
An oxidizing agent is a substance in a redox chemical reaction that gains or "accepts"/"receives" an electron from a reducing agent. In other words, an oxidizer is any substance that oxidizes another substance. The oxidation state, which describes the degree of loss of electrons, of the oxidizer decreases while that of the reductant increases; this is expressed by saying that oxidizers "undergo reduction" and "are reduced" while reducers "undergo oxidation" and "are oxidized". Common oxidizing agents are oxygen, hydrogen peroxide, and the halogens.

Potassium permanganate is an inorganic compound with the chemical formula KMnO4. It is a purplish-black crystalline salt, that dissolves in water as K+ and MnO−
4, an intensely pink to purple solution.

A permanganate is a chemical compound with the manganate(VII) ion, MnO−
4, the conjugate base of permanganic acid. Because the manganese atom has a +7 oxidation state, the permanganate(VII) ion is a strong oxidising agent. The ion is a transition metal ion with a tetrahedral structure. Permanganate solutions are purple in colour and are stable in neutral or slightly alkaline media. The exact chemical reaction depends on the carbon-containing reactants present and the oxidant used. For example, trichloroethane (C2H3Cl3) is oxidised by permanganate ions to form carbon dioxide (CO2), manganese dioxide (MnO2), hydrogen ions (H+), and chloride ions (Cl−).

In inorganic nomenclature, a manganate is any negatively charged molecular entity with manganese as the central atom. However, the name is usually used to refer to the tetraoxidomanganate(2−) anion, MnO2−
4, also known as manganate(VI) because it contains manganese in the +6 oxidation state. Manganates are the only known manganese(VI) compounds.

Sodium manganate is the inorganic compound with the formula Na2Mn O4. This deep green solid is a rarely encountered analogue of the related salt K2MnO4. Sodium manganate is rare because it cannot be readily prepared from the oxidation of manganese dioxide and sodium hydroxide. Instead this oxidation reaction tends to stop at producing sodium hypomanganate, Na3MnO4, and even this Mn(V) salt is unstable in solution. Sodium manganate can be produced by reduction of sodium permanganate under basic conditions:
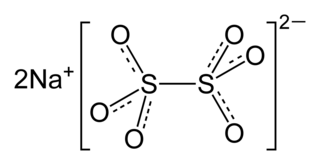
Sodium dithionate Na2S2O6 is an important compound for inorganic chemistry. It is also known under names disodium dithionate, sodium hyposulfate, and sodium metabisulfate. The sulfur can be considered to be in its +5 oxidation state.
Permanganic acid (or manganic(VII) acid) is the inorganic compound with the formula HMnO4. This strong oxoacid has been isolated as its dihydrate. It is the conjugate acid of permanganate salts. It is the subject of few publications and its characterization as well as its uses are very limited.

Sodium pertechnetate is the inorganic compound with the formula NaTcO4. This colourless salt contains the pertechnetate anion, TcO−
4 that has slightly distorted tetrahedron symmetry both at 296 K and at 100 K while the coordination polyhedron of the sodium cation is different from typical for scheelite structure. The radioactive 99m
Tc
O−
4 anion is an important radiopharmaceutical for diagnostic use. The advantages to 99m
Tc
include its short half-life of 6 hours and the low radiation exposure to the patient, which allow a patient to be injected with activities of more than 30 millicuries. Na[99m
Tc
O
4] is a precursor to a variety of derivatives that are used to image different parts of the body.
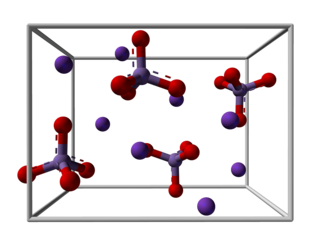
Potassium manganate is the inorganic compound with the formula K2MnO4. This green-colored salt is an intermediate in the industrial synthesis of potassium permanganate, a common chemical. Occasionally, potassium manganate and potassium permanganate are confused, but each compound's properties are distinct.
In chemistry, hypomanganate, also called manganate(V) or tetraoxidomanganate(3−), is a trivalent anion (negative ion) composed of manganese and oxygen, with formula MnO3−
4.
Potassium hypomanganate is the inorganic compound with the formula K3MnO4. Also known as potassium manganate(V), this bright blue solid is a rare example of a salt with the hypomanganate or manganate(V) anion, where the manganese atom is in the +5 oxidation state. It is an intermediate in the production of potassium permanganate and the industrially most important Mn(V) compound.

Sodium permanganate is the inorganic compound with the formula NaMnO4. It is closely related to the more commonly encountered potassium permanganate, but it is generally less desirable, because it is more expensive to produce. It is mainly available as the monohydrate. This salt absorbs water from the atmosphere and has a low melting point. Being about 15 times more soluble than KMnO4, sodium permanganate finds some applications where very high concentrations of MnO4− are sought.

Ammonium permanganate is the chemical compound NH4MnO4, or NH3·HMnO4. It is a water soluble, violet-brown or dark purple salt.

Barium manganate is an inorganic compound with the formula BaMnO4. It is used as an oxidant in organic chemistry. It belongs to a class of compounds known as manganates in which the manganese resides in a +6 oxidation state. Manganate should not be confused with permanganate which contains manganese(VII). Barium manganate is a powerful oxidant, popular in organic synthesis and can be used in a wide variety of oxidation reactions.
Barium permanganate is a chemical compound, with the formula Ba(MnO4)2. It forms violet to brown crystals that are sparingly soluble in water.
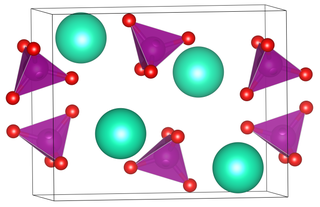
Rubidium permanganate is the permanganate salt of rubidium, with the chemical formula RbMnO
4.

Caesium permanganate is the permanganate salt of caesium, with the chemical formula CsMnO4.
Magnesium permanganate is an inorganic compound with the chemical formula Mg(MnO4)2. It can be used as an oxidant.
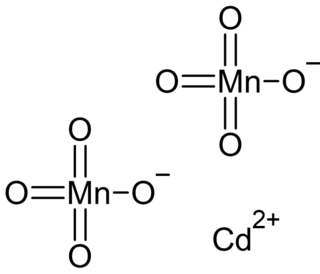
Cadmium permanganate is an inorganic compound with the chemical formula Cd(MnO4)2. It can form the hexahydrate Cd(MnO4)2·6H2O.














