
A dye is a colored substance that chemically bonds to the substrate to which it is being applied. This distinguishes dyes from pigments which do not chemically bind to the material they color. Dye is generally applied in an aqueous solution, and may require a mordant to improve the fastness of the dye on the fiber.

Indigo is a deep color close to the color wheel blue, as well as to some variants of ultramarine, based on the ancient dye of the same name. The word "indigo" comes from the Latin word indicum, meaning "Indian", as the dye was originally exported to Europe from India.

Violet is the color of light at the short wavelength end of the visible spectrum, between blue and invisible ultraviolet. It is one of the seven colors that Isaac Newton labeled when dividing the spectrum of visible light in 1672. Violet light has a wavelength between approximately 380 and 435 nanometers. The color's name is derived from the violet flower.

Purple is any of a variety of colors with hue between red and blue. In the RGB color model used in computer and television screens, purples are produced by mixing red and blue light. In the RYB color model historically used by painters, purples are created with a combination of red and blue pigments. In the CMYK color model used in printing, purples are made by combining magenta pigment with either cyan pigment, black pigment, or both.

A pigment is a colored material that is completely or nearly insoluble in water. In contrast, dyes are typically soluble, at least at some stage in their use. Generally dyes are often organic compounds whereas pigments are often inorganic compounds. Pigments of prehistoric and historic value include ochre, charcoal, and lapis lazuli.

Indigo dye is an organic compound with a distinctive blue color. Historically, indigo was a natural dye extracted from the leaves of some plants of the Indigofera genus, in particular Indigofera tinctoria; dye-bearing Indigofera plants were commonly grown and used throughout the world, in Asia in particular, as an important crop, with the production of indigo dyestuff economically important due to the previous rarity of some blue dyestuffs historically.
Tyrian purple, also known as Phoenician red, Phoenician purple, royal purple, imperial purple, or imperial dye, is a reddish-purple natural dye. The name Tyrian refers to Tyre, Lebanon. It is secreted by several species of predatory sea snails in the family Muricidae, rock snails originally known by the name 'Murex'. In ancient times, extracting this dye involved tens of thousands of snails and substantial labor, and as a result, the dye was highly valued. The colored compound is 6,6′-dibromoindigo.
A lake pigment is a pigment made by precipitating a dye with an inert binder, or mordant, usually a metallic salt. Unlike vermilion, ultramarine, and other pigments made from ground minerals, lake pigments are organic. Manufacturers and suppliers to artists and industry frequently omit the lake designation in the name. Many lake pigments are fugitive because the dyes involved are not lightfast. Red lakes were particularly important in Renaissance and Baroque paintings; they were often used as translucent glazes to portray the colors of rich fabrics and draperies.

Azo compounds are compounds bearing the functional group diazenyl.
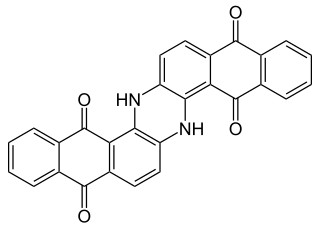
Indanthrone blue, also called indanthrene, is an organic compound with the formula (C14H6O2NH)2. It is a dark blue solid that is a common dye as well as a precursor to other dyes.

Azo dyes are organic compounds bearing the functional group R−N=N−R′, in which R and R′ are usually aryl. They are a commercially important family of azo compounds, i.e. compounds containing the linkage C-N=N-C. Azo dyes are widely used to treat textiles, leather articles, and some foods. Chemically related to azo dyes are azo pigments, which are insoluble in water and other solvents.

Colored smoke is a kind of smoke created by an aerosol of small particles of a suitable pigment or dye.
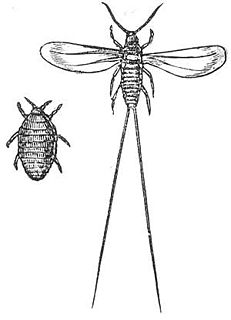
The cochineal (, , is a scale insect in the suborder Sternorrhyncha, from which the natural dye carmine is derived. A primarily sessile parasite native to tropical and subtropical South America through North America, this insect lives on cacti in the genus Opuntia, feeding on plant moisture and nutrients. The insects are found on the pads of prickly pear cacti, collected by brushing them off the plants, and dried.

Alizarin crimson is a shade of red that is biased slightly more towards purple than towards orange on the color wheel and has a blue undertone. It is named after the organic dye alizarin, found in the madder plant, and the related synthetic lake pigment alizarin crimson. William Henry Perkin had co-discovered a way to synthesize the pigment alizarin, which became known as the color alizarin crimson. Its consistency and lightfastness quickly made it a favourite red pigment for artists.

Anthraquinone dyes are an abundant group of dyes comprising a anthraquinone unit as the shared structural element. Anthraquinone itself is colourless, but red to blue dyes are obtained by introducing electron donor groups such as hydroxy or amino groups in the 1-, 4-, 5- or 8-position. Anthraquinone dyestuffs are structurally related to indigo dyestuffs and are classified together with these in the group of carbonyl dyes.

Vat Yellow 1 is a vat dye, yellow in appearance under some conditions used in cloth dyeing. Its synonyms are flavanthrone and Indofast Yellow, and it is in the class of anthraquinone-type compounds.

Pigment Violet 29 is an organic compound that is used as a pigment and vat dye. Its colour is dark red purple, or bordeaux.

Violanthrone, also known as dibenzanthrone, is an organic compound that serves as a vat dye and a precursor to other vat dyes. X-ray crystallography confirms that the molecule is planar with C2v symmetry. Isomeric with violanthrone is isoviolanthrone, which has a centrosymmetric structure.
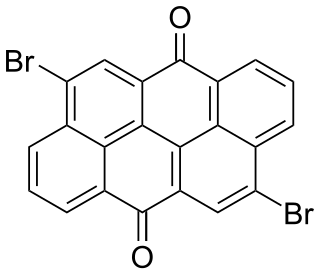
Dibromoanthanthrone is a scarlet or orange-red-hue synthetic organic colourant.

A colorant is any substance that changes the spectral transmittance or reflectance of a material. Synthetic colorants are those created in a laboratory or industrial setting. The production and improvement of colorants was a driver of the early synthetic chemical industry, in fact many of today's largest chemical producers started as dye-works in the late 19th or early 20th centuries, including Bayer AG(1863). Synthetics are extremely attractive for industrial and aesthetic purposes as they have they often achieve higher intensity and color fastness than comparable natural pigments and dyes used since ancient times. Market viable large scale production of dyes occurred nearly simultaneously in the early major producing countries Britain (1857), France (1858), Germany (1858), and Switzerland (1859), and expansion of associated chemical industries followed. The mid-nineteenth century through WWII saw an incredible expansion of the variety and scale of manufacture of synthetic colorants. Synthetic colorants quickly became ubiquitous in everyday life, from clothing to food. This stems from the invention of industrial research and development laboratories in the 1870s, and the new awareness of empirical chemical formulas as targets for synthesis by academic chemists. The dye industry became one of the first instances where directed scientific research lead to new products, and the first where this occurred regularly.


















