
Leukemia is a group of blood cancers that usually begin in the bone marrow and result in high numbers of abnormal blood cells. These blood cells are not fully developed and are called blasts or leukemia cells. Symptoms may include bleeding and bruising, bone pain, fatigue, fever, and an increased risk of infections. These symptoms occur due to a lack of normal blood cells. Diagnosis is typically made by blood tests or bone marrow biopsy.
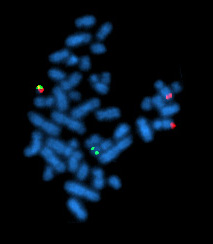
The Philadelphia chromosome or Philadelphia translocation (Ph) is a specific genetic abnormality in chromosome 22 of leukemia cancer cells. This chromosome is defective and unusually short because of reciprocal translocation, t(9;22)(q34;q11), of genetic material between chromosome 9 and chromosome 22, and contains a fusion gene called BCR-ABL1. This gene is the ABL1 gene of chromosome 9 juxtaposed onto the breakpoint cluster region BCR gene of chromosome 22, coding for a hybrid protein: a tyrosine kinase signaling protein that is "always on", causing the cell to divide uncontrollably by interrupting the stability of the genome and impairing various signaling pathways governing the cell cycle.

Chronic myelogenous leukemia (CML), also known as chronic myeloid leukemia, is a cancer of the white blood cells. It is a form of leukemia characterized by the increased and unregulated growth of myeloid cells in the bone marrow and the accumulation of these cells in the blood. CML is a clonal bone marrow stem cell disorder in which a proliferation of mature granulocytes and their precursors is found. It is a type of myeloproliferative neoplasm associated with a characteristic chromosomal translocation called the Philadelphia chromosome.
Tumors of the hematopoietic and lymphoid tissues or tumours of the haematopoietic and lymphoid tissues are tumors that affect the blood, bone marrow, lymph, and lymphatic system. Because these tissues are all intimately connected through both the circulatory system and the immune system, a disease affecting one will often affect the others as well, making aplasia, myeloproliferation and lymphoproliferation closely related and often overlapping problems. While uncommon in solid tumors, chromosomal translocations are a common cause of these diseases. This commonly leads to a different approach in diagnosis and treatment of hematological malignancies. Hematological malignancies are malignant neoplasms ("cancer"), and they are generally treated by specialists in hematology and/or oncology. In some centers "hematology/oncology" is a single subspecialty of internal medicine while in others they are considered separate divisions. Not all hematological disorders are malignant ("cancerous"); these other blood conditions may also be managed by a hematologist.

Acute lymphoblastic leukemia (ALL) is a cancer of the lymphoid line of blood cells characterized by the development of large numbers of immature lymphocytes. Symptoms may include feeling tired, pale skin color, fever, easy bleeding or bruising, enlarged lymph nodes, or bone pain. As an acute leukemia, ALL progresses rapidly and is typically fatal within weeks or months if left untreated.
Lymphoid leukemias are a group of leukemias affecting circulating lymphocytes, a type of white blood cell. The lymphocytic leukemias are closely related to lymphomas of the lymphocytes, to the point that some of them are unitary disease entities that can be called by either name. Such diseases are all lymphoproliferative disorders. Most lymphoid leukemias involve a particular subtype of lymphocytes, the B cells.

Acute myeloid leukemia (AML) is a cancer of the myeloid line of blood cells, characterized by the rapid growth of abnormal cells that build up in the bone marrow and blood and interfere with normal blood cell production. Symptoms may include feeling tired, shortness of breath, easy bruising and bleeding, and increased risk of infection. Occasionally, spread may occur to the brain, skin, or gums. As an acute leukemia, AML progresses rapidly, and is typically fatal within weeks or months if left untreated.
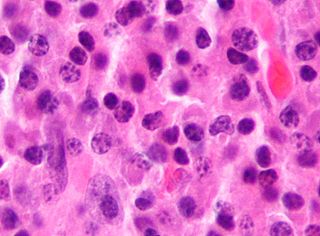
Acute monocytic leukemia is a type of acute myeloid leukemia. In AML-M5 >80% of the leukemic cells are of monocytic lineage. This cancer is characterized by a dominance of monocytes in the bone marrow. There is an overproduction of monocytes that the body does not need in the periphery. These overproduced monocytes interfere with normal immune cell production which causes many health complications for the affected individual.

Chronic myelomonocytic leukemia (CMML) is a type of leukemia, which are cancers of the blood-forming cells of the bone marrow. In adults, blood cells are formed in the bone marrow, by a process that is known as haematopoiesis. In CMML, there are increased numbers of monocytes and immature blood cells (blasts) in the peripheral blood and bone marrow, as well as abnormal looking cells (dysplasia) in at least one type of blood cell.

Acute myeloblastic leukemia with maturation (M2) is a subtype of acute myeloid leukemia (AML).

ETV6 protein is a transcription factor that in humans is encoded by the ETV6 gene. The ETV6 protein regulates the development and growth of diverse cell types, particularly those of hematological tissues. However, its gene, ETV6 frequently suffers various mutations that lead to an array of potentially lethal cancers, i.e., ETV6 is a clinically significant proto-oncogene in that it can fuse with other genes to drive the development and/or progression of certain cancers. However, ETV6 is also an anti-oncogene or tumor suppressor gene in that mutations in it that encode for a truncated and therefore inactive protein are also associated with certain types of cancers.

