
Osmotic pressure is the minimum pressure which needs to be applied to a solution to prevent the inward flow of its pure solvent across a semipermeable membrane. It is also defined as the measure of the tendency of a solution to take in its pure solvent by osmosis. Potential osmotic pressure is the maximum osmotic pressure that could develop in a solution if it were separated from its pure solvent by a semipermeable membrane

Brine is water with a high-concentration solution of salt. In diverse contexts, brine may refer to the salt solutions ranging from about 3.5% up to about 26%. Brine forms naturally due to evaporation of ground saline water but it is also generated in the mining of sodium chloride. Brine is used for food processing and cooking, for de-icing of roads and other structures, and in a number of technological processes. It is also a by-product of many industrial processes, such as desalination, so it requires wastewater treatment for proper disposal or further utilization.

Desalination is a process that removes mineral components from saline water. More generally, desalination is the removal of salts and minerals from a substance. One example is soil desalination. This is important for agriculture. It is possible to desalinate saltwater, especially sea water, to produce water for human consumption or irrigation. The by-product of the desalination process is brine. Many seagoing ships and submarines use desalination. Modern interest in desalination mostly focuses on cost-effective provision of fresh water for human use. Along with recycled wastewater, it is one of the few water resources independent of rainfall.
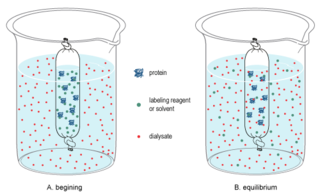
In chemistry, dialysis is the process of separating molecules in solution by the difference in their rates of diffusion through a semipermeable membrane, such as dialysis tubing.

Forward osmosis (FO) is an osmotic process that, like reverse osmosis (RO), uses a semi-permeable membrane to effect separation of water from dissolved solutes. The driving force for this separation is an osmotic pressure gradient, such that a "draw" solution of high concentration, is used to induce a net flow of water through the membrane into the draw solution, thus effectively separating the feed water from its solutes. In contrast, the reverse osmosis process uses hydraulic pressure as the driving force for separation, which serves to counteract the osmotic pressure gradient that would otherwise favor water flux from the permeate to the feed. Hence significantly more energy is required for reverse osmosis compared to forward osmosis.
Water potential is the potential energy of water per unit volume relative to pure water in reference conditions. Water potential quantifies the tendency of water to move from one area to another due to osmosis, gravity, mechanical pressure and matrix effects such as capillary action. The concept of water potential has proved useful in understanding and computing water movement within plants, animals, and soil. Water potential is typically expressed in potential energy per unit volume and very often is represented by the Greek letter ψ.
Electrodialysis reversal (EDR) is an electrodialysis reversal water desalination membrane process that has been commercially used since the early 1960s. An electric current migrates dissolved salt ions, including fluorides, nitrates and sulfates, through an electrodialysis stack consisting of alternating layers of cationic and anionic ion exchange membranes. Periodically, the direction of ion flow is reversed by reversing the polarity of the applied electric current.
Solar desalination is a desalination technique powered by solar energy. The two common methods are direct (thermal) and indirect (photovoltaic).

In chemical biology, tonicity is a measure of the effective osmotic pressure gradient; the water potential of two solutions separated by a partially-permeable cell membrane. Tonicity depends on the relative concentration of selective membrane-impermeable solutes across a cell membrane which determine the direction and extent of osmotic flux. It is commonly used when describing the swelling-versus-shrinking response of cells immersed in an external solution.

Electrodialysis (ED) is used to transport salt ions from one solution through ion-exchange membranes to another solution under the influence of an applied electric potential difference. This is done in a configuration called an electrodialysis cell. The cell consists of a feed (dilute) compartment and a concentrate (brine) compartment formed by an anion exchange membrane and a cation exchange membrane placed between two electrodes. In almost all practical electrodialysis processes, multiple electrodialysis cells are arranged into a configuration called an electrodialysis stack, with alternating anion and cation-exchange membranes forming the multiple electrodialysis cells. Electrodialysis processes are different from distillation techniques and other membrane based processes in that dissolved species are moved away from the feed stream, whereas other processes move away the water from the remaining substances. Because the quantity of dissolved species in the feed stream is far less than that of the fluid, electrodialysis offers the practical advantage of much higher feed recovery in many applications.

