Related Research Articles

A thermophile is an organism—a type of extremophile—that thrives at relatively high temperatures, between 41 and 122 °C. Many thermophiles are archaea, though some of them are bacteria and fungi. Thermophilic eubacteria are suggested to have been among the earliest bacteria.
DNA primase is an enzyme involved in the replication of DNA and is a type of RNA polymerase. Primase catalyzes the synthesis of a short RNA segment called a primer complementary to a ssDNA template. After this elongation, the RNA piece is removed by a 5' to 3' exonuclease and refilled with DNA.

Cytochromes P450 are a superfamily of enzymes containing heme as a cofactor that mostly, but not exclusively, function as monooxygenases. In mammals, these proteins oxidize steroids, fatty acids, and xenobiotics, and are important for the clearance of various compounds, as well as for hormone synthesis and breakdown. In 1963, Estabrook, Cooper, and Rosenthal described the role of CYP as a catalyst in steroid hormone synthesis and drug metabolism. In plants, these proteins are important for the biosynthesis of defensive compounds, fatty acids, and hormones.
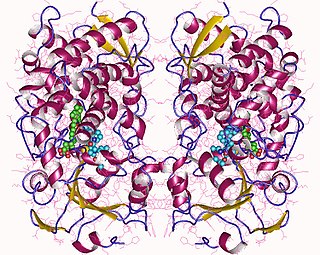
Cytochrome P450 3A4 is an important enzyme in the body, mainly found in the liver and in the intestine. It oxidizes small foreign organic molecules (xenobiotics), such as toxins or drugs, so that they can be removed from the body. It is highly homologous to CYP3A5, another important CYP3A enzyme.
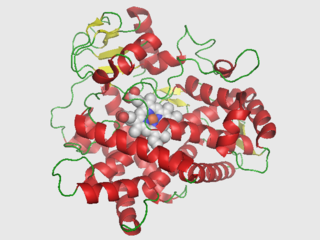
Cytochrome P450 2D6 (CYP2D6) is an enzyme that in humans is encoded by the CYP2D6 gene. CYP2D6 is primarily expressed in the liver. It is also highly expressed in areas of the central nervous system, including the substantia nigra.

Cytochrome P450 2E1 is a member of the cytochrome P450 mixed-function oxidase system, which is involved in the metabolism of xenobiotics in the body. This class of enzymes is divided up into a number of subcategories, including CYP1, CYP2, and CYP3, which as a group are largely responsible for the breakdown of foreign compounds in mammals.

Sulfolobus is a genus of microorganism in the family Sulfolobaceae. It belongs to the archaea domain.

Cytochrome P450 2A6 is a member of the cytochrome P450 mixed-function oxidase system, which is involved in the metabolism of xenobiotics in the body. CYP2A6 is the primary enzyme responsible for the oxidation of nicotine and cotinine. It is also involved in the metabolism of several pharmaceuticals, carcinogens, and a number of coumarin-type alkaloids. CYP2A6 is the only enzyme in the human body that appreciably catalyzes the 7-hydroxylation of coumarin, such that the formation of the product of this reaction, 7-hydroxycoumarin, is used as a probe for CYP2A6 activity.

A thermoacidophile is an extremophilic microorganism that is both thermophilic and acidophilic; i.e., it can grow under conditions of high temperature and low pH. The large majority of thermoacidophiles are archaea or bacteria, though occasional eukaryotic examples have been reported. Thermoacidophiles can be found in hot springs and solfataric environments, within deep sea vents, or in other environments of geothermal activity. They also occur in polluted environments, such as in acid mine drainage.
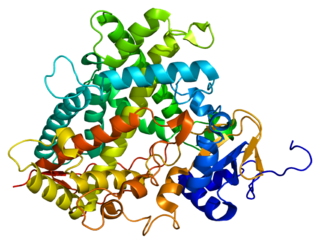
Cytochrome P450 1A2, a member of the cytochrome P450 mixed-function oxidase system, is involved in the metabolism of xenobiotics in the human body. In humans, the CYP1A2 enzyme is encoded by the CYP1A2 gene.