Histone-lysine N-methyltransferase 2A, also known as acute lymphoblastic leukemia 1 (ALL-1), myeloid/lymphoid or mixed-lineage leukemia1 (MLL1), or zinc finger protein HRX (HRX), is an enzyme that in humans is encoded by the KMT2A gene.
Acute myelomonocytic leukemia (AMML) is a form of acute myeloid leukemia that involves a proliferation of CFU-GM myeloblasts and monoblasts. AMML occurs with a rapid increase amount in white blood cell count and is defined by more than 20% of myeloblast in the bone marrow. It is classified under "M4" in the French-American-British classification (FAB). It is classified under "AML, not otherwise classified" in the WHO classification.

Precursor B-cell lymphoblastic leukemia is a form of lymphoid leukemia in which too many B-cell lymphoblasts are found in the blood and bone marrow. It is the most common type of acute lymphoblastic leukemia (ALL). It is sometimes additionally classified as a lymphoma, as designated leukemia/lymphoma. ALL is the most prevalent childhood malignancy, with precursor B-cell ALL (B-ALL) accounting for approximately 75–80% of newly diagnosed pediatric ALL cases.

Childhood leukemia is leukemia that occurs in a child and is a type of childhood cancer. Childhood leukemia is the most common childhood cancer, accounting for 29% of cancers in children aged 0–14 in 2018. There are multiple forms of leukemia that occur in children, the most common being acute lymphoblastic leukemia (ALL) followed by acute myeloid leukemia (AML). Survival rates vary depending on the type of leukemia, but may be as high as 90% in ALL.
FLAG is a chemotherapy regimen used for relapsed and refractory acute myeloid leukemia (AML). The acronym incorporates the three primary ingredients of the regimen:
- Fludarabine: an antimetabolite that, while not active toward AML, increases formation of an active cytarabine metabolite, ara-CTP, in AML cells;
- Arabinofuranosyl cytidine : an antimetabolite that has been proven to be the most active toward AML among various cytotoxic drugs in single-drug trials; and
- Granulocyte colony-stimulating factor (G-CSF): a glycoprotein that shortens the duration and severity of neutropenia.
AI-10-49 is a small molecule inhibitor of leukemic oncoprotein CBFβ-SMHHC developed by the laboratory of John Bushweller with efficacy demonstrated by the laboratories of Lucio H. Castilla and Monica Guzman. AI-10-49 allosterically binds to CBFβ-SMMHC and disrupts protein-protein interaction between CBFβ-SMMHC and tumor suppressor RUNX1. This inhibitor is under development as an anti-leukemic drug.
Clonal hypereosinophilia, also termed primary hypereosinophilia or clonal eosinophilia, is a grouping of hematological disorders all of which are characterized by the development and growth of a pre-malignant or malignant population of eosinophils, a type of white blood cell that occupies the bone marrow, blood, and other tissues. This population consists of a clone of eosinophils, i.e. a group of genetically identical eosinophils derived from a sufficiently mutated ancestor cell.
Mixed-phenotype acute leukemia (MPAL) is a group of blood cancers (leukemia) which have combined features of myeloid and lymphoid cancers. It is a rare disease, constituting about 2–5% of all leukemia cases. It mostly involve myeloid with either of T lymphocyte or B lymphocyte progenitors, but in rare cases all the three cell lineages. Knowledge on the cause, clinical features and cellular mechanism is poor, making the treatment and management (prognosis) difficult.

T-cell acute lymphoblastic leukemia (T-ALL) is a type of acute lymphoblastic leukemia with aggressive malignant neoplasm of the bone marrow. Acute lymphoblastic leukemia (ALL) is a condition where immature white blood cells accumulate in the bone marrow, subsequently crowding out normal white blood cells and create build-up in the liver, spleen, and lymph nodes. The two most common types of ALL are B-lymphocytes and T-lymphocytes, where the first protects the body against viruses and bacteria through antibody production which can directly destroy target cells or trigger others to do so, whilst the latter directly destroy bacteria or cells infected with viruses. Approximately 20% of all ALL patients are categorized specifically to suffer from T-ALL and it is seen to be more prevalent in the adult population in comparison to children, with incidences shown to diminish with age. Amongst T-ALL cases in the pediatric population, a median onset of age 9 has been identified and the disease is particularly prominent amongst adolescents. The disease stems from cytogenic and molecular abnormalities, resulting in disruption of developmental pathways controlling thymocyte development, tumor suppressor development, and alterations in control of cell growth and proliferation. Distinct from adult T-cell leukemia where T-cell lymphotropic virus Type I causes malignant maturation of T-cells, T-ALL is a precursor for lymphoid neoplasm. Its clinical presentation most commonly includes infiltration of the central nervous system (CNS), and further identifies mediastinal mass presence originating from the thymus, along with extramedullary involvement of multiple organs including the lymph node as a result of hyperleukocytosis.












