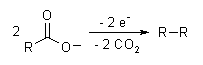Mechanism and side-reactions
The reaction mechanism involves a two-stage radical process: electrochemical oxidation first gives a alkylcarboxyl radical, which decarboxylates almost immediately to give an alkyl radical intermediate. The alkyl radicals which combine to form a covalent bond. [2] As an example, electrolysis of acetic acid yields ethane and carbon dioxide:
- CH3COOH → CH3COO− → CH3COO· → CH3· + CO2
- 2CH3· → CH3CH3
Another example is the synthesis of 2,7-dimethyl-2,7-dinitrooctane from 4-methyl-4-nitrovaleric acid: [3]
-

Other compounds can trap the radicals formed by decarboxylation, and the Kolbe reaction has also been occasionally used in cross-coupling reactions. If a mixture of two different carboxylates are used, the radical cross-coupling reaction generally gives all combinations of them: [4]
- R1COO− + R2COO− → R1−R1 and/or R1−R2 and/or R2−R2
The reaction process can be enhanced and the Hofer–Moest reaction alternative suppressed, by performing the reaction under weakly acidic conditions in protic solvents, and using a high current density and a platinum anodic electrode. [4]
In 2022, it was discovered that the Kolbe electrolysis is enhanced if an alternating square wave current is used instead of a direct current. [5] [6]
Hofer–Moest reaction
In the Hofer–Moest reaction, the alkyl radical undergo further oxidation to form a carbocation, rather than coupling with another alkyl radical, which then reacts with an available nucleophile. [7] The Hofer–Moest reaction, rather than Kolbe radical-coupling, always occurs if the carboxylic acid bears a carbocation-stabilizing side-substituent at the α position, but only sometimes otherwise. [4]
-

This page is based on this
Wikipedia article Text is available under the
CC BY-SA 4.0 license; additional terms may apply.
Images, videos and audio are available under their respective licenses.