Osmotic power, salinity gradient power or blue energy is the energy available from the difference in the salt concentration between seawater and river water. Two practical methods for this are reverse electrodialysis (RED) and pressure retarded osmosis (PRO). Both processes rely on osmosis with membranes. The key waste product is brackish water. This byproduct is the result of natural forces that are being harnessed: the flow of fresh water into seas that are made up of salt water.
A solar-powered desalination unit produces potable water from saline water through direct or indirect methods of desalination powered by sunlight. Solar energy is the most promising renewable energy source due to its ability to drive the more popular thermal desalination systems directly through solar collectors and to drive physical and chemical desalination systems indirectly through photovoltaic cells.
Reverse osmosis (RO) is a water purification process that uses a semi-permeable membrane to separate water molecules from other substances. RO applies pressure to overcome osmotic pressure that favors even distributions. RO can remove dissolved or suspended chemical species as well as biological substances, and is used in industrial processes and the production of potable water. RO retains the solute on the pressurized side of the membrane and the purified solvent passes to the other side. It relies on the relative sizes of the various molecules to decide what passes through. "Selective" membranes reject large molecules, while accepting smaller molecules.

Osmosis is the spontaneous net movement or diffusion of solvent molecules through a selectively-permeable membrane from a region of high water potential to a region of low water potential, in the direction that tends to equalize the solute concentrations on the two sides. It may also be used to describe a physical process in which any solvent moves across a selectively permeable membrane separating two solutions of different concentrations. Osmosis can be made to do work. Osmotic pressure is defined as the external pressure required to be applied so that there is no net movement of solvent across the membrane. Osmotic pressure is a colligative property, meaning that the osmotic pressure depends on the molar concentration of the solute but not on its identity.
Osmoregulation is the active regulation of the osmotic pressure of an organism's body fluids, detected by osmoreceptors, to maintain the homeostasis of the organism's water content; that is, it maintains the fluid balance and the concentration of electrolytes to keep the body fluids from becoming too diluted or concentrated. Osmotic pressure is a measure of the tendency of water to move into one solution from another by osmosis. The higher the osmotic pressure of a solution, the more water tends to move into it. Pressure must be exerted on the hypertonic side of a selectively permeable membrane to prevent diffusion of water by osmosis from the side containing pure water.

Pressure retarded osmosis (PRO) is a technique to separate a solvent from a solution that is more concentrated and also pressurized. A semipermeable membrane allows the solvent to pass to the concentrated solution side by osmosis. The technique can be used to generate power from the salinity gradient energy resulting from the difference in the salt concentration between sea and river water.

Marine energy or marine power refers to the energy carried by ocean waves, tides, salinity, and ocean temperature differences. The movement of water in the world's oceans creates a vast store of kinetic energy, or energy in motion. Some of this energy can be harnessed to generate electricity to power homes, transport and industries.
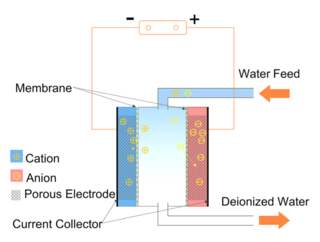
Capacitive deionization (CDI) is a technology to deionize water by applying an electrical potential difference over two electrodes, which are often made of porous carbon. In other words, CDI is an electro-sorption method using a combination of a sorption media and an electrical field to separate ions and charged particles. Anions, ions with a negative charge, are removed from the water and are stored in the positively polarized electrode. Likewise, cations are stored in the cathode, which is the negatively polarized electrode.

Australia is the driest habitable continent on Earth and its installed desalination capacity has been increasing. Until a few decades ago, Australia met its demands for water by drawing freshwater from dams and water catchments. As a result of the water supply crisis during the severe 1997–2009 drought, state governments began building desalination plants that purify seawater using reverse osmosis technology. Approximately one percent of the world's drinkable water originates from desalination plants.
Osmosis is the movement of molecules through a membrane.














