Examples
The stereoisomers β-D-glucopyranose and β-D-mannopyranose are epimers because they differ only in the stereochemistry at the C-2 position. The hydroxy group in β-D-glucopyranose is equatorial (in the "plane" of the ring), while in β-D-mannopyranose the C-2 hydroxy group is axial (up from the "plane" of the ring). These two molecules are epimers but, because they are not mirror images of each other, are not enantiomers. (Enantiomers have the same name, but differ in D and L classification.) They are also not sugar anomers, since it is not the anomeric carbon involved in the stereochemistry. Similarly, β-D-glucopyranose and β-D-galactopyranose are epimers that differ at the C-4 position, with the former being equatorial and the latter being axial.
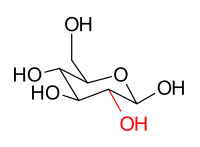 |  |
β-D-glucopyranose | β-D-mannopyranose |
In the case that the difference is the -OH groups on C-1, the anomeric carbon, such as in the case of α-D-glucopyranose and β-D-glucopyranose, the molecules are both epimers and anomers (as indicated by the α and β designation). [3]
 |  |
α-D-glucopyranose | β-D-glucopyranose |
Other closely related compounds are epi-inositol and inositol, and lipoxin and epilipoxin.
 | 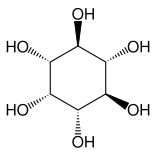 |  |  |
epi-inositol | Inositol | Lipoxin | Epilipoxin |
Doxorubicin and epirubicin are two epimers that are used as drugs.
 |
Doxorubicin–epirubicin comparison |