Cytochrome P450 17A1 is an enzyme of the hydroxylase type that in humans is encoded by the CYP17A1 gene on chromosome 10. It is ubiquitously expressed in many tissues and cell types, including the zona reticularis and zona fasciculata of the adrenal cortex as well as gonadal tissues. It has both 17α-hydroxylase and 17,20-lyase activities, and is a key enzyme in the steroidogenic pathway that produces progestins, mineralocorticoids, glucocorticoids, androgens, and estrogens. More specifically, the enzyme acts upon pregnenolone and progesterone to add a hydroxyl (-OH) group at carbon 17 position (C17) of the steroid D ring, or acts upon 17α-hydroxyprogesterone and 17α-hydroxypregnenolone to split the side-chain off the steroid nucleus.

Steroid 21-hydroxylase is an enzyme that hydroxylates steroids at the C21 position and is involved in biosynthesis of aldosterone and cortisol. The enzyme converts progesterone and 17α-hydroxyprogesterone into 11-deoxycorticosterone and 11-deoxycortisol, respectively, within metabolic pathways that ultimately lead to aldosterone and cortisol. Deficiency in the enzyme may cause congenital adrenal hyperplasia.

Steroid 11β-hydroxylase, also known as steroid 11β-monooxygenase, is a steroid hydroxylase found in the zona glomerulosa and zona fasciculata of the adrenal cortex. Named officially the cytochrome P450 11B1, mitochondrial, it is a protein that in humans is encoded by the CYP11B1 gene. The enzyme is involved in the biosynthesis of adrenal corticosteroids by catalyzing the addition of hydroxyl groups during oxidation reactions.

Sulfolobaceae are a family of the Sulfolobales belonging to the domain Archaea. The family consists of several genera adapted to survive environmental niches with extreme temperature and low pH conditions.
Icerudivirus is a genus of viruses in the family Rudiviridae. These viruses are non-enveloped, stiff-rod-shaped viruses with linear dsDNA genomes, that infect hyperthermophilic archaea of the species Sulfolobus islandicus. There are three species in the genus.

Sulfolobus turreted icosahedral virus 1 is a species of virus that infects the archaeon Sulfolobus solfataricus.
Hexaprenyl diphosphate synthase is an enzyme with systematic name geranylgeranyl-diphosphate:isopentenyl-diphosphate transferase . This enzyme catalyses the following chemical reaction
Saccharolobus solfataricus is a species of thermophilic archaeon. It was transferred from the genus Sulfolobus to the new genus Saccharolobus with the description of Saccharolobus caldissimus in 2018.
Saccharolobus shibatae is an archaeal species belongs to the phylum Thermoproteota. Saccharolobus shibatae was described for the first time as Sulfolobus shibatae in 1990, after being isolated from geothermal pools in Beppu, Japan. It was transferred from the genus Sulfolobus to the new genus Saccharolobus with the description of Saccharolobus caldissimus in 2018.
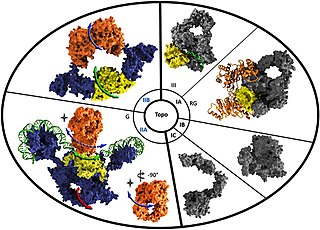
Reverse gyrase is a type I topoisomerase that introduces positive supercoils into DNA, contrary to the typical negative supercoils introduced by the type II topoisomerase DNA gyrase. These positive supercoils can be introduced to DNA that is either negatively supercoiled or fully relaxed. Where DNA gyrase forms a tetramer and is capable of cleaving a double-stranded region of DNA, reverse gyrase can only cleave single stranded DNA. More specifically, reverse gyrase is a member of the type IA topoisomerase class; along with the ability to relax negatively or positively supercoiled DNA, type IA enzymes also tend to have RNA-topoisomerase activities. These RNA topoisomerases help keep longer RNA strands from becoming tangled in what are referred to as "pseudoknots." Due to their ability to interact with RNA, it is thought that this is one of the most ancient class of enzymes found to date.
References
- ↑ Wright RL, Harris K, Solow B, White RH, Kennelly PJ (April 1996). "Cloning of a potential cytochrome P450 from the archaeon Sulfolobus solfataricus". FEBS Letters. 384 (3): 235–9. doi: 10.1016/0014-5793(96)00322-5 . PMID 8617361.
- ↑ Suzuki R, Hirakawa H, Nagamune T (December 2014). "Electron donation to an archaeal cytochrome P450 is enhanced by PCNA-mediated selective complex formation with foreign redox proteins". Biotechnology Journal. 9 (12): 1573–81. doi: 10.1002/biot.201400007 . PMID 24924478. S2CID 5231123.