, is an anticonvulsant which is used for the treatment of epilepsy in Europe, Japan, and Mexico. It is a GABA analogue, or an analogue of the neurotransmitter γ-aminobutyric acid (GABA), and has been found to be an endogenous metabolite of GABA.

The γ-hydroxybutyrate (GHB) receptor (GHBR), originally identified as GPR172A, is an excitatory G protein-coupled receptor (GPCR) that binds the neurotransmitter and psychoactive drug γ-hydroxybutyric acid (GHB). As solute carrier family 52 member 2 (SLC52A2), it is also a transporter for riboflavin.

Aceburic acid (INN), also known as 4-acetoxybutanoic acid or 4-hydroxybutyric acid acetate, is drug described as an analgesic which was never marketed. It is the acetyl ester of gamma-hydroxybutyrate, and based on its structural relation to GHB, is likely to behave as a prodrug to it.
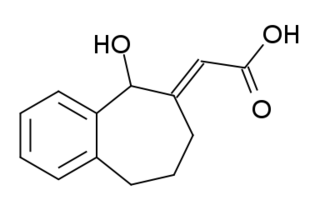
NCS-382 is a moderately selective antagonist for the GHB receptor. It blocks the effects of GHB in animals and has both anti-sedative and anticonvulsant effects. It has been proposed as a treatment for GHB overdose in humans as well as the genetic metabolic disorder succinic semialdehyde dehydrogenase deficiency (SSADHD), but has never been developed for clinical use.
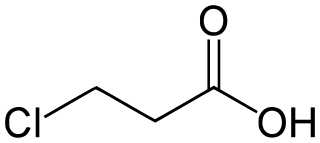
3-Chloropropanoic acid (also 3-chloropropionic acid) is the organic compound with the formula ClCH2CH2CO2H. A white or colorless solid, it is used as a drug and a synthetic intermediate. The compound is produced by the hydrochlorination of acrylic acid.
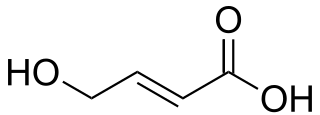
trans-4-Hydroxycrotonic acid (T-HCA), also known as γ-hydroxycrotonic acid (GHC), is an agent used in scientific research to study the GHB receptor. It is an analogue of γ-hydroxybutyric acid (GHB), as well as an active metabolite of GHB. Similarly to GHB, T-HCA has been found to be endogenous to the rat central nervous system, and as a metabolite of GHB, is almost certain to be endogenous to humans as well. T-HCA binds to the high-affinity GHB receptor with 4-fold greater affinity than GHB itself, where it acts as an agonist, but does not bind to the low-affinity GHB binding site, the GABAB receptor. Because of this, T-HCA does not produce sedation. T-HCA has been shown to cause receptor activation-evoked increases in extracellular glutamate concentrations, notably in the hippocampus.

γ-Hydroxyvaleric acid (GHV), also known as 4-methyl-GHB, is a designer drug related to γ-hydroxybutyric acid (GHB). It is sometimes seen on the grey market as a legal alternative to GHB, but with lower potency and higher toxicity, properties which have tended to limit its recreational use.

HA-966 or (±) 3-Amino-1-hydroxy-pyrrolidin-2-one is a molecule used in scientific research as a glycine receptor and NMDA receptor antagonist / low efficacy partial agonist. It has neuroprotective and anticonvulsant, anxiolytic, antinociceptive and sedative / hypnotic effects in animal models. Pilot human clinical trials in the early 1960s showed that HA-966 appeared to benefit patients with tremors of extrapyramidal origin.

Gabapentinoids, also known as α2δ ligands, are a class of drugs that are derivatives of the inhibitory neurotransmitter gamma-Aminobutyric acid (GABA) which block α2δ subunit-containing voltage-dependent calcium channels (VDCCs). This site has been referred to as the gabapentin receptor, as it is the target of the drugs gabapentin and pregabalin.

A GABA analogue is a compound which is an analogue or derivative of the neurotransmitter gamma-Aminobutyric acid (GABA).